Review Article - Imaging in Medicine (2012) Volume 4, Issue 2
PET/CT findings in gastric cancer: potential advantages and current limitations
Saadet Atay-Rosenthal*, Richard L Wahl & Elliot K FishmanThe Russell H. Morgan Department of Radiology & Radiological Sciences, Johns Hopkins Hospital, 601 N. Caroline Street, Johns Hopkins Outpatient Center, Office 3171D, Baltimore, MD 21287, USA
- Corresponding Author:
- Saadet Atay-Rosenthal
The Russell H. Morgan Department of Radiology & Radiological Sciences
Johns Hopkins Hospital, 601 N. Caroline Street
Johns Hopkins Outpatient Center, Office 3171D, Baltimore, MD 21287, USA
Tel: +1 410 502 8052
Fax: +1 443 287 6422
E-mail: satayro1@jhmi.edu
Abstract
18F-fluorodeoxyglucose (18F-FDG) as a substrate for tumors with high glucose metabolism has been the most commonly used 18F-FDG-PET radiopharmaceutical in oncologic imaging. However, the usefulness of 18F-FDG-PET in gastric cancer has not yet been fully established. The intent of this review is to help to better understand the impact of 18F‑FDG-PET on the imaging of gastric cancer, to offer a greater understanding of how gastric malignancies may present and be recognized on 18F‑FDG‑PET and to assess the complementary value of PET/CT in gastric cancer staging.
Keywords
18F-FDG-PET ▪ 18F-fluorodeoxyglucose ▪ gastric cancer staging ▪ gastric carcinoma ▪ PET/CT ▪ TNM staging
Successful management of gastric cancer depends on early detection and accurate staging of disease as surgery is the only curative treatment method for localized disease. Endoscopic ultrasound has been the most reliable nonsurgical method in evaluation of the primary tumor, however, recent studies comparing the preoperative staging of gastric cancer by endoscopic ultrasound with multidetector CT show that the two modalities demonstrate very close accuracy in determining the individual T and N stage [1,2]. Multidetector CT is currently the staging modality of choice in identifying the primary tumor, assessing the local spread of the tumor, detecting local and distant nodal disease and metastasis [3–5]. Considering that on average 25% of patients with newly diagnosed gastric carcinoma have undetectable intra-abdominal M1 disease (metastasis to peritoneum, liver or nonregional lymph nodes) by current imaging modalities and yet detected at surgical staging [6], there is a need for better imaging to evaluate the extent of disease and avoid unnecessary surgery.
The role of 18F-fluorodeoxyglucose (18F-FDG)- PET in detection of primary gastric cancer and lymph node metastasis has been controversial. There is limited data on use of PET/CT for preoperative staging of gastric cancer, although there is growing interest in this topic [7]. 18F-FDGPET can be useful in postoperative follow-up of gastric cancer patients with suspected recurrent gastric cancer, especially when the recurrence is suspected clinically because of its high positive predictive value [8,9]. Treatment decisions were changed in 30.4% patients when PET/CT was introduced to conventional follow-up [10]. Significant correlation was found between 18F-FDG-PET uptake and patient survival [11].
In this article we review some of the gastric 18F-FDG uptake patterns and their importance in gastric cancer detection, and describe the additional value of PET/CT on gastric cancer staging illustrated with case examples. The studies were performed in routine setting with dedicated PET/CT system with no special preparation in terms of gastric distention other than oral contrast or water on the table before imaging.
Gastric 18F-FDG-uptake pattern & significance
Variable physiologic 18F-FDG uptake simply caused by visceral thickening, physiological emptying, or inflammatory disease in the alimentary tract often makes it difficult to differentiate normal uptake from pathology. Highly variable normal gastric 18F-FDG uptake limits the PET detection rate for early gastric cancer. Only 60% of locally advanced gastric carcinoma was detected by 18F-FDG-PET in a report by Stahl et al. [12].
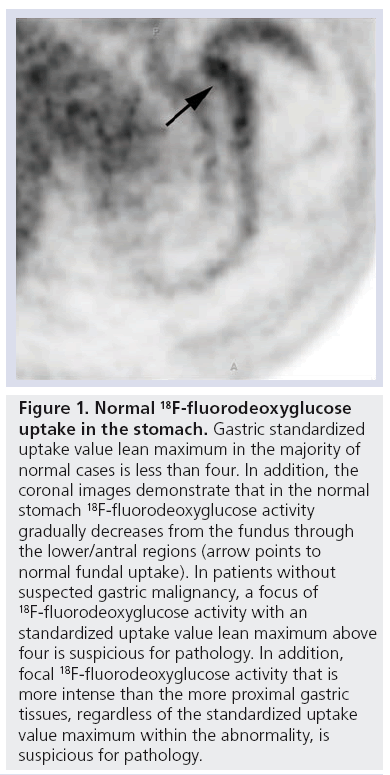
There is high patient-to-patient variation in the concentration of tracer at the gastroesophageal junction (GEJ) and gastric antrum [13]. However, a specific pattern of gastric 18F-FDG uptake was demonstrated by Koga et al. in a review of 22 patients without any gastric lesions. The gastric regions were classified into U (upper)- area, M (middle)-area and L (lower)-area. In all cases, the 18F-FDG uptake in the U-area was equal to or higher than in the M- and L-areas (U > M > L) [14]. Thus, higher focal uptake in the M- or L-area compared with the upper area may be suggestive of pathologic uptake (Figures 1–3).
Figure 1: Normal 18F-fluorodeoxyglucose uptake in the stomach. Gastric standardized uptake value lean maximum in the majority of normal cases is less than four. In addition, the coronal images demonstrate that in the normal stomach 18F-fluorodeoxyglucose activity gradually decreases from the fundus through the lower/antral regions (arrow points to normal fundal uptake). In patients without suspected gastric malignancy, a focus of 18F-fluorodeoxyglucose activity with an standardized uptake value lean maximum above four is suspicious for pathology. In addition, focal 18F-fluorodeoxyglucose activity that is more intense than the more proximal gastric tissues, regardless of the standardized uptake value maximum within the abnormality, is suspicious for pathology.
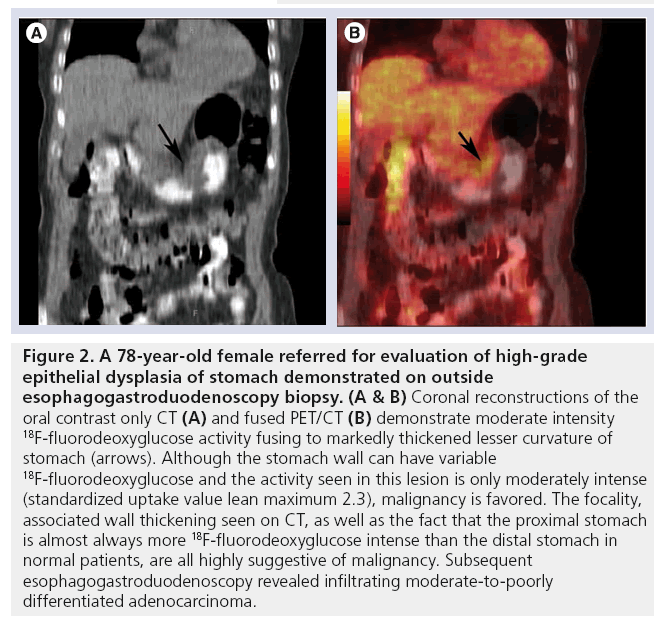
Figure 2: A 78‑year-old female referred for evaluation of high-grade epithelial dysplasia of stomach demonstrated on outside esophagogastroduodenoscopy biopsy. (A & B) Coronal reconstructions of the oral contrast only CT (A) and fused PET/CT (B) demonstrate moderate intensity 18F-fluorodeoxyglucose activity fusing to markedly thickened lesser curvature of stomach (arrows). Although the stomach wall can have variable 18F-fluorodeoxyglucose and the activity seen in this lesion is only moderately intense (standardized uptake value lean maximum 2.3), malignancy is favored. The focality, associated wall thickening seen on CT, as well as the fact that the proximal stomach is almost always more 18F-fluorodeoxyglucose intense than the distal stomach in normal patients, are all highly suggestive of malignancy. Subsequent esophagogastroduodenoscopy revealed infiltrating moderate-to-poorly differentiated adenocarcinoma.
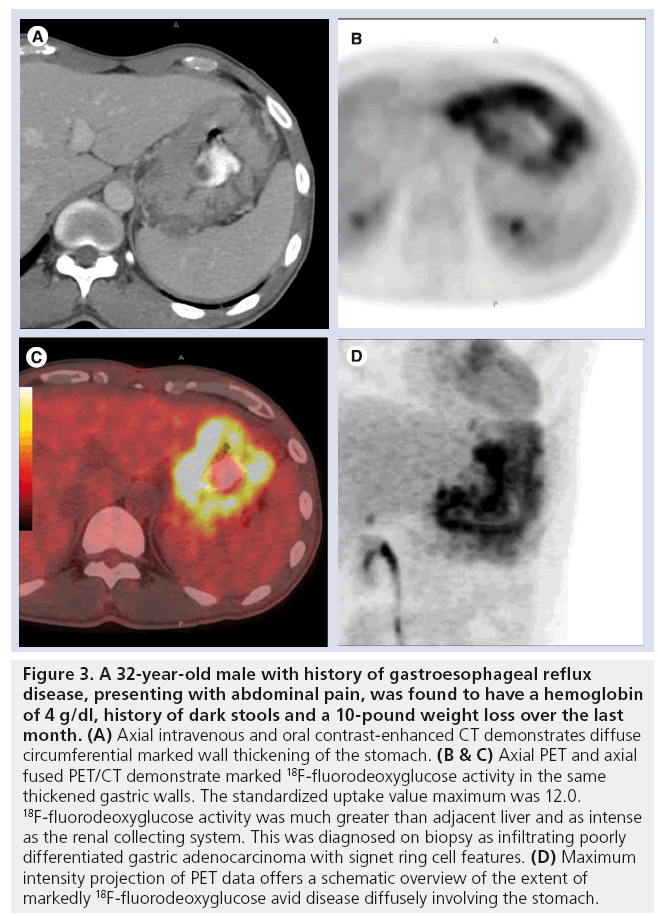
Figure 3: A 32-year-old male with history of gastroesophageal reflux disease, presenting with abdominal pain, was found to have a hemoglobin of 4 g/dl, history of dark stools and a 10-pound weight loss over the last month. (A) Axial intravenous and oral contrast-enhanced CT demonstrates diffuse circumferential marked wall thickening of the stomach. (B & C) Axial PET and axial fused PET/CT demonstrate marked 18F-fluorodeoxyglucose activity in the same thickened gastric walls. The standardized uptake value maximum was 12.0. 18F-fluorodeoxyglucose activity was much greater than adjacent liver and as intense as the renal collecting system. This was diagnosed on biopsy as infiltrating poorly differentiated gastric adenocarcinoma with signet ring cell features. (D) Maximum intensity projection of PET data offers a schematic overview of the extent of markedly 18F-fluorodeoxyglucose avid disease diffusely involving the stomach.
Kamimura et al. in a prospective evaluation of 60 patients, however, did not find a significant difference in each gastric area when an additional 400 ml of water was given prior to imaging. The standardized uptake value (SUV) remains high, however, in the GEJ than in the U-area [15].
The physiological gastric uptake can be somewhat reduced in some instances using gastric distention. The potential importance of additional water intake or vesicant just before the study to differentiate physiological from pathological uptake in primary and recurrent tumors has been demonstrated in several articles [15–19]. Gastric distention enabled better delineation of the lesions and reduction in nonspecific uptake. Kamimura et al. reported increased specificity from 50 to 100% and negative predictive value from 72 to 94% in patients with locally advanced gastric carcinomas after ingestion of 400 ml of water and additional spot imaging of the stomach. The average SUV of the stomach declined from 2.6 to 1.65 following gastric distention (SUV units are g/ml, however in the following discussion we adopt conventional practice and present SUV in dimensionless units) [18].
Histological types of gastric cancer can cause significant variation in 18F-FDG uptake. Moderately differentiated type tubular adenocarcinoma and intestinal type of advanced gastric cancer reveal significantly higher uptake (SUV: 7.7–13.2). Nonintestinal diffuse type, mucinous adenocarcinoma and carcinomas containing signet ring cells display low detectability on PET. This is, in part, due to high content of metabolically quite inert mucus and low tumor cell density. One other reason may be lack of expression of the glucose transporter Glut-1 on the cell membrane of most signet ring cells and mucinous adenocarcinoma [20–23]. Both the depth of tumor invasion and histological subtypes were found to be independent factors influencing the 18F-FDG avidity. Glut-1 expression was the most influential factor for the degree of 18F-FDG uptake in gastric cancer in multiple regression analysis (p < 0.01) [24]. In nonintestinal type gastric cancer with a high number of signet ring cells, thymidine analog 3´-deoxy-3´-18F-fluorothymidine (FLT) PET may be used for imaging, which has shown 100% sensitivity compared with 69% with 18F-FDG-PET in locally advanced gastric cancer in a pilot study of 45 patients [25]. In 21 patients with newly diagnosed advanced gastric cancer the sensitivities of FLT-PET and 18F-FDG-PET were 95.2 and 95%, respectively. The FLT-PET signal is often quite low, however [26].
We demonstrate a case of signet ring carcinoma with moderately positive 18F-FDG uptake (Figures 4 & 5).
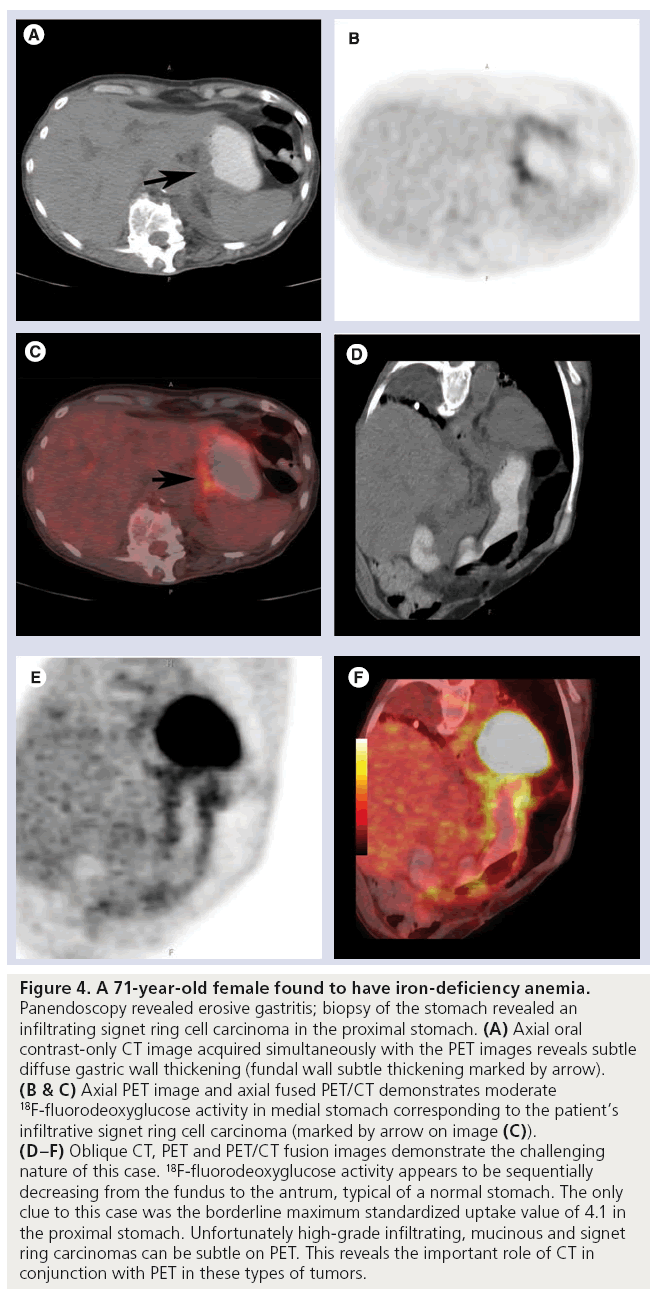
Figure 4: A 71-year-old female found to have iron-deficiency anemia. Panendoscopy revealed erosive gastritis; biopsy of the stomach revealed an infiltrating signet ring cell carcinoma in the proximal stomach. (A) Axial oral contrast-only CT image acquired simultaneously with the PET images reveals subtle diffuse gastric wall thickening (fundal wall subtle thickening marked by arrow). (B & C) Axial PET image and axial fused PET/CT demonstrates moderate 18F-fluorodeoxyglucose activity in medial stomach corresponding to the patient’s infiltrative signet ring cell carcinoma (marked by arrow on image (C)). (D–F) Oblique CT, PET and PET/CT fusion images demonstrate the challenging nature of this case. 18F-fluorodeoxyglucose activity appears to be sequentially decreasing from the fundus to the antrum, typical of a normal stomach. The only clue to this case was the borderline maximum standardized uptake value of 4.1 in the proximal stomach. Unfortunately high-grade infiltrating, mucinous and signet ring carcinomas can be subtle on PET. This reveals the important role of CT in conjunction with PET in these types of tumors.
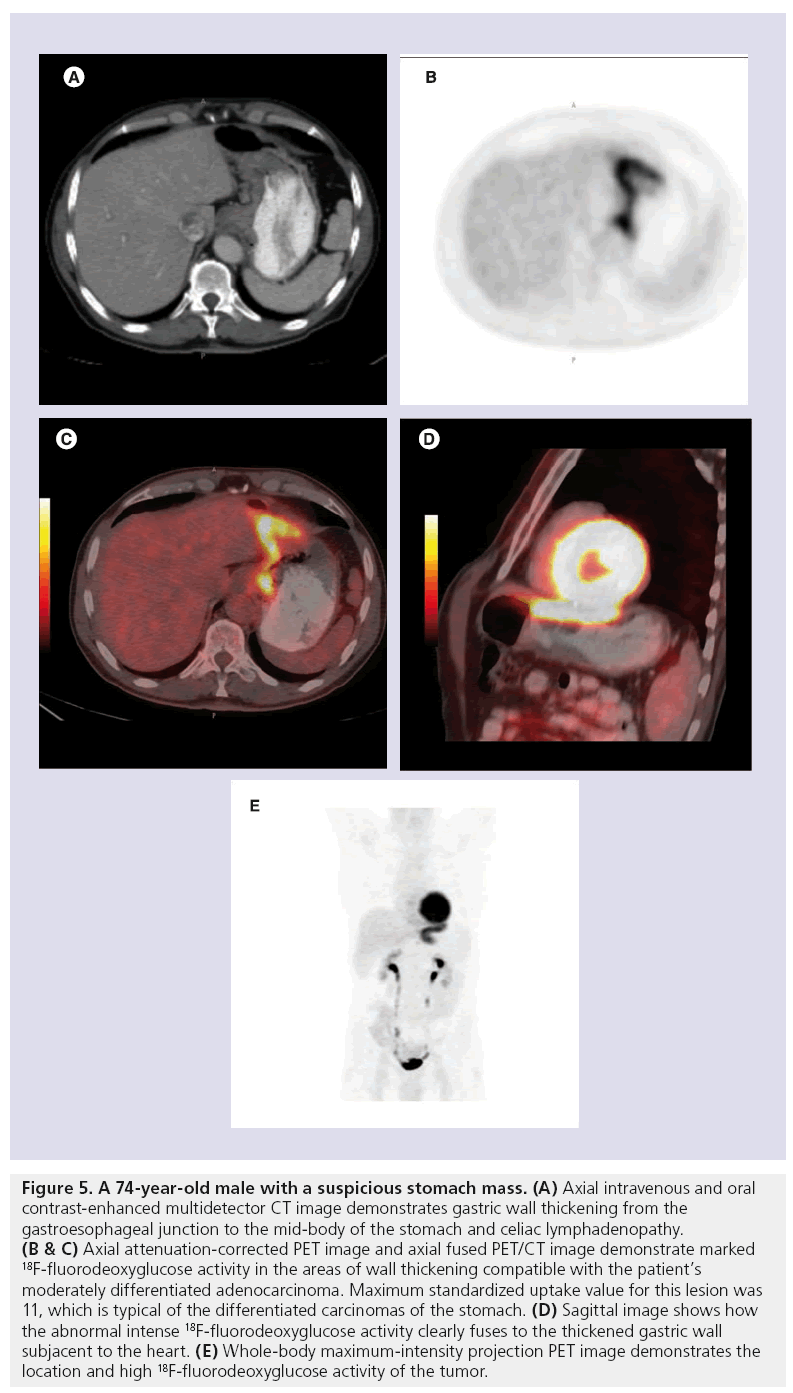
Figure 5: A 74-year-old male with a suspicious stomach mass. (A) Axial intravenous and oral contrast-enhanced multidetector CT image demonstrates gastric wall thickening from the gastroesophageal junction to the mid-body of the stomach and celiac lymphadenopathy. (B & C) Axial attenuation-corrected PET image and axial fused PET/CT image demonstrate marked 18F-fluorodeoxyglucose activity in the areas of wall thickening compatible with the patient’s moderately differentiated adenocarcinoma. Maximum standardized uptake value for this lesion was 11, which is typical of the differentiated carcinomas of the stomach. (D) Sagittal image shows how the abnormal intense 18F-fluorodeoxyglucose activity clearly fuses to the thickened gastric wall subjacent to the heart. (E) Whole-body maximum-intensity projection PET image demonstrates the location and high 18F-fluorodeoxyglucose activity of the tumor.
The site of primary tumor may also influence the detection rate by 18F-FDG-PET such that proximal lesions are detected at a higher rate; however, the results are controversial [22,23].
One of the larger series is by Takahashi et al. studying the significance of gastric 18F-FDG accumulation in a retrospective analysis of 599 patients. The main cause for nonspecific accumulation was inf lammatory mucosal changes forming a background for the development of cancer or malignant lymphoma. In this study although the 18F-FDG uptake was thought to be nonspecific in four out of 91 positive cases, endoscopy revealed three cases of early gastric cancer and one case of mucosa-associated lymphoid tissue lymphoma. Two of the early gastric cancers were signet ring type with the lesion sizes of 5 and 14 mm. Incidental lesions also included benign disorders, such as gastric and duodenal ulcer and gastric submucosal tumor [27].
SUV & its implications
18F-FDG uptake in the tumor is semiquantitatively assessed using SUV. SUVs are dependent on many parameters including, but not limited to, time after 18F-FDG injection, tumor size and blood glucose level and are also affected by the methods of both image reconstruction and attenuation correction. Although there is no consensus on the cutoff level, 95% of patients with disease-free stomach demonstrate a peak SUV of less than four in the GEJ and gastric antrum [13].
SUV values are important in estimating prognosis and follow-up of patients. Significant correlation is observed between SUV and the primary size of tumors. The gastric cancers with high 18F-FDG uptake tend to have higher malignant aggressiveness as there is significant correlation between the primary tumor SUV and lymph node metastases [20]. High 18F-FDG uptake in primary tumors increases the accuracy of FDG-PET assessment of lymph node stage [28]. Patients with high tumor SUV had poorer prognosis with lower survival rates than those with low SUVs [23].
Serial 18F-FDG-PET imaging is used to monitor cytotoxic therapy and for early prediction of histopathological response to chemotherapy. In a study by Stahl et al., relative tumor SUV changes were not influenced by any of the methodological variations, such as time delay, after 18F-FDG injection, acquisition protocol, reconstruction algorithm or normalization of SUV, as long as they were the same method for pre- and post-treatment imaging, in a retrospective evaluation of 43 patients with serial imaging. When a decline in tumor SUV of approximately 40% was used as a cutoff value between responders and nonresponders, the accuracy for prediction of response was high at 80% [29]. Similar results were obtained in other studies assessing the relative changes in tumor 18F-FDG uptake for prediction of outcome as early as 2 weeks after initiation of therapy [30,31].
The therapy induced reduction of 18F-FDG-PET predicted early metabolic response according to RECIST, in patients with advanced gastric adenocarcinoma treated with chemotherapy plus cetuximab at 6 weeks with
Patients with a significant histopathological response to preoperative therapy also have better prognosis and higher survival rates. The 2-year survival was 87% compared with 44% in nonresponders (p = 0.02) in a study by Wieder et al. in patients with locally advanced adenocarcinoma of the esophagogastric junction [30].
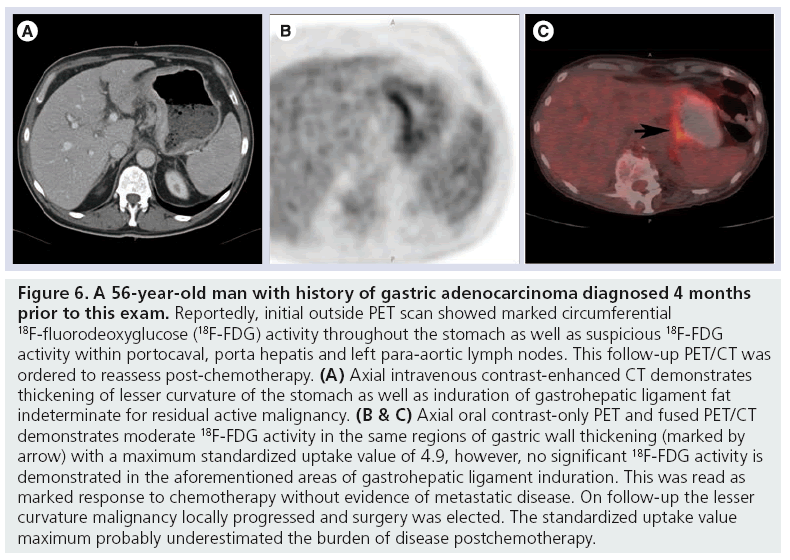
When a tumor is partially treated, whether by chemotherapy or radiation, its maximum SUV can frequently be below four yet harbor significant residual disease (Figure 6).
Figure 6: A 56-year-old man with history of gastric adenocarcinoma diagnosed 4 months prior to this exam. Reportedly, initial outside PET scan showed marked circumferential 18F-fluorodeoxyglucose (18F-FDG) activity throughout the stomach as well as suspicious 18F-FDG activity within portocaval, porta hepatis and left para-aortic lymph nodes. This follow-up PET/CT was ordered to reassess post-chemotherapy. (A) Axial intravenous contrast-enhanced CT demonstrates thickening of lesser curvature of the stomach as well as induration of gastrohepatic ligament fat indeterminate for residual active malignancy. (B & C) Axial oral contrast-only PET and fused PET/CT demonstrates moderate 18F-FDG activity in the same regions of gastric wall thickening (marked by arrow) with a maximum standardized uptake value of 4.9, however, no significant 18F-FDG activity is demonstrated in the aforementioned areas of gastrohepatic ligament induration. This was read as marked response to chemotherapy without evidence of metastatic disease. On follow-up the lesser curvature malignancy locally progressed and surgery was elected. The standardized uptake value maximum probably underestimated the burden of disease postchemotherapy.
Staging
■ Detection of primary tumors: T-staging
The sensitivity rate for detecting primary gastric tumors by 18F-FDG-PET alone ranges between 58 and 94% among studies (median 81.5%); the specificity, however, ranges from 78 to 100% (median 100%) [21]. The detection rate is even lower for early stage gastric cancers. In early gastric cancer (EGC), only the intestinal type was detectable with 18F-FDG-PET [33]. Advancedstage gastric cancer is detected by PET imaging in 90% of patients as opposed to 40% in early stage gastric cancers [23].
The screening sensitivity of 18F-FDG with PET-only imaging was found to be even lower (12.5%) in an asymptomatic patient group of 2861 subjects when endoscopy (chromoendoscopy with enhanced ability to detect small lesions) findings were used as a gold standard. The study, however, reveals high specificity and negative predictive values of 99.2 and 99.4%, respectively [34].
18F-FDG-PET improves TNM staging. In a prospective study by Chen et al. a high sensitivity of 94% was found when 68 patients with biopsy proven early and advanced gastric cancer were enrolled for preoperative staging [20].
Yun et al. reported that 18F-FDG-PET is as accurate as CT for detecting primary tumors of the stomach in either EGC or advanced gastric cancer [35].
Limited data are available on combined PET/CT that would further improve the diagnostic performance. In a recent article, Kim et al. showed high sensitivity of PET/CT for the detection of primary tumors (n = 66 out of 71; 93%), similar to the detection rate for contrast-enhanced CT (n = 64 out of 71, 90%; p = 0.55) [7].
■ Detection of regional lymph node metastases: N-staging
Lymph node status is an important prognostic factor regarding long-term survival [36]. The extent of lymph node metastasis at primary diagnosis is the most important independent factor associated with the survival time and determining the timing of tumor recurrence [21,37].
CT has major limitations detecting cancerous involvement of normal-sized lymph nodes and cannot differentiate between reactive hyperplasia and metastatic enlargement. Regional lymph nodes are considered to represent local metastasis if they are solitary or separate nodes of 8 mm or greater in the long axis diameter, round shape, or exhibiting central necrosis with marked or heterogeneous enhancement [36,38]. However, in the literature 55% of the metastatic lymph nodes are found to be 5 mm or less in diameter. When 348 lymph nodes less than 3 mm were analyzed prospectively in 31 gastrectomy specimens, 14.5% were found to have metastasis in histological evaluation [36].
18F-FDG-PET has limited detection of locoregional lymph node metastases from gastric cancer [39]. Perigastric lymph nodes may not be distinguished from the primary tumor or the normal stomach wall. The sensitivity of CT for N1 disease was found to be significantly higher than that of PET. However, the sensitivity, specificity and accuracy of 18F-FDG-PET were not significantly different from CT for primary tumors or for N2 and N3 metastases in EGC and advanced gastric cancer. In clinical practice, detection of N2 or N3 disease status may change the extent of lymphadenectomy. In advanced gastric cancer, all patients undergo D1 dissection anyway so detection of N1 status may not change the treatment [35].
During the evaluation of lymph node staging 18F-FDG-PET shows 23.3% sensitivity and 100% specificity whereas CT shows 65% sensitivity and 77% specificity in a study of 85 patients when the lymph node was considered positive if the long axis diameter was more than 1 cm [23]. Kim et al. reported that according to multiple logistic regression analysis the SUV of the primary tumors was the only independent variable to be significantly related to sensitivity for lymph node metastases [28].
Current 18F-FDG-PET scanners have 4–10 mm spatial resolution, which may also explain the low sensitivity especially in detection of perigastric N1 lymph nodes. PET/CT fusion provides both anatomic and functional information and improves localization of foci with increased uptake than standalone PET (Figures 7 & 8). The tumor detection task is complicated by small size, low uptake in tumors and high normal 18F-FDG background uptake.
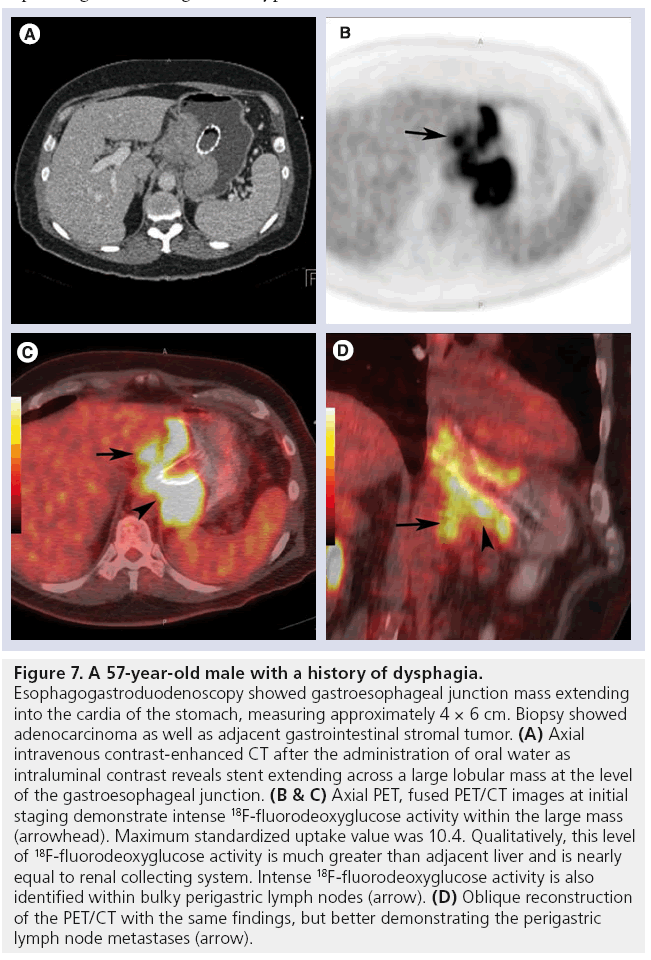
Figure 7: A 57-year-old male with a history of dysphagia. Esophagogastroduodenoscopy showed gastroesophageal junction mass extending into the cardia of the stomach, measuring approximately 4 × 6 cm. Biopsy showed adenocarcinoma as well as adjacent gastrointestinal stromal tumor. (A) Axial intravenous contrast-enhanced CT after the administration of oral water as intraluminal contrast reveals stent extending across a large lobular mass at the level of the gastroesophageal junction. (B & C) Axial PET, fused PET/CT images at initial staging demonstrate intense 18F-fluorodeoxyglucose activity within the large mass (arrowhead). Maximum standardized uptake value was 10.4. Qualitatively, this level of 18F-fluorodeoxyglucose activity is much greater than adjacent liver and is nearly equal to renal collecting system. Intense 18F-fluorodeoxyglucose activity is also identified within bulky perigastric lymph nodes (arrow). (D) Oblique reconstruction of the PET/CT with the same findings, but better demonstrating the perigastric lymph node metastases (arrow).
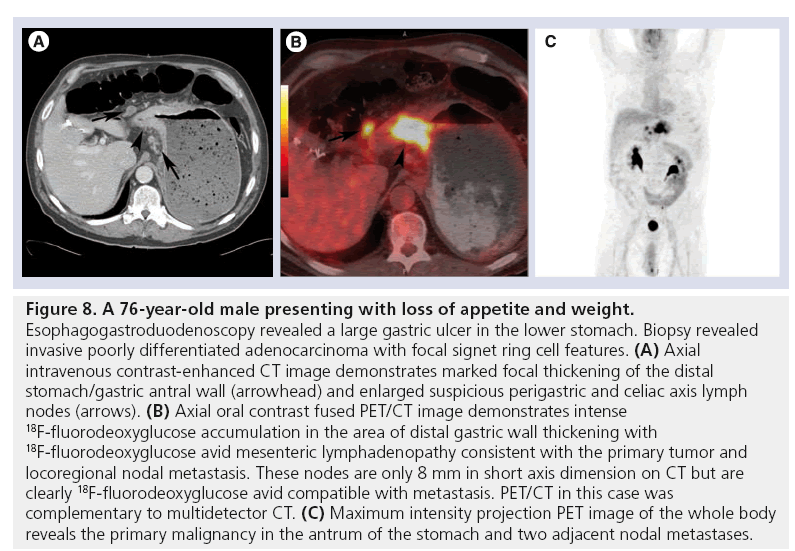
Figure 8: A 76-year-old male presenting with loss of appetite and weight. Esophagogastroduodenoscopy revealed a large gastric ulcer in the lower stomach. Biopsy revealed invasive poorly differentiated adenocarcinoma with focal signet ring cell features. (A) Axial intravenous contrast-enhanced CT image demonstrates marked focal thickening of the distal stomach/gastric antral wall (arrowhead) and enlarged suspicious perigastric and celiac axis lymph nodes (arrows). (B) Axial oral contrast fused PET/CT image demonstrates intense 18F-fluorodeoxyglucose accumulation in the area of distal gastric wall thickening with 18F-fluorodeoxyglucose avid mesenteric lymphadenopathy consistent with the primary tumor and locoregional nodal metastasis. These nodes are only 8 mm in short axis dimension on CT but are clearly 18F-fluorodeoxyglucose avid compatible with metastasis. PET/CT in this case was complementary to multidetector CT. (C) Maximum intensity projection PET image of the whole body reveals the primary malignancy in the antrum of the stomach and two adjacent nodal metastases.
The sensitivity and accuracy of PET/CT was found to be inferior to contrast-enhanced CT, however, specificity and positive predictive value for PET/CT was significantly higher than for CT in a series of 59 patients with confirmed regional lymph node metastases [7].
■ Detection of distant metastases: M-staging
Detection of metastatic lymph nodes is found to be particularly poor with PET with a sensitivity of only 20.6% [23]. Both 18F-FDG-PET and CT have low sensitivity in detecting N2 and N3 lymph node stations between 33–46.2 and 44–63.1%, respectively. The 18F-FDG-PET specificity on the other hand ranges between 91 and 100% (median: 96%) for N1 and N2 stations.
The sensitivity of 18F-FDG-PET with regard to lymph node metastases increases to 65% with a 90% specificity in cases with high primary tumor SUV. Signet ring cell carcinoma reveals the lowest sensitivity (15%), whereas other cell types can be detected with moderate sensitivity (30–71%) [28].
18F-FDG-PET has a better positive predictive value for lymph node metastases compared with CT, which affects treatment strategy in considering palliative therapy. When there is involvement of N3 lymph nodes a palliative approach is considered rather than curative resection [21].
Kim et al. demonstrate that in a series of 11 patients with distant metastases all of the four distant lymph node metastases were detected by combined PET/CT and one was missed by CT alone owing to small size [7]. PET/CT would be particularly important in distant metastases detection.
PET has very limited value in detecting peritoneal carcinomatosis with a low sensitivity (range: 9–50%; median: 32.5%), but relatively higher specificity of (63–99%; median: 88.5%). Low sensitivity may be explained by associated fibrosis, small size of peritoneal lesions (<5 mm) and low number of tumor cells in ascites, pleural and bone metastases [21].
Limited sensitivity for detecting peritoneal carcinomatosis with 18F-FDG-PET or CT scanning alone (57 and 43%, respectively) has been reported. When the images were evaluated together the sensitivity increased to 78% with a high positive predictive value of 95% in a study by Turlakow et al. [40]. The sensitivity and specificity of PET/CT was found to be similar to contrast-enhanced CT in all sites of recurrence in a study by Sim et al. except peritoneal seeding in which contrast-enhanced CT was more sensitive [41].
Evaluation of distant metastasis by PET alone is limited with reported sensitivity and specificity of 85 and 74% in detection of liver metastasis; 67 and 88% for lung metastases; 24 and 76% for ascites; 4 and 100% for pleural carcinomatosis; and 30 and 82% for bone metastases, respectively [21]. The results are better with a dedicated PET/CT system with a per-lesionbased positive predictive value of up to 89% in detecting recurrence after surgical resection. A study by Park et al. demonstrated that among the 108 recurrence sites, lymph node and lung metastases showed a higher rate of 18F-FDG uptake while nearly half of bowel metastases showed negative uptake and all uptake in lymph nodes and bones were true positive [8].
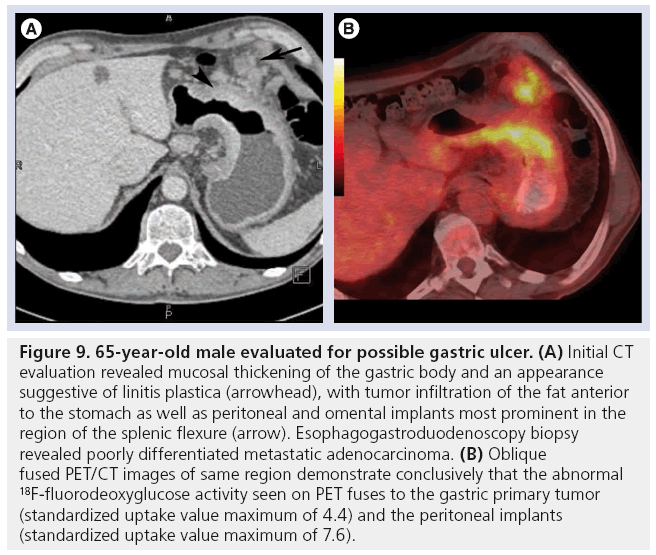
Combined imaging with the higher sensitivity of CT and higher specificity of PET will be more helpful in these cases than either imaging alone (Figure 9).
Figure 9: 65-year-old male evaluated for possible gastric ulcer. (A) Initial CT evaluation revealed mucosal thickening of the gastric body and an appearance suggestive of linitis plastica (arrowhead), with tumor infiltration of the fat anterior to the stomach as well as peritoneal and omental implants most prominent in the region of the splenic flexure (arrow). Esophagogastroduodenoscopy biopsy revealed poorly differentiated metastatic adenocarcinoma. (B) Oblique fused PET/CT images of same region demonstrate conclusively that the abnormal 18F-fluorodeoxyglucose activity seen on PET fuses to the gastric primary tumor (standardized uptake value maximum of 4.4) and the peritoneal implants (standardized uptake value maximum of 7.6).
Conclusion
18F-FDG-PET alone has a limited role in detecting and evaluating the local extent of primary gastric cancer with a low detection rate of early gastric cancer and variable uptake depending on histological subtype. PET/CT has a better positive predictive value for lymph node metastasis compared with CT alone, with reasonable sensitivity for liver and lung metastasis. 18F-FDG-PET provides valuable information on tumor aggressiveness and estimation of prognosis. The combination of PET/ CT overcoming the limitations of either modality alone and improving the system resolution will be more valuable in preoperative staging of gastric cancer, especially in evaluation of distant metastases.
Future perspective
PET/CT imaging in gastric cancer, with increased accuracy in preoperative staging precluding unnecessary surgery and increased sensitivity for detection of recurrence in postoperative follow-up of patients, will be utilized more in the future in the correct clinical setting.
Prediction of early response to therapy for 18F-FDG-avid tumors, which helps to estimate prognosis and survival rates, will help to monitor individualized therapy, preventing unnecessary prolonged chemotherapy and therapy-related toxicity.
Clinical utility of PET/CT will increase if the patient population is selected carefully. The selection parameters would depend on histopathological subtype (intestinal vs nonintestinal); stage (early vs advanced gastric cancer); and according to the baseline 18F-FDG tumor uptake (high vs low), which would increase the test sensitivity in detection rate of metastasis and prediction of therapy response.
Depending on histopathological subtype of tumor, new tracers such as FLT-PET and possibly new tracers and metabolic agents being developed may play a role to further increase PET usage. We still need better tracers for gastric cancer.
The clinical utility of PET/CT will further increase with application of better techniques such as good gastric distention and with future developments in dedicated PET/CT imaging systems with higher spatial resolution and faster imaging.
Financial & competing interests disclosure
The authors have no relevant affiliations or financial involvement with any organization or entity with a financial interest in or financial conflict with the subject matter or materials discussed in the manuscript. This includes employment, consultancies, honoraria, stock ownership or options, expert testimony, grants or patents received or pending, or royalties.
No writing assistance was utilized in the production of this manuscript.

References
Papers of special note have been highlighted as:
• • of considerable interest
- Haberman CR, Weiss F, Riecken R et al. Preoperative staging of gastric adenocarcinoma: comparison of helical CT and endoscopic US. Radiology 230, 465–471 (2004).
- Hwang SW, Lee DH, Lee SH et al. Preoperative staging of gastric cancer by endoscopic ultrasonography and multidetectorrow computed tomography. J. Gastroenterol. Hepatol. 25(3), 512–518 (2010).
- Horton KM, Fishman EK. Current role of CT in imaging of the stomach. Radiographics 23, 75–87 (2003).
- Gore RM. Upper gastrointestinal tract tumors: diagnosis and treating strategies. Cancer Imaging 5, 95–98 (2005).
- Kim HJ, Kim AY, Oh ST et al. Gastric cancer staging at multi-detector row CT gastrography: comparison of transverse and volumetric CT scanning. Radiology 236, 879–885 (2005).
- Sarela AI, Miner TJ, Karpeh MS et al. Clinical outcomes with laparoscopic stage M1, unresected gastric adenocarcinoma. Ann. Surg. 243, 189–195 (2006).
- Kim EY, Lee WJ, Choi D et al. The value of PET-CT for preoperative staging of advanced gastric cancer: comparison with contrastenhanced CT. Eur. J. Radiol. 79, 183–188 (2011).
- Park MJ, Lee WJ, Lim HK, Park KW, Choi JY, Kim BT. Detecting recurrence of gastric cancer: the value of FDG PET/CT. Abdom. Imaging 34, 441–447 (2009).
- Nakamoto Y, Togashi K, Kaneta T et al. Clinical value of whole body PET for recurrent gastric cancer: a multicenter study. Jap. J. Clin. Oncol. 39(5), 297–302 (2009).
- Sun L, Su XH, Guan YS et al. Clinical role of 18F fluorodeoxyglucose positron emission tomography/computed tomography in post-operative follow up of gastric cancer: initial results. World J. Gastroenterol. 14(29), 4627–4632 (2008).
- Potter TD, Flamen P, Cutsem V et al. Whole body PET with FDG for the diagnosis of recurrent gastric cancer. Eur. J. Nucl. Med. 29(4), 525–529 (2002).
- Stahl A, Ott K, Weber WA et al. FDG PET imaging of locally advanced gastric carcinomas: correlation with endoscopic and histopathological findings. Eur. J. Nucl. Med. 30(2), 288–295 (2003).
- Salaun PY, Grewal RK, Dodamane I et al. An analysis of the 18F-FDG uptake pattern in the stomach. J. Nucl. Med. 46, 48–51 (2005).
- Koga H, Sasaki M, Kuwabara Y et al. An analysis of the physiological FDG uptake pattern in the stomach. Ann. Nucl. Med. 17(8), 733–738 (2003).
- Kamimura K, Fujita S, Nishii R et al. An analysis of physiological FDG uptake in the stomach with the water gastric distention method. Eur. J. Nucl. Med. Mol. Imaging 34, 1815–1818 (2007).
- Imperiale A, Cimarelli S, Sellem DB, Blondet C, Contantinesco A. Focal F-18 FDG uptake mimicking malignant gastric localizations disappearing after water ingestion on PET/CT images. Clin. Nucl. Med. 31(12), 835–837 (2006).
- Tian J, Chen L, Wei B et al. The value of vesicant 18F-FDG PET in gastric malignancies. Nucl. Med. Commun. 25, 825–831 (2004).
- Kamimura K, Nagamachi S, Wakamatsu H et al. Role of gastric distention with additional water in differentiating locally advanced gastric carcinomas from physiological uptake in the stomach on 18F-fluoro-2-deoxy-dglucose PET. Nucl. Med. Commun. 30, 431–439 (2009).
- Yun M, Choi HS, Yoo E, Bong JK, Ryu YH, Lee JD. The role of gastric distention in differentiating recurrent tumor from physiological uptake in the remnant stomach on 18F-FDG PET. J. Nucl. Med. 46, 953–957 (2005).
- Chen J, Cheong JH, Yun MJ et al. Improvement in preoperative staging of gastric adenocarcinoma with positron emission tomography. Cancer 103, 2383– 2390 (2005).
- Dassen AE, Lips DJ, Hoekstra CJ et al. FDGPET has no definite role in preoperative imaging in gastric cancer. Eur. J. Surg. Oncol. 35, 449–455 (2009).
- Yoshioka T, Yamaguchi K, Kubota K et al. Evaluation of F18-FDG PET in patients with advanced, metastatic, or recurrent gastric cancer. J. Nucl. Med. 44, 690–699 (2003).
- Mochiki E, Kuwano H, Katoh H, Asao T, Oriuchi N, Endo K. Evaluation of 18F-2- deoxy-2-fluoro-d-glucose positron emission tomography for gastric cancer. World J. Surg. 28, 247–253 (2004).
- Yamada A, Oguchi K, Fukushima M, Imai Y, Kadoya M. Evaluation of 2-deoxy-2-18F fluoro-d-glucose positron emission tomography in gastric carcinoma: relation to histological subtypes, depth of tumor invasion, and glucose transporter-1 expression. Ann. Nucl. Med. 20, 597–604 (2006).
- Herrmann K, Ott K, Buck AK et al. Imaging gastric cancer with PET and the radiotracers 18F-FLT and 18F-FDG: a comparative analysis. J. Nucl. Med. 48, 1945–1950 (2007).
- Kameyama R, Yamamato Y, Izuishi K et al. Detection of gastric cancer using 18F-FLT PET: comparison with 18F-FDG PET. Eur. J. Nucl. Med. Mol. Imaging 36, 382–388 (2009).
- Takahashi H, Ukawa K, Ohkawa N et al. Significance of F18-2-deoxy-2-fluoro-glucose accumulation in the stomach on positron emission tomography. Ann. Nucl. Med. 23, 391–397 (2009).
- Kim SK, Kang KW, Lee JS et al. Assessment of lymph node metastases using 18F-FDG PET in patients with advanced gastric cancer. Eur. J. Nucl. Med. Mol. Imaging 33, 148–155 (2006).
- Stahl A, Ott K, Schwaiger M, Weber WA. Comparison of different SUV based methods for monitoring cytotoxic therapy with FDG PET. Eur. J. Nucl. Med. Mol. Imaging 31, 1471–1479 (2004).
- Wieder HA, Ott K, Lordick F et al. Prediction of tumor response by FDG-PET: comparison of the accuracy of single and sequential studies in patients with adenocarcinoma of the esophagogastric junction. Eur. J. Nucl. Med. Mol. Imaging 34, 1925–1932 (2007).
- Ott BK, Fink U, Becker K et al. Prediction of response to preoperative chemotherapy in gastric carcinoma by metabolic imaging: results of a prospective trial. J. Clin. Gastric Cancer 10, 221–227 (2007).
- Di Fabio F, Pinto C, Rojas Llimpe FL et al. The predictive value of 18F-FDG-PET early evaluation in patients with metastatic gastric adenocarcinoma treated with chemotherapy plus cetuximab. Gastric Cancer 10(4), 221–227 (2007).
- Mukai K, Ishida Y, Okajima K, Isozaki H, Morimoto T, Nishiyama S. Usefulness of preoperative FDG-PET for detection of gastric cancer. Gastric Cancer 9, 192–196 (2006).
- Shoda H, Kakugawa Y, Saito D et al. Evaluation of 18F-2-deoxy-2-fluoro-glucose positron emission tomography for gastric cancer screening in asymptomatic individuals undergoing endoscopy. Br. J. Cancer 97, 1493–1498 (2007).
- Yun M, Lim JS, Noh SH et al. Lymph node staging of gastric cancer using 18F-FDG PET: a comparison study with CT. J. Nucl. Med. 46, 1582–1588 (2005).
- Kwee RM, Kwee TC. Imaging in assessing lymph node status in gastric cancer. Gastric Cancer 12, 6–22 (2009).
- Shiraishi N, Inomata M, Osawa N, Yasuda K, Adachi Y, Kitano S. Early and late recurrence after gastrectomy for gastric carcinoma. Univariate and multivariate analyses. Cancer 89, 255–261 (2000).
- Chen CY, Hsu JS, Wu DC et al. Gastric cancer: preoperative local staging with 3D multidetector row CT-correlation with surgical and histopathologic results. Radiology 242, 472–482 (2007).
- Lim JS, Yun MJ, Kim MJ et al. CT and PET in stomach cancer: preoperative staging and monitoring of response to therapy. Radiographics 26, 143–156 (2006).
- Turlakow A, Yeung HW, Salmon AS, Macapinlac HA, Larson SM. Peritoneal carcinomatosis: role of 18F-FDG PET. J. Nucl. Med. 44, 1407–1412 (2003).
- Sim SH, Kim YJ, Oh DY et al. The role of PET/CT in detection of gastric cancer recurrence. BMC Cancer 9, 73 (2009).
• • Good overview of multidetector CT technique and applications on gastric imaging.
• • One of the earliest papers on the use of PET/CT for preoperative staging of gastric cancer and in comparison to multidetector CT.
• • One of the early few studies on PET/CT in patients with suspected recurrent gastric cancer with a high positive predictive value.
• • Multicenter study on clinical usefulness of 18F-fluorodeoxyglucose‑PET in patients with suspected recurrent gastric cancer, demonstrating change in therapeutic management in up to 48% of the patients in a selected patient group.
• • Valuable information is provided improving the detection rate with additional water and regional imaging with increased specificity, positive predictive value and accuracy.
• • Very good prospective study with combined virtual gastroscopy and dynamic contrastenhanced multiplanar reformations illustrated with very good cases and detailed teachings on preoperative gastric cancer imaging.
• • Excellent overview on preoperative staging of gastric cancer and follow-up supported with excellent case examples.