Review Article - International Journal of Clinical Rheumatology (2019) Volume 14, Issue 5
The evidence for dietary manipulation in inflammatory arthritis
- Corresponding Author:
- Hannah Jethwa
Department of Rheumatology
Cambridge University Hospitals NHS Foundation Trust, UK
E-mail: hannahjethwa@nhs.net
Abstract
Over the past few decades there has been an increasing interest in the role that diet plays on the development and clinical manifestations of inflammatory arthritis. Although much remains to be elucidated, prospective cohort studies reviewing the relation of diet and rheumatoid arthritis development are suggestive that dietary components may play a fundamental role. Furthermore, data on microbiome dysbiosis in the pathogenesis of inflammatory disease further adds strength to the thought that dietary manipulation in patients may have ameliorative effects on their inflammatory disease; it may be that dietary alterations in patients at risk of inflammatory arthritis may reduce the development of clinical disease. In this review, we discuss the role that diet may play in the pathogenesis of inflammatory arthritis and highlight the clinical evidence available on dietary manipulation for the amelioration of disease activity.
Keywords
diet, inflammatory arthritis, microbiome
Introduction
“Let food be thy medicine and medicine be thy food” – Hippocrates, 431 BC.
The concept that dietary intake has a significant effect on human health and disease dates back many centuries, however focus on this area of research has been relatively limited until recent decades. The mechanisms by which dietary constituents result in inflammatory arthritis remain to be elucidated, however microbiome dysbiosis and diet-related immune complex formation have been suggested.
Microbiome dysbiosis
Microbiome dysbiosis in rheumatoid arthritis
It is well known that patients with Rheumatoid Arthritis (RA) have an increased incidence of periodontal disease and research has identified the presence of high levels of antibodies against the DNA of bacteria implicated in periodontal disease in the serum and synovial fluid of these patients [1]. Furthermore, metagenomics analysis of gut microbiota in patients with RA have demonstrated notable differences compared to healthy volunteers and studies in germ-free mice have further implicated the role of the microbiome in autoimmune disease [1]. These data are suggestive that autoimmunity may be triggered in the gut and the fact that many patient’s positive RA-associated antibodies prior to the onset of inflammatory joint disease further suggests that autoimmunity may not triggered in the joints themselves.
One theory for how gut bacteria result in autoimmunity is that increased permeability of the gut wall lumen exposes the immune system to commensal microorganisms which would usually not result in disease [1]. Pianta et al. recently identified two HLA-DR-presented auto-antigens with high sequence homology to gut microbes, such as Prevotella sp., which result in both T- and B-cell responses in patients with RA, suggesting the role of molecular mimicry in pathogenesis [2]. It may be, therefore, that dietary manipulation, probiotics or targeted antibiotic therapy in patients either at high risk of the development of RA or in those who demonstrate seropositivity without joint inflammation may reduce the likelihood of clinical disease.
Microbiome dysbiosis in spondyloarthritis
The microbiome is also thought to be key in the pathogenesis of spondyloarthropathy. Hammer et al. developed a strain of human HLA-B27 transgenic rats that develop spondyloarthritis associated with intestinal inflammation; interestingly, germ-free rats did not develop inflammatory features until they were exposed to specific pathogen-free enteric bacteria [3]. Furthermore, a mouse model initially developed to mimic rheumatoid arthritis developed spondyloarthritis-like disease involving ankles, feet and Achilles tendons, plantar fasciitis, spondylitis and ileitis following exposure to curdlan (a β-1,3-glucan derived from yeast, fungal and bacterial cell walls) [4]. The CD4+ T cells in these mice were demonstrated to be initiated via a IL-23 dependent pathway, resulting in production of IL-1, IL-6 and TNFα, all of which are fundamental in spondyloarthritis pathogenesis [4].
The presence of subclinical klebsiella infections has also been proposed in AS pathogenesis. This is supported by studies demonstrating that rabbits immunised with HLA-B27- positive lymphocytes demonstrate increased haemaggluninating activity against red cells coated with klebsiella lipopolysaccharide, anti- HLA-B27 monoclonal antibodies bind to various bacteria including klebsiella and a homologous amino acid sequence present in HLA-B27 has molecular similarity to another sequence within klebsiella pneumonia nitrogenase reductase [5]. Furthermore, high levels of antibodies against klebsiella have been demonstrated in serum of patients with AS and antibodies to klebsiella nitrogenase reductase peptides have been shown to bine preferentially to synovial tissues in these patients [5]. Combining these data, molecular mimicry or cross-reactivity of klebsiella peptides are suggested to play a role in AS pathogenesis.
Studies reviewing the impact of the microbiome in psoriatic arthritis remain limited, however it has been hypothesized that individuals with psoriasis who develop arthritis may have an altered skin microbiome composition compared to those with only skin involvement [6]. Furthermore, studies have demonstrated that intestinal dysbiosis can determine the direction of naïve CD4+ T cell differentiation into either effector or regulatory T cells which can lead to chronic inflammatory disease; furthermore, CD4+ and CD8+ T cell expansion has been noted in the peripheral blood and synovium of patients with psoriatic arthritis, highlighting the importance of T cells in disease pathogenesis [6].
Diet-related immune-complex formation
Immune-complex formation and rheumatoid arthritis
Panush et al. studied a patient with RA who allegedly had symptomatic exacerbations of her arthritis following ingestion of meat, milk and beans [7]. Clinical assessment included tender and swollen joint count in addition to subjective and objective disease severity indices. They noted a significant amelioration of her inflammatory symptoms when fasting or when taking a glutenand lactose-free oral supplement compared to when she had a normal balanced diet. Furthermore, following a 33-day gluten- and lactose-free diet, four milk challenges resulted in disease flares; symptomatic deterioration peaked at 24-48 hours’ post-challenge and resolved within 48 hours from onset. Immunologic studies suggested both delayed and immediate cutaneous reactivity to milk; anti-milk IgG levels were mildly elevated, with large amounts of IgG4 anti-milk. IgE antibodies to foods, including milk, were not elevated.
Hvatum et al. [8] measured antibodies to several dietary antigens (α- and β-lactalbumin and casein from milk, cereals, ovalbumin from hen’s eggs, cod fish and pork meat) in serum and jejunal perfusion fluid from 14 patients with RA and 20 healthy controls. The most marked variation between groups was a significant elevation in jejunal fluid total IgM levels in RA patients. Furthermore, jejunal IgA, IgG and IgM activities to almost all tested antigens were at least moderately increased in RA patients compared to controls. Interestingly, median intestinal levels of IgG did not differ between groups, suggesting Non-Steroidal Anti-Inflammatory Drug (NSAID) treatment did not result in any general increase in mucosal permeability of intact proteins.
Three of the five patients who initially had raised levels of intestinal dietary antibodies were treated with a 16-week course of sulfasalazine; interestingly, antibody levels normalised after treatment [8]. Mechanisms by which sulfasalazine affects the microbiota, however, remain largely unclear, though it is known that colonic bacterial azoreductase enzymes hydrolyze the drug to its active components; eradication of relevant bacteria, for example with specific antibiotic use, has thus been shown to suppress sulfasalazine metabolism [9].
Immune-complex formation and spondyloarthritis
Diet-related immune-complex formation has also been suggested in the pathogenesis of spondyloarthritis. A study by Togrol et al. reviewed the presence of HLA-B27, anti-gliadin and endomysial antibodies in 30 patients with AS and 19 age- and sex-matched controls; positive anti-gliadin antibodies were not demonstrated in the control group but were positive in 36.7% of patients with AS [10]. Only one of these patients, however, demonstrated typical coeliac histology on intestinal biopsy, suggesting the possibility that anti-gliadin antibody positivity may contribute to AS pathogenesis without resulting causing coeliac disease, which may be a result of increased intestinal permeability to microorganisms.
In addition to noting an increased frequency of coeliac disease in patients with PsA, Lindqvist et al. reported a higher prevalence of IgA anti-gliadin, but not anti-endomysial, antibodies in 109 patients with psoriasis and PsA who reported no known food intolerances compared to healthy controls [11]. Furthermore, individuals with antibody positivity demonstrated elevated inflammatory markers and reported longer durations of morning stiffness [11]; this data further suggestive that such antibodies may be relevant in the pathogenesis of spondyloarthopathies and adds to the possibility that a gluten-free diet may have either preventative or ameliorative effects on this group of diseases. A subsequent study by Kia et al. however, identified no statistically significant difference in the prevalence of anti-gliadin antibodies between patients with psoriasis, those with both psoriasis and PsA and healthy controls [12].
Diet and inflammatory arthritis development
Dietary risk factors and rheumatoid arthritis development
Epidemiological studies prompted early hypotheses that geographic variations in RA prevalence may be associated with diet. For example, indigenous populations with a diet high in omega-3 fatty acids from fish have a significantly reduced incidence of RA compared to European populations [13], suggesting a protective effect.
A 1996 population-based case-control study of dietary intake in women identified more specifically that an increased consumption of broiled or baked fish was associated with reduced risk of RA development; intake of other types of fish did not affect disease risk. This association strengthened when analysis was limited to seropositive RA patients [14].
A later prospective study of over 29,000 women aged 55-69 years revealed the use of supplemental zinc and beta-cryptoxanthin to be statistically significant predictors of the development of RA, leading to increased disease risk. Inverse associations with risk of disease development were also noted in those who consumed greater intakes of fruit and cruciferous vegetables, suggesting a protective factor [15].
Giuseppe et al. performed a population-based case-control study of 1726 patients with RA and 3683 controls which suggested a 23% reduction in the odds of RA development in individuals who have high dietary adherence to a Mediterranean diet; the association was stronger amongst men and only noted in seropositive patients [16].
Further prospective studies have also demonstrated a lower risk of RA development in those with a high intake of vitamin C, wholegrains or poultry and increased risk in those who consume large amount of red/ processed meat, refined grains, french fries or desserts [17,18].
Dietary risk factors and spondyloarthritis development
To date there are no prospective cohort studies reviewing dietary risk factors for the development of spondyloarthritis, however it has been suggested that the change in dietary habits from a high protein, low-starch diet to a Westernised high-starch diet among the Inuit population of Alaska and Canada (in addition to HLA-B27 association) may explain the increased incidence of Ankylosing Spondylitis (AS) in this demographic [19].
A questionnaire-based study in individuals with self-reported psoriasis and PsA suggested an increased risk of disease development in individuals with a high intake of omega-3 fatty acids; an inverse risk association was noted in those with high consumption of omega-6 fatty acids and linoleic, however prospective studies with clinician assessments of disease would be required for further validation [20].
Dietary manipulation in inflammatory arthritis
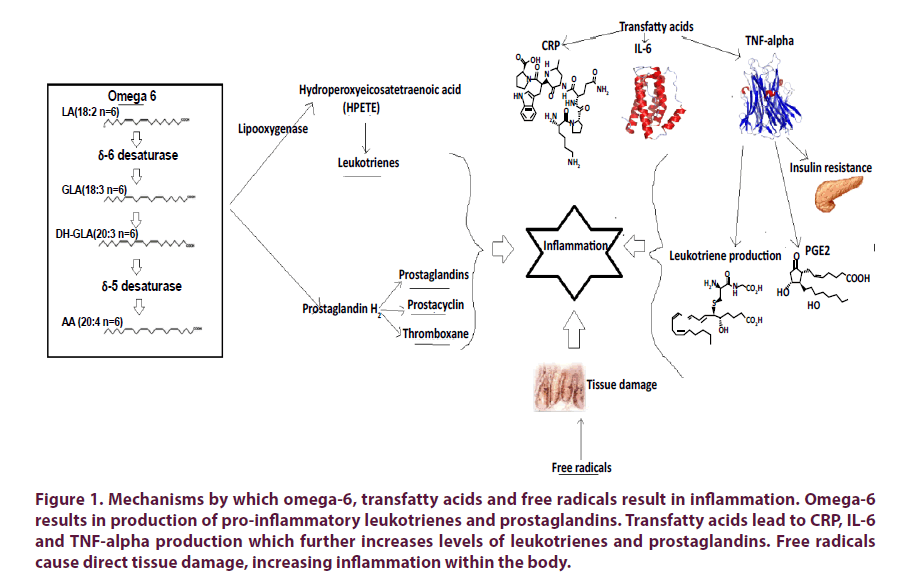
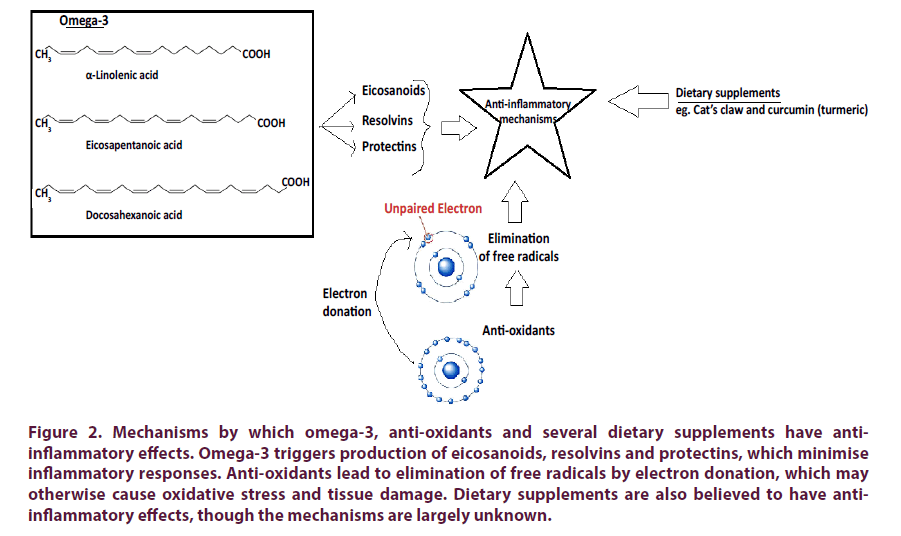
Various support groups for Rheumatoid Arthritis (RA) recommend avoiding foods high in sugar, saturated fat, trans-fat, omega-6 fatty acids and refined carbohydrates and encourage foods rich in unsaturated fats, especially those containing omega-3 fatty acids, vitamin C and anti-oxidants [21-24]. These recommendations were initially based on anecdotal evidence, however more recent studies have identified mechanisms by which these dietary components may result in clinical effects (Figures 1 and 2).
Figure 1: Mechanisms by which omega-6, transfatty acids and free radicals result in inflammation. Omega-6 results in production of pro-inflammatory leukotrienes and prostaglandins. Transfatty acids lead to CRP, IL-6 and TNF-alpha production which further increases levels of leukotrienes and prostaglandins. Free radicals cause direct tissue damage, increasing inflammation within the body.
Figure 2: Mechanisms by which omega-3, anti-oxidants and several dietary supplements have antiinflammatory effects. Omega-3 triggers production of eicosanoids, resolvins and protectins, which minimise inflammatory responses. Anti-oxidants lead to elimination of free radicals by electron donation, which may otherwise cause oxidative stress and tissue damage. Dietary supplements are also believed to have antiinflammatory effects, though the mechanisms are largely unknown.
Clinical data -dietary supplements
Anti-oxidants
Edmonds et al. carried out a prospective placebocontrolled, double-blinded randomised control trial to review the effects of α-tocopherol (a form of vitamin E) on RA disease activity [25]. Forty-two patients were enrolled and divided into either the treatment group (n=20; 600mg α-tocopherol twice daily) or the placebo group (n=22). Clinical efficacy by means of the Ritchie articular index, early morning stiffness and swollen joint count was recorded after 1,4,8 and 12 weeks of treatment [25]. Pain scores, inflammatory markers and assays for oxidative modification were also assessed. Interestingly, although pain scores were reduced in patients in the treatment group, there was no statistical significance between the other parameters recorded. The authors hypothesise the action of α-tocopherol on central nervous pathways, which may be mediated by the suppressive action on nitric oxide (implicated in central pain processing).
A later study in 2003 demonstrated that blood concentrations of anti-oxidants (total-thioles, glutathione and vitamin C) were significantly lower in patients with RA patients compared to healthy controls, while concentrations of malondialdehyde (a marker of oxidative stress) were much higher [26]. The addition of a combined dose of vitamins A, C and E to conventional treatment resulted in a significant increase in total thioles, glutathione and vitamin C compared to patients with RA on conventional treatment alone, along with a decrease in the concentration of malondialdehyde. Furthermore, the treatment group who were given supplemental vitamin A, C and E demonstrated a greater improvement in their disease activity score measured using the Rheumatoid Arthritis Disease Activity Index (RADAI). This data demonstrates a compromised antioxidant defence system in patients with RA and suggests co-administration of such anti-oxidants along with conventional drugs may be beneficial in reducing disease activity as well as long-term tissue damage [26].
A systematic review by Canter et al. assessing the efficacy antioxidants in inflammatory arthritis identified five studies that compared selenium use with placebo [27]. The supplements were tested in combination with other existing medications such as Disease-Modifying Anti-Rheumatic Drugs (DMARDs) or Non-Steroidal Anti- Inflammatory Drugs (NSAIDs). The treatment groups reported a reduction in analgesia use, however no statistically significant difference in clinical outcome measures was noted.
Omega-3 fatty acids
Eicosapentaenoid acid (EPA) and docoshexenoic acid (DHA) are the active constituents of omega-3 fatty acids. Volker et al. carried out a placebo controlled, double blind, randomized 15-week study of 8 patients with RA to assess the efficacy of fish oil-derived 60% omega-3 fatty acid supplementation (40mg/kg) [28]. All patients had a daily omega-6 fatty acid intake of less than 10 g/day. Results indicated a 20% improvement in six of the nine American College of Rheumatology clinical variables (ACR 20) for patients in the active arm at 15 weeks; statistically significant improvements in inflammatory markers or tender joint count, however, was not reported.
A subsequent 24-week randomised control trial of 35 patients with RA divided patients into three groups: treatment group (EPA-i-DHA 840mg/kg daily), a placebo group and a control group [29]. The former two groups were advised to consume less than 12.5g omega-6 fatty acids daily. The group receiving omega-3 supplements demonstrated a reduction in C-reactive protein, however differences in the tender or swollen joint count, self-reported pain score, patient global assessment and modified health assessment questionnaire were not noted amongst the three groups.
An early meta-analysis of 3-month dietary oily fish supplementation demonstrated a statistically significant improvement in tender joint count; an improvement in morning stiffness duration was also noted [30]. A subsequent meta-analysis of 17 randomised clinical trials assessing the use of omega-3 fatty acids as an adjunct to NSAIDs in various patient groups including those with RA demonstrated a statistically significant reduction in joint pain intensity, the number of tender joints, morning stiffness duration and NSAID consumption at 3-4 months [31].
Supplemental use of omega-3 fatty acids has also been reviewed in patients with AS. Sundstrom et al. performed a trial in which 18 patients with AS were randomised to either a low dose (1.95g/day) or a high dose (4.55g/day) omega-3 supplementation; patients in the high dose group exhibited a significant decrease in their Bath Ankylosing Spondylitis Disease Activity Index (BASDAI) compared to those in the low dose group [32].
Cat’s claw
Two of the most commonly used species of Cat’s claw are Uncaria tomentosa and Uncaria guianens. A 24-week randomised double-blind clinical trial including 40 patients with RA demonstrated a significant reduction in painful joints in those using Uncaria tomentosa extract compared to placebo, however overall pain scores, swollen joint count and joint stiffness did not significantly differ between groups [33].
Curcumin (Turmeric)
Multiple studies have demonstrated the ability of curcumin to modulate various cell signalling molecules, for example such as pro-inflammatory cytokines, apoptotic proteins, NF–κB, cyclooxygenase-2, prostaglandin E2, adhesion molecules and phosphorylase kinase [34].
Clinical trials assessing curcumin efficacy on IA are limited, however a pilot study in 45 patients with RA demonstrated the greatest improvement in disease activity scores (DAS, ACR 20, 50 and 70) in those taking 500mg curcumin daily, compared to those on diclofenac sodium (50mg/daily) or on combination therapy [35]. Studies assessing the efficacy of curcumin on spondyloarthritis disease activity have not been published, however it shown modest improvement in those with cutaneous psoriasis [36,37].
Clinical data - dietary changes
Clinical data on drastic dietary changes are limited due to high dropout rates however the efficacy of various diets on disease activity in IA has been reviewed. Beri et al. studied the effects of a basal isocaloric diet free from pulses, cereals, milk and/or non-vegetarian protein food in DMARD-naïve patients with RA [38]. Only 52% of the 27 patients enrolled were able to adhere to the study. Five diets ((1) an isocaloric diet consisting of fruit, vegetables, sugar and refined oil, (2) diet 1 plus pulses, (3a) diet 1 plus wheat and wheat products, (3b) diet 1 plus rice and rice products, (4) diet 1 plus milk and milk products and (5) diet 1 plus eggs, meat, fish and chicken) were prescribed in succession for two weeks each and clinical assessment (including morning stiffness, joint pain score, articular index, patient global assessment and ESR) was recorded prior to each dietary alteration. A significant clinical improvement was noted in 10 patients (71%). One responded to elimination of pulses alone, four to elimination of wheat and rice, two to elimination of rice and rice products; three patients improved only upon elimination of multiple food items. Interestingly, clinical deterioration was noted in several patients following dietary changes. The overall clinical significance of this study remains low given the small number of patients included and, furthermore, the high degree of individual variation to dietary response leads to difficulties in data interpretation. This does, however, imply that dietary effects on disease activity are heterogeneous and likely patient-specific.
Mediterranean diet
Mediterranean diets which are high in olive oil, fish and fresh fruit and vegetables have been proposed as protective against inflammatory arthritis development due to the abundance of long chain n-3 polyunsaturated fatty acids, n-9 monounsaturated oleic acid, eicosatrienoic acid and anti-oxidants which mediate antiinflammatory responses [39,40].
Skoldstam et al. carried out a three month randomised clinical trial comparing disease activity in 51 patients with stable RA who were equally allocated to either a Mediterranean diet or control group [41]. Patients in the diet group demonstrated a median reduction in DAS-28. of 0.56 as well as patient-reported improvement in physical function and vitality; the control group demonstrated no significant changes [41].
A later prospective study reviewed the diets of 208 patients with RA and calculated Mediterranean diet scores in comparison to healthy controls [42]. Monounsaturated fatty acid (MUFA) intake, a key component of the Mediterranean diet, was found to be significantly lower in patients with RA and the consumed MUFA: saturated fatty acid ratio differed significantly within the patient group following sub-classification with disease activity scores; furthermore, disease activity (DAS28-ESR) significantly correlated with MUFA/SFA intake after age adjustment, suggesting that MUFA intake may be the key dietary component of the Mediterranean diet which suppresses disease activity in RA [42].
Lactovegetarian diet
Fasting has been documented to have beneficial effects on RA disease activity [39]. Fraser et al. demonstrated that fasting for 7-10 days with limited amounts of vitamin and mineral supplementation, carbohydrate and energy provided in the form of vegetable juice led to a decreased CD4+ lymphocyte activation and increased interleukin-4 production in peripheral blood of rheumatoid arthritis patients after acute starvation which suggests transient immunosuppression as significant decreases in swollen and tender joints, pain, ESR and CRP were noted [43]. A 2013 retrospective case control study by Su et al. [44] compared the DAS28, intake of medication, side effects and diet of a fasting cohort (n=39) and a non-fasting cohort (n=32) before and during Ramadhan. The mean DAS28 score was higher before Ramadhan in the fasting cohort however this was not significant although significant reduction in morning stiffness and function was found [44].
Skoldstam et al. assessed the clinical response to a 9-week lactovegetarian diet (vegetarian, inclusive of dairy but excluding eggs) in 16 patients with RA after a 7-10 day fast. A significant improvement in pain, stiffness, medication and clinical/biochemical findings was noted during fasting, before a rapid deterioration when commencing the lactovegetarian diet [45]. No differences were found in symptoms, drug consumption or clinical/biochemical variables between diet patients and controls.
A subsequent trial of 20 patients with RA on a 3-month vegan diet who were pre-fasted demonstrated subjective improvement, according to physician’s general assessment, in 55% of patients; 35% and 10% of patients reported either no change or deterioration in symptoms, respectively [46]. Statistically significant changes in ESR and CRP levels, however, were not noted between groups.
A 13-month prospective randomised placebocontrolled single-blind trial by Kjeldsen-Kragh et al. tested the effects of fasting on RA patients for 7-10 days before commencing an individually based gluten-free vegan diet for 3.5 months followed by an individual lactovegetarian diet for 9 months [47]. A significant improvement in number of tender/swollen joints, Ritchie’s articular index, pain score, duration of morning stiffness, grip strength and inflammatory markers was noted in the fasting and vegetarian diet groups compared to controls [47].
Beneficial effects in disease activity was also reported in a subsequent clinical trial in patients undergoing dietary elimination combined with a vegetarian diet; a statistically significant improvement according to the Paulus criteria (20% improvement in 4 of 6 measures: joint tenderness scores, joint swelling scores, physician and patient global assessments, Erythrocyte Sedimentation Rate (ESR) and morning stiffness) was noted in the diet group, which resulted in an exacerbation of symptoms in 59% following meat intake [47]. A follow up study 2 years later showed that the responders had continued with the diet and still had significantly reduced clinical disease variables and ESR levels [48].
Vegan diet
Veganism is a type of vegetarian diet that excludes meat, eggs, dairy products and all other animal-derived ingredients. A one-year randomised clinical trial assessed disease activity in 38 patients with RA on a vegan, gluten-free diet and 28 patients on a well-balanced non-vegan diet demonstrated a significant decrease in serum IgG antibodies to gliadin and beta-lactoglobulin in the vegan diet group; furthermore, according 40.9% of patients who completed the vegan diet for at least 9 months demonstrated an ACR 20 response compared with 4% in the non nonvegan group. after 12 months as the vegan diet responders exhibited significant improvement in all variables except for CRP [49]. These data suggest a vegan diet may result in a diminished immune response to food antigens, however due to strict dietary limitations a high dropout rate was noted, with only 58% of patients in the vegan diet group completing more than 9 months of the study [49].
Improvements in functional limitation, joint tenderness/swelling score, degree of pain and morning stiffness severity following a 4-week low-fat vegan diet in 24 patients with RA was also demonstrated in a later study; morning stiffness duration, however, did not differ between diet and control groups [50].
Gluten-free diet
A gluten-free diet has been proposed to have ameliorative effects on inflammatory arthritis, though studies reviewing the incidence of anti-gliadin antibodies in such patients have demonstrated variable results [51-53]. have focused on spondyloarthropathies given the data showing an increased incidence of anti-gliadin antibodies in both patients with AS and PsA. Limited objective data is available to suggest this diet is beneficial, however cases have been reported of PsA disease improvement following dietary gluten elimination [54] and links between psoriasis and coeliac disease have been proposed [55].
Low starch diet
Starch is one of the main dietary substrates required for colonic microbial growth, and therefore a low starch diet has been proposed to have ameliorative effects in AS by reducing the bulk of potentially pathogenic organisms such as klebiella in the faecal microflora [56].
A 9-month longitudinal study of 36 patients with active AS demonstrated an improvement in inflammatory markers, total IgA concentrations and a reduction in anti-inflammatory drug intake in the majority of patients following a decrease in dietary starch intake [56].
Discussion
Over recent decades there has been an increasing appreciation of the role that diet plays on the pathogenesis of IA.
Historical studies reviewing the impact of diet on inflammatory arthritis predominantly focus on RA, for example early prospective population based studies have suggested a high intake of omega-3 fatty acids, fruits and vegetables may be protective against disease development. Interestingly, some studies demonstrate this correlation is stronger with seropositive RA, suggesting the interaction between such dietary components and B cells may result in autoimmunity and antibody production [13-18].
Recent clinical trials suggest supplementation with vitamins A, C and E and curcumin use in RA, and omega-3 supplementation in RA and AS may improve overall disease activity; use of α-tocopherol, selenium or cat’s claw, however, may reduce pain scores but do not significantly affect overall disease activity [25-37].
More drastic dietary changes, however, demonstrate more impressive results; for example, a statistically significant improvement in disease activity scores was noted in patient’s adherent to a Mediterranean diet [41,42]. Trials reviewing the benefit of such dietary changes however show high drop-out rates, though individuals who notice a subjective improvement in disease activity are more likely to comply.
Conclusion and future work
Although a handful of small trials demonstrate a statistically significant impact of dietary change on IA disease activity, robust data to advocate dietary advice in a clinical setting remains limited; larger clinical trials are required, especially for ankylosing spondylitis and psoriatic arthritis, where data remains sparse. Studies including sub-analysis of patients with both early and established disease, individuals who are DMARD or biologic-naïve and those comparing seropositive with seronegative RA would be noteworthy to assess which patient groups would be most likely to benefit. Specific dietary components which demonstrate a significant impact on disease activity would warrant further research reviewing specific mechanisms of immune modulation, for example microbiome manipulation.
References
- Jethwa H, Abraham S. The evidence for microbiome manipulation in inflammatory arthritis. Rheumatology. 56(9), 1452-1460 (2017).
- Pianta A, Arvikar SL, Strle K et al. Two rheumatoid arthritis-specific autoantigens correlate microbial immunity with autoimmune responses in joints. J. Clin. Invest. 127(8), 2946-2956 (2017).
- Hammer RE, Maika SD, Richardson JA et al. Spontaneous inflammatory disease in transgenic rats expressing HLA-B27 and human β2m: An animal model of HLA-B27-associated human disorders. Cell. 63(5), 1099-1112 (1990).
- Ruutu M, Thomas G, Steck R et al. β‐glucan triggers spondylarthritis and Crohn's disease–like ileitis in SKG mice. Arthritis Rheum. 64(7), 2211-2222 (2012).
- Rashid T, Wilson C, Ebringer A. The Link between Ankylosing Spondylitis, Crohns Disease, Klebsiella, and Starch Consumption. Clin. Dev. Immunol. 2013, (2013).
- Eppinga H, R Konstantinov S, Peppelenbosch M et al. The Microbiome and Psoriatic Arthritis. Curr. Rheumatol. Rep. 16(3), 407 (2014).
- Panush RS, Stroud RM, Webster EM. Food-induced (allergic) arthritis: inflammatory arthritis exaccerbated by milk. Arthritis. Rheum. 29(2), 220-227 (1986).
- Hvatum M, Kanerud L, Hällgren R et al. The gut-joint axis: cross reactive food antibodies in rheumatoid arthritis. Gut. 55(9), 1240 (2006).
- Kim DH. Gut microbiota-mediated drug-antibiotic interactions. Drug. Metab. Dispos. 43(10), 158 -1589 (2015).
- Togrol RE, Nalbant S, Solmazgul E et al. The significance of coeliac disease antibodies in patients with ankylosing spondylitis: a case-controlled study. J. Int. Med. Res. 37(1), 220-226 (2009).
- Lindqvist U, Rudsander A, Bostrom A et al. IgA antibodies to gliadin and coeliac disease in psoriatic arthritis. Rheumatology. 41(1), 31-37 (2002).
- Kia KF, Nair RP, Ike RW et al. Prevalence of antigliadin antibodies in patients with psoriasis is not elevated compared with controls. Am. J. Clin. Dermatol. 8(5), 301-305 (2007).
- Abdel-Kader Martín N, H Cardiel M. Geographical factors in rheumatoid arthritis. Int. J. Clin. Rheumatol. 6(3), 259-261 (2011).
- Shapiro JA, Koepsell TD, Voigt LF et al. Diet and Rheumatoid Arthritis in Women: A Possible Protective Effect of Fish Consumption. Epidemiology. 7(3), 256-263 (1996).
- Cerhan JR, Saag KG, Merlino LA et al. Antioxidant micronutrients and risk of rheumatoid arthritis in a cohort of older women. Am. J. Epidemiol. 157(4), 345-354 (2003).
- Di Giuseppe D, Johansson K, Askling J et al. Mediterranean diet is inversely associated with risk of Rheumatoid Arthritis: A Swedish population based case control study. Arthritis. Rheumatol. (2017).
- Pattison D, Silman A, Goodson N et al. Vitamin C and the risk of developing inflammatory polyarthritis: prospective nested case-control study. Ann. Rheum. Dis. 63(7), 843-847 (2004).
- Lu B Hu Y, Sparks JA, Costenbader KH et al. Prospective Study of Dietary Patterns and Risk of Rheumatoid Arthritis in Women. Arthritis. Rheumatol. 67(10) (2015).
- Rashid T, Wilson C, Ebringer A. Raised incidence of ankylosing spondylitis among Inuit populations could be due to high HLA-B27 association and starch consumption. Rheumatol. Int. 35(6), 945-951 (2015).
- Park MK, Li W, Paek SY et al. Consumption of polyunsaturated fatty acids and risk of incident psoriasis and psoriatic arthritis from the Nurses’ Health Study II. Br. J. Dermatol. 177(1), 302-306 (2017).
- https://www.mayoclinic.org/nutrition-and-pain/art-20208638
- https://www.arthritisresearchuk.org/arthritis-information/conditions/rheumatoid-arthritis/self-help/diet.aspx
- http://www.papaa.org/further-information/you-and-your-diet.
- https://nass.co.uk/about-as/living-well-with-as/diet-and-as/
- Edmonds S, Winyard P, Guo R et al. Putative analgesic activity of repeated oral doses of vitamin E in the treatment of rheumatoid arthritis. Results of a prospective placebo controlled double blind trial. Ann. Rheum. Dis. 56(11), 649-655 (1997).
- Jaswal S, Mehta HC, Sood AK et al. Antioxidant status in rheumatoid arthritis and role of antioxidant therapy. Clin. Chim. Acta. 338(1-2), 123-129 (2003).
- Canter PH, Wider B, Ernst E. The antioxidant vitamins A, C, E and selenium in the treatment of arthritis: a systematic review of randomized clinical trials. Rheumatology. 46(8), 1223-1233 (2007).
- Volker D, Fitzgerald P, Major G et al. Efficacy of fish oil concentrate in the treatment of rheumatoid arthritis. J. Rheumatol. 27(10), 2343-2346 (2000).
- Sundrarjun T, Komindr S, Archararit N et al. Effects of n-3 fatty acids on serum interleukin-6, tumour necrosis factor-alpha and soluble tumour necrosis factor receptor p55 in active rheumatoid arthritis. J. Int. Med. Res. 32(5), 443-454 (2004).
- Fortin PR, Lew RA, Liang MH et al. Validation of a meta-analysis: The effects of fish oil in rheumatoid arthritis. J. Clin. Epidemiol. 48(11), 1379-1390 (1995).
- Goldberg RJ, Katz J. A meta-analysis of the analgesic effects of omega-3 polyunsaturated fatty acid supplementation for inflammatory joint pain. Pain. 129(1-2), 210-223 (2007).
- Sundström B, Stålnacke K, Hagfors L et al. Supplementation of omega-3 fatty acids in patients with ankylosing spondylitis. Scand. J. Rheumatol. 35(5), 359-362 (2006).
- Mur E, Hartig F, Eibl G et al. Randomized double blind trial of an extract from the pentacyclic alkaloid-chemotype of uncaria tomentosa for the treatment of rheumatoid arthritis. J. Rheumatol. 29(4), 678-681 (2002).
- Gupta SC, Patchva S, Aggarwal BB. Therapeutic Roles of Curcumin: Lessons Learned from Clinical Trials. AAPS. J. 15(1), 195-218 (2013).
- Chandran B, Goel A. A Randomized Pilot Study to Assess the Efficacy and Safety of Curcumin in Patients with Active Rheumatoid Arthritis. Phytother. Res. 2012; 1719-1725.
- Heng MC, Song MK, Harker J et al. Drug‐induced suppression of phosphorylase kinase activity correlates with resolution of psoriasis as assessed by clinical, histological and immunohistochemical parameters. Br. J. Dermatol. 143(5), 937-949 (2000).
- Kurd SK, Smith N, VanVoorhees A et al. Oral curcumin in the treatment of moderate to severe psoriasis vulgaris: A prospective clinical trial. J. Am. Acad. Dermatol. 58(4), 625-631 (2008).
- Beri D, Malaviya AN, Shandilya R et al. Effects of dietary restrictions on disease actvitiy in rheumatoid arthritis. Ann. Rheum. Dis. 47(1), 69-72 (1988).
- Haugen M, Fraser D, Forre O. Diet therapy for the patient with rheumatoid arthritis? Rheumatology. 38(11), 1039 (1999).
- Rayman MP, Pattison DJ. Dietary manipulation in musculoskeletal conditions. Best. Pract. Res. Clin. Rheumatol. 22(3), 535-561 (2008).
- Skoldstam L, Hagfors L, Johansson G. An experimental study of a Mediterranean diet intervention for patients with rheumatoid arthritis. Ann. Rheum. Dis. 62(3), 208-214 (2003).
- Matsumoto Y, Sugioka Y, Tada M et al. Monounsaturated fatty acids might be key factors in the Mediterranean diet that suppress rheumatoid arthritis disease activity: The TOMORROW study. Clin. Nutr. 37(2), 675-680 (2018).
- Fraser DA, Thoen J, Reseland JE et al. Decreased CD4+ lymphocyte activation and increased interleukin-4 production in peripheral blood of rheumatoid arthritis patients after acute starvation. Clin. Rheumatol. 18(5), 394-401 (1999).
- Su VX, Azahar NA, Jeans Y et al. AB0244 Retrospective study on effects of ramadhan month fasting on rheumatoid arthritis patients. Ann. Rheum. Dis. 72(Suppl 3), A861 (2013).
- Sköldstam L, Larsson L, Lindström FD. Effects Of Fasting and Lactovegetarian Diet on Rheumatoid Arthritis. Scand. J. Rheumatol. 8(4), 249-55 (1979).
- Skoldstam L. Fasting and vegan diet in rheumatoid arthritis. Scand. J. Rheumatol. 15(2), 219-212 (1986).
- Kjeldsen-Kragh J, Haugen M, Borchgrevink CF et al. Controlled trial of fasting and one-year vegetarian diet in rheumatoid arthritis. Lancet. 338(8772), 899-902 (1991).
- Kjeldsen-Kragh J, Haugen M, Borchgrevink CF et al. Vegetarian diet for patients with rheumatoid arthritis: status two years after introduction of diet. Clin. Rheumatol. 13(3), 475-482 (1994).
- Hafström I, Ringertz B, Spångberg A et al. A vegan diet free of gluten improves the signs and symptoms of rheumatoid arthritis: the effects on arthritis correlate with a reduction in antibodies to food antigens. Rheumatology. 40(10), 1175-1179 (2001).
- McDougall J, Bruce B, Spiller G et al. Effects of a very low-fat, vegan diet in subjects with rheumatoid arthritis. J. Altern. Complement. Med. 8(1), 71-75 (2002).
- Coaccioli S, Landucci P, Fatati G et al. Prevalence of coeliac disease in rheumatoid and psoriatic arthritis and in psoriasis. Med. J. Nutrition. Metab. 3(1), 61-64 (2010).
- Roth EB, Stenberg P, Book C et al. Antibodies against transglutaminases, peptidylarginine deiminase and ciltrulline in rheumatoid arthritis: New pathways to epitope spreading. Clin. Exp. Rheumatol. 24, 2–8 (2006).
- Riente L, Chimenti D, Pratesi F et al. Antibodies to tissue transglutaminase and Saccharomyces cerevisiae in ankylosing spondylitis and psoriatic arthritis. J. Rheumatol. 31, 920–924 (2004).
- Fitzgerald K, Hyman M, Swift K. Psoriatic arthritis. Glob. Adv. Health. Med. 1(4), 54-61 (2012).
- Ludvigsson JF, Lindelof B, Zingone F et al. Psoriasis in a nationwide cohort study of patients with celiac disease. J. Investig. Dermatol. 131(1), 2010-2016 (2011).
- Ebringer A, Wilson C. The Use of a Low Starch Diet in the Treatment of Patients Suffering from Ankylosing Spondylitis. Clin. Rheumatol. 15(S1), 62-66 (1996).