Review Article - Imaging in Medicine (2011) Volume 3, Issue 5
Role of ultrasound in the diagnosis of testicular lesions
Phillip FC Lung1 & Paul S Sidhu†11King’s College London, Department of Radiology, King’s College Hospital, Denmark Hill, London SE5 9RS, UK
- Corresponding Author:
- Paul S Sidhu
King’s College London
Department of Radiology, King’s College Hospital
Denmark Hill, London SE5 9RS, UK
Tel: +44 203 299 4164
Fax: +44 203 299 3157
E-mail: paulsidhu@nhs.net
Abstract
Ultrasound is the main imaging technique in the assessment of the scrotal contents, providing unparalleled imaging of exquisite resolution. Ultrasound is effectively the sole scrotal imaging technique that a patient will undergo prior to definitive surgery. The accurate assessment of focal intratesticular lesions in the adult patient is of paramount importance, although most focal lesions will be malignant and require an orchidectomy, the recognition of the benign entity is essential to the individual patient. This article covers the salient features of benign and malignant focal intratesticular lesions to allow a confident diagnosis and to prevent unnecessary orchidectomy. We detail the range of intratesticular malignancies from germ cell tumors to lymphoma and metastases. Operator confidence is necessary to establish a benign diagnosis and we highlight important benign lesions that may benefit from a ‘watch and wait’ policy, such as infarction, abscess formation, focal orchitis, rete testis; as well as more bizarre unusual pathologies: intratesticular varicocele and adrenal rest tumors to name but a few. New ultrasound imaging techniques will be reviewed and their role discussed.
Keywords
color Doppler ▪ contrast-enhanced ultrasound ▪ elastography ▪ germ-cell tumor ▪ seminoma ▪ testis ▪ tumors ▪ ultrasound
Scrotal lesions present in many different ways, with the patient often locating a ‘lump’ or experiencing nonlocalizing pain. These patients will be investigated through a combination of clinical examination and radiological assessment. Ultrasound is the imaging modality of choice, and is exceptional in being able to rapidly establish the site of the scrotal lesion, either intra- or extratesticular, which is crucial with extratesticular lesions in the adult patient typically being benign. Despite the diagnostic challenge presented by intratesticular lesions, the combination of B-mode and color Doppler ultrasound commonly provides sufficient information to differentiate between benign and malignant disease. We will detail the typical findings seen in a range of intratesticular lesions in order to allow the reader to understand ultrasound features that indicate a benign rather than a malignant intratesticular lesion.
Intratesticular malignant tumors
There has been an unexplained increase in the prevalence of testicular carcinoma over the last 70 years, and testicular carcinoma is predominantly a cancer of white males [1]. In the UK, testicular cancer accounts for approximately 1% of all male cancers with just under 2000 new cases per year, although geographical variations exist with the highest incidence recorded in Denmark, with an incidence approaching six per 100,000 patients [2]. Testicular cancer most commonly occurs in young and middle aged men, affecting men between the ages of 20 and 45 years of age, with older men more susceptible to lymphoma, leukemia or metastatic disease. Although the prognosis of testicular cancer has improved markedly with the majority being curable, early identification of these tumors remains essential. Germ cell tumors constitute 95% of all testicular tumors and are divided into seminomas and nonseminomas [3]. The remainder of the testicular tumors are mainly made up of lymphoma (4%) with the rest a mixture of tumors of rare etiology. Risk factors for testicular tumors include cryptorchidism, infertility and siblings with testicular cancer [4].
The resolution afforded by B-mode imaging provides ultrasound with a high sensitivity for the detection of testicular masses. Nonetheless, it is the detection of vascularity with color Doppler that helps to differentiate benign (invariably avascular) from malignant (increase in vascularity and changes in the pattern of vascularity) condition [5]. Therefore a combination of B-mode ultrasound features and color Doppler findings will invariably lead to the correct diagnosis. A review of the salient features of a number of these focal lesions is detailed later.
■ Seminomatous germ cell tumor
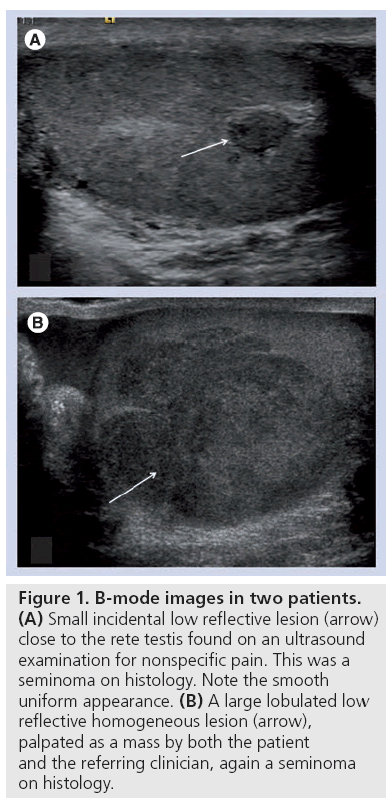
Seminomas represent 40–50% of the germ cell tumors with a peak incidence in the 35–45-year age group [6]. At presentation, approximately 75% of cases are limited to the testis, with retroperitoneal lymphadenopathy in 20% and extranodal metastases in 5% [7]. Bilateral disease may occur in 2% of cases and are then nearly always asynchronous. A seminoma most commonly presents with early disease and also boasts survival rates in excess of 95%. Human chorionic gonadotrophin is produced by both seminomas and nonseminomas, whereas the detection of α-fetoprotein can be used to differentiate between the two, as it is not secreted by pure seminomas. On B-mode ultrasound, a seminoma is usually homogeneous and of low reflectivity, which corresponds to the histological findings of uniform cells containing clear cytoplasm [8]. They vary in size from small incidental nodules to large infiltrative masses replacing the entire testicular parenchyma (Figure 1). The majority display a well-defined margin and cystic elements are occasionally seen with a reported incidence of 10% [8]. Vascularity is usually readily visualized with color Doppler and where this is increased, malignancy is likely, although small tumors may not display color flow [5].
Figure 1: B-mode images in two patients. (A) Small incidental low reflective lesion (arrow) close to the rete testis found on an ultrasound examination for nonspecific pain. This was a seminoma on histology. Note the smooth uniform appearance. (B) A large lobulated low reflective homogeneous lesion (arrow), palpated as a mass by both the patient and the referring clinician, again a seminoma on histology.
■ Nonseminomatous germ cell tumor
Embryonal carcinoma, choriocarcinoma, teratoma and yolk sac tumors all belong to the nonseminomatous germ cell tumors (NSGCTs) and are usually found in combination as mixed germ cell tumors with common combined configurations such as; teratomas and embryonal carcinoma (25%) or embryonal carcinoma and seminomas (15%) or teratoma, embryonal carcinoma and seminomas (15%) [6]. NSGCTs account for 60% of the germ cell tumors: 40–45% of GCTs contain only nonseminomatous elements, whilst approximately 20% are mixed and contain both seminomatous and nonseminomatous cells; these are treated as NSGCTs. NSGCTs occur earlier than the seminomas, with a peak incidence in the 20–35-year age group; the mixed seminomatous/nonseminomatous group maintain an incidence inbetween the two groups.
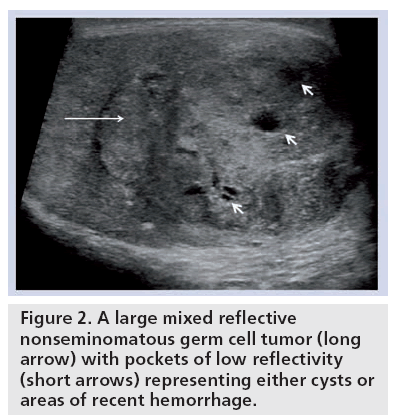
The ultrasound features on B-mode ref lect the diversity of the underlying cell type of the NSGCTs and tend to be much more heterogeneous in nature, with mixed reflectivity, areas of necrosis, hemorrhage and calcification (Figure 2). In common with all malignant intratesticular tumors, the tumor, if large enough, will display an increase in color Doppler flow. If the cell type of the NSGCTs is more pure, then different patterns on the B-mode ultrasound may emerge. Details for these appearances, incidence and associations are documented later.
Figure 2: A large mixed reflective nonseminomatous germ cell tumor (long arrow) with pockets of low reflectivity (short arrows) representing either cysts or areas of recent hemorrhage.
Embryonal carcinoma
An embryonal carcinoma may represent the stem cell for all NSGCTs. Pure embryonal carcinomas represent only 3% of the GCT population, but an embryonal carcinoma element is found in over 87% of mixed GCTs, which makes it only second to seminomas as the most common type of testicular tumor. As a pure tumor, an embryonal carcinoma is usually smaller than a seminoma, but is much more aggressive; which is evident at histology where invasion of the tunica may occur with irregular and illdefined margins. This is reflected on ultrasound where the mass appears heterogeneous with irregular borders. Hemorrhage and calcification commonly occur and are seen as echogenic foci within the lesion [9].
Yolk sac tumor
Pure forms of yolk sac tumors are almost completely confined to the pediatric population, with the majority occurring in children before 2 years of age. In the adult population, they exist as part of a mixed germ cell tumor in 45% of cases, similar to teratomas, and their presence engenders a poorer prognosis. The yolk sac elements secrete α-fetoprotein, which can be used to monitor treatment. On histology, hemorrhage, necrosis and cystic degeneration often occur. These elements are reproduced on ultrasound as heterogeneous masses that can contain cystic areas and echogenic foci of hemorrhage [10].
Teratoma
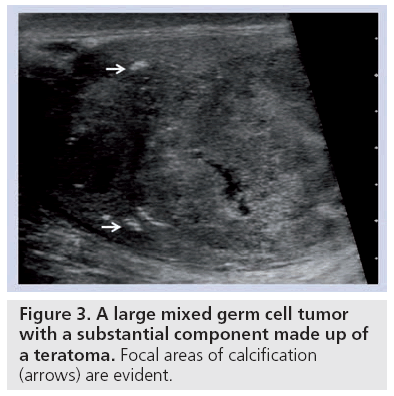
Teratomas are also common in the pediatric population as a pure type; whereas this is only found in 2–3% in adults. Teratomas are usually large at presentation and heterogeneous, comprising of all three germ cell layers, with solid and cystic components. This is evident on ultrasound as a large, well defined complex mass that commonly contains cysts. Regions of calcification, cartilage and fibrosis appear echogenic and may demonstrate acoustic shadowing. The presence of necrosis and hemorrhage is usually found in tumors containing embryonal carcinoma or choriocarcinoma. Ultrasound features are similar to the other NSGCTs but calcification may be a feature (Figure 3).
Choriocarcinoma
Choriocarcinoma is a rare, highly malignant tumor that accounts for less than 1% of GCTs in its pure form; 8% of mixed GCTs contain foci of choriocarcinoma. Human chorionic gonadotropin is produced and high levels indicate a poor prognosis. These tumors are typically small in size, but rapid growth may exceed the blood supply resulting in a scar, often in the presence of distant metastatic disease; this is often referred to as a ‘burned-out’ tumor [11]. Hemorrhagic necrosis is regularly seen appearing as areas of mixed reflectivity.
Lymphoma, leukemia & metastasis
Testicular involvement in acute leukemia is estimated at 64% and in chronic leukemia at 25%. Primary leukemia of the testis is rare, although it is a common site of recurrence in children.
The blood–testis barrier prevents chemotherapy agents from dealing with intratesticular leukemia cells. Testicular lymphoma may be the primary site of involvement, the initial manifestation of widespread disease or the site of recurrence. Testicular lymphoma, which accounts for 5% of testicular tumors, is the most common testicular tumor in the over 60-year-old age group, the most common secondary tumor of the testis and is the most common bilateral tumor. Metastasis to the testis is unusual, with the most frequent primary sites being the prostate, lung, melanoma, colon and kidney. Metastases occur most commonly in patients over 50 years of age. All three entities may be indistinguishable on ultrasound from a primary tumor [12,13].
Intratesticular benign tumors
■ Sex cord stromal tumors
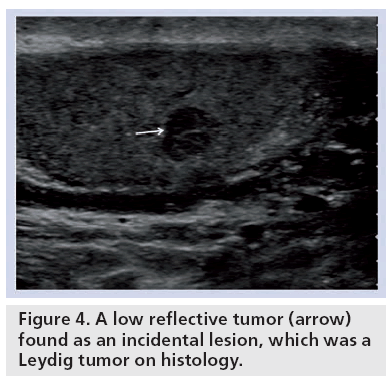
Whilst 90% of the sex cord stromal tumors are benign (of which the two most important are Leydig cell and Sertoli cell tumors), 10% exhibit malignant behavior. There is no radiological finding that enables reliable differentiation between the two tumors [8] and, as a result, all undergo excision; 4% of testicular malignancies are sex cord stromal tumors. Leydig cell tumors account for the majority in this group and arise from the interstitial components of the sex cords [14]. These tumors occur at any age, although the majority of cases are between 20 and 60 years of age. Leydig cell tumors present with a testicular mass in 43% of cases, but they may manifest clinically as gynecomastia, reduced libido or precocious puberty in as many as 30% due to their capacity to secrete androgens and/ or estrogens. They are usually smaller than 5 cm and appear as hypoechoic lesions on ultrasound (Figure 4). Necrosis, hemorrhage and cyst formation may occur and this variation makes Leydig cell tumors similar in ultrasound appearance to that of germ cell tumors.
Sertoli cell tumors are derived from the sex cords and may also secrete androgens or estrogens, but whilst occasionally causing gynecomastia, they are often not produced in sufficient quantities to be hormonally active. Their appearances are typically well-defined, rounded masses, but have been reported as causing a diffuse heterogeneous enlargement of the testis [15]. The large-cell calcifying Sertoli cell tumor is a benign variant in which approximately 30% are associated with a genetic syndrome; Peutz-Jeghers and Carney syndromes being the most common. B-mode characteristically demonstrates considerable areas of calcification.
Other intratesticular benign focal lesions
■ Testicular calcification
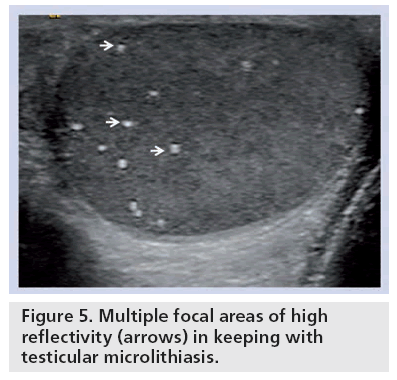
Testicular microlithiasis (TM) can be defined as the intratesticular presence of small echogenic foci measuring 1–3 mm and is characterized by microcalcification within the seminiferous tubules with a prevalence of 2%. The appearances of TM range from several scattered echogenic foci to diffuse areas of calcification (Figure 5). They can be differentiated from other forms of intratesticular calcification by the lack of acoustic shadowing, likely due to their small size. There is a suggestion of an increase in the prevalence of testicular cancer with TM with several studies reporting this association [16]. However, recently, an increase in the prevalence of testicular tumor with any form of calcification has been suggested [17]. It is not apparent if there is a risk of developing a malignancy in the presence of TM, and as such in the absence of any evidence to the contrary, follow-up of these patients is advocated [18].
■ Intratesticular cysts
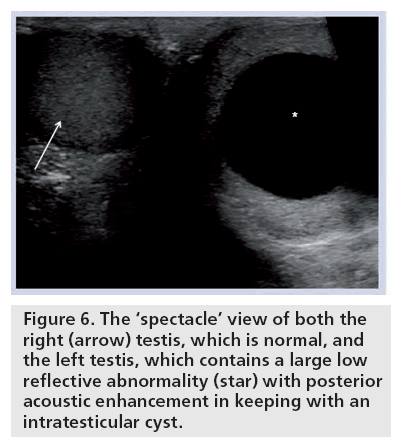
The incidental finding of a simple intratesticular cyst is common, ranging from 2 to 10% [19]. These cysts are usually asymptomatic (Figure 6). The cysts are impalpable and tend to be located in a subcapsular position near the mediastinum testis. The appearances are of a solitary, anechoic round cyst enclosed by an indiscernible wall and displaying posterior acoustic enhancement. To enable a confident diagnosis of a simple cyst, it is important to ensure that the cyst does not contain any solid components and that the wall is not thickened and irregular. If these features are identified, a cystic testicular tumor, such as a teratoma, should be considered. There should be no evidence of color Doppler flow to the fluid component of the cyst, but care is needed to avoid artifact from acoustic streaming [20].
■ Epidermoid cysts
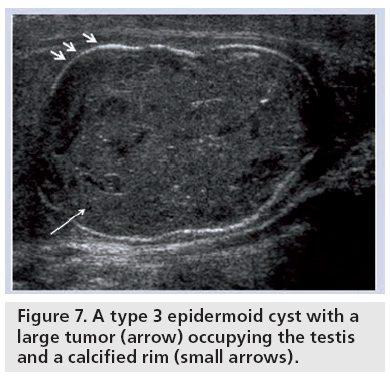
A total of 1% of all testicular tumors are epidermoid cysts making it the most common benign testicular neoplasm [21]. Epidermoid cysts are filled with layers of keratin enclosed within a fibrous capsule. Ultrasound appearances are classified into four types [22]: type 1 is the classic onion skin pattern with concentric echogenic rings; type 2 is that of a densely calcified mass demonstrating significant acoustic shadowing concealing the presence of a cyst; type 3 cysts display rim, peripheral or central calcification; and Type 4 is a mixed type with a heterogeneous echo pattern and ill-defined margins (Figure 7). The dilemma posed by the discovery of an epidermoid cyst is the possible simulation of these appearances by a malignant process. Indeed, similar appearances have been reported in teratomas [23]. Features that support a benign process include lack of vascularity on color Doppler and negative tumor markers. Current management varies from a conservative ultrasound follow-up approach, enucleating the tumor to an orchidectomy.
Figure 7: A type 3 epidermoid cyst with a large tumor (arrow) occupying the testis and a calcified rim (small arrows).
■ Testicular infarction
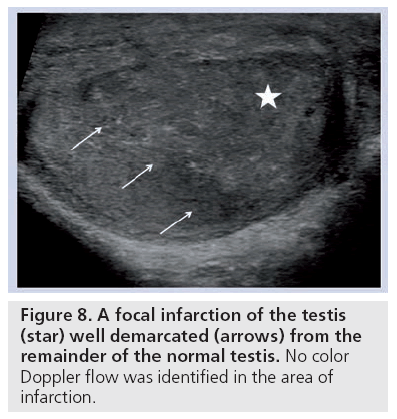
In the past, radionuclide scintigraphy was utilized to assess testicular perfusion in patients with suspected testicular infarction. With the advances in color Doppler technology, ultrasound has replaced nuclear medicine as the investigation of choice. Segmental testicular infarction is a rare disorder classically presenting with acute scrotal pain and is secondary to a variety of conditions including epididymoorchitis, trauma and sickle cell disease but may also be idiopathic [24]. Whilst the B-mode appearances of segmental infarction are variable, the key to making the diagnosis is the feature of reduced or absent color Doppler flow. Typical features include a wedge shaped or round lesion of mixed or low echogenicity; they are usually solitary and may be found in either testis; rarely, they may appear to exert mass effect simulating a testicular tumor (Figure 8). Venous infarction is a rare complication in cases of severe epididymo-orchitis where the progressive worsening of edema produces venous impairment culminating in ischemia. The ultrasound changes reflect the underlying pathology with an initial generalized reduction in reflectivity representing edema with focal areas becoming increasingly heterogeneous and hypoechoic. These round hypoechoic lesions do not demonstrate color Doppler flow and in time, will regress. In prolonged cases, there may be evidence of hemorrhage or abscess formation, which may necessitate orchidectomy. Testicular tumor is the primary differential for testicular infarction and can be distinguished by an absence of color Doppler flow in an area of infarction. However, it is again important to ensure that the clinical history is compatible and close follow-up should confirm that the lesion resolves. Severe orchitis, a consequence of progression from epididymitis may present in a variety of forms, and may be focal in nature simulating a testicular tumor, with an increase in color Doppler flow and must be differentiated from tumors with follow-up ultrasound imaging [25].
■ Intratesticular varicocele
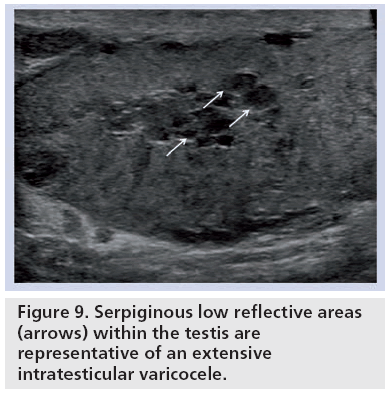
An intratesticular varicocele carries an incidence of less than 2%, rising to 16% in those with infertility. They are located near the mediastinum testis and are serpiginous or ovoid in shape. In the presence of an associated extratesticular varicocele, continuity with the intratesticular segment may be demonstrable [26,27]. Vascularity on color Doppler with retrograde f low on Valsalva manoeuvre is characteristic (Figure 9).
■ Intratesticular hematoma
An intratesticular hematoma is an uncommon consequence to testicular trauma and a clear history of such is often elicited [28]. In the acute phase, the hematoma is seen as a heterogeneous area of mixed reflectivity; a hypoechoic rim is often seen representing edema. With time, the hematoma retracts and appears progressively more hypoechoic until it eventually resolves completely or becomes a scar. An essential feature is the absence of color Doppler flow within the lesion which, in combination with an appropriate clinical history and expected evolution on follow-up studies, allows differentiation from malignancy.
■ Intratesticular abscess
An intratesticular abscess is rare, but are most commonly seen following severe epididymoorchitis; other causes include trauma, mumps and infarction. Ultrasound appearances are typical for an abscess with a complex hypoechoic lesion surrounded by irregular and poorly defined walls. No color Doppler flow should be identifiable within the lesion. In severe cases with large abscess formation and little residual testicular parenchyma, orchidectomy may be performed.
■ Testicular adrenal rest tumors
In patients with a known diagnosis of congenital adrenal hyperplasia, the presence of eccentrically located low reflective masses should alert the clinician to the possibility of testicular adrenal rest tumors, with a prevalence as high as 94%; occasionally, the intratesticular mass may be the first manifestation of congenital adrenal hyperplasia. Testicular adrenal rest tumors are often lobulated, may display acoustic shadowing due to fibrosis and are found near the mediastinum testis [29]. Interrogation with color Doppler demonstrates a ‘spoke-wheel’ arrangement of increased blood flow.
■ Rete testis
Rete testis dilatation is a benign condition and is often found in a postinfectious or post-traumatic patient. Multiple round low reflective structures are arranged adjacent to the mediastinum testis, ranging in size from a few millimetres to as large as 7 cm [30]. The rete testis is characteristically avascular on color Doppler ultrasound differentiating this entity from an intratesticular varicocele. However, dilated rete testis has been described secondary to seminomas and teratoma, so careful inspection of the testis for an associated tumor is advocated.
Summary of ultrasound features
Ultrasound will invariably detect the intratesticular focal lesion, often as an incidental finding when the patient is examined for an unconnected reason. The large lesion is characterized on the B-mode ultrasound and the addition of color aids the interpretation into benign and malignant pathologies; albeit with some important caveats, particularly in focal orchitis. Many characteristics seen on B-mode imaging coupled with a relevant clinical history will establish the underlying cause. However when the lesion is small (<5 mm) it may not demonstrate sufficient color Doppler signal to establish the presence of vascularity, leaving the operator uncertain of the nature of the abnormality. Newer techniques such as contrast-enhanced ultrasound and even elastography may help.
Future perspective
The utility of ultrasound in the investigation of scrotal disease is well established, particularly in the ability to separate intra- and extratesticular lesions. Furthermore, the low cost, ubiquity of ultrasound and noninvasive nature, all contribute to an incomparable technique for scrotal imaging. The use of radionuclide scanning has become redundant with the advancement of color Doppler, whilst CT imaging is confined to staging of malignant disease. However, ultrasound is not without limitations. Ultrasound is highly operator dependent with diagnostic confidence linked to sonographic experience. Where a diagnosis is uncertain, there is a dearth of available alternative methods of investigation, usually resulting in unnecessary scrotal exploration. MRI has been touted as a potential problem solving alternative to ultrasound for the investigation of testicular lesions, being able to correctly identify intratesticular malignancy, with contrast-enhanced subtraction MRI able to determine testicular perfusion. Despite this, there has been no study performed comparing ultrasound to MRI in the assessment of focal testicular lesions [31]. This, together with high cost and potential complications of MR contrast, has limited the adoption of this technique. Contrast-enhanced ultrasound is a new technique taking advantage of intravenous microbubble contrast to provide information on vascular enhancement characteristics of testicular lesions. The advantages have already been proven in vascular and abdominal imaging, and can potentially improve upon the limitations of ultrasound [32,33]. This technique will allow for the very sensitive depiction of the presence of vascularity in a lesion, and perhaps differentiate the benign from the malignant. This technique has been reported as being successful in establishing the lack of vascularity in segmental testicular infarction [34]. A further development is the concept of tissue elastography, essentially the assessment of the ‘hardness’ of tissue. This technique has been used in the thyroid, prostate, liver and the breast but is as yet unproven in the testis [35]. Perhaps the absence of ‘hardness’ of an intratesticular lesion may aid in a more confident interpretation, allowing a ‘watchfulwaiting’ approach avoiding surgery.
Financial & competing interests disclosure
The authors have no relevant affiliations or financial involvement with any organization or entity with a financial interest in or financial conflict with the subject matter or materials discussed in the manuscript. This includes employment, consultancies, honoraria, stock ownership or options, expert testimony, grants or patents received or pending, or royalties.
No writing assistance was utilized in the production of this manuscript.
References
Papers of special note have been highlighted as:
• of interest
- Greenlee RT, Hill-Harmon MB, Murray T, Thun M. Cancer Statistics, 2001. CA Cancer J. Clin. 51, 15–36 (2001).
- Osterlind A. Diverging trends in incidence and mortality of testicular cancer in Denmark. Br. J. Cancer 53, 501–506 (1986).
- Dearnaley DP, Huddart RA, Horwich A. Managing testicular cancer. Br. Med. J. 322, 1583–1588 (2001).
- Horwich A, Shipley J, Huddart RA. Testicular germ cell cancer. Lancet 367, 754–765 (2006).
- Sidhu PS, Sriprasad S, Bushby LH, Sellars ME, Muir GH. Impalpable testis cancer. BJU Int. 93, 888 (2004).
- Ro JY, Kim KR, Ayala AG. Tumors of the male genital tract. In: Diagnostic Histopathology of Tumors. Fletcher CDM (Ed.). Elsiever, 749–879 (2007).
- Ulbright TM, Amin MB, Young RH. Miscellanous primary tumors of the testis, adenxa and spermatic cord. In: Atlas of Tumor Pathology. Rosai J, Sobin LH (Eds). Armed Forces Institute of Pathology, 235–366 (1999).
- Woodward PJ, Sohaey R, O’Donoghue MJ, Green DE. Tumors and tumor-like lesions of the testis: radiologic-pathologic correlation. Radiographics 22, 189–216 (2002).
- Dogra VS, Gottlieb RH, Oka M, Rubens DJ. Sonography of the scrotum. Radiology 227(1), 18–36 (2003).
- Frush DP , Sheldon CA. Diagnostic imaging for pediatric scrotal disorders. Radiographics 18, 969–985 (1998).
- Comiter CV, Renshaw AA, Benson CB, Loughlin KR. Burned-out primary testicular cancer: sonographic and pathological characteristics. J. Urol. 156, 85–88 (1996).
- Rayor RA, Scheible W, Brock WA, Leopold GR. High resolution ultrasonography in the diagnosis of testicular relapse in patients with acute lymphoblastic leukemia. J. Urol. 128, 602–603 (1982).
- Garcia-Gonzalez R, Pinto J, Val-Bernal JF. Testicular metastases from solid tumors: an autopsy study. Ann. Diagn. Pathol. 4, 397–400 (2000).
- Kim I, Young RH, Scully RE. Leydig cell tumors of the testis: a clinicopathological analysis of 40 cases and review of the literature. Am. J. Surg. Pathol. 9, 177–192 (1985).
- Lui P, Thorner P. Sonographic appearance of Sertoli cell tumor with pathologic correlation. Pediatr. Radiol. 23, 127–128 (1993).
- Miller FNAC, Sidhu PS. Does testicular microlithiasis matter? A review. Clin. Radiol. 57, 883–890 (2002).
- Miller FNAC, Rosairo S, Clarke JL, Sriprasad S, Muir GH, Sidhu PS. Testicular calcification and microlithiasis: association with primary intratesticular malignancy in 3,477 patients. Euro. Radiol. 17, 363–369 (2006).
- Backus ML, Mack LA, Middleton WD, King BF, Winter TC, True LD. Testicular microlithiasis: imaging appearances and pathologic correlation. Radiology 192, 781–785 (1994).
- Hamm B, Fobbe F, Loy V. Testicular cysts: differentiation with US and clinical findings. Radiology 168, 19–23 (1988).
- Clarke L, Edwards A, Graham E. Acoustic streaming: an in vitro study. Ultrasound in Med. Biol. 30, 559–562 (2004).
- Price EB. Epidermoid cysts of the testis: a clinical and pathologic analysis of 69 cases from the testicular tumor registry. J. Urol. 102, 708–713 (1969).
- Atchley JTM, Dewbury KC. Ultrasound appearances of testicular epidermoid cysts. Clin. Radiol. 55, 493–502 (2000).
- Maizlin ZV, Belenky A, Baniel J, Gottlieb P, Sandbank J, Strauss S. Epidermoid cyst and teratoma of the testis: sonographic and histologic similarities. J. Ultrasound Med. 24, 1403–1409 (2005).
- Bilagi P, Sriprasad S, Clarke J, Sellars M, Muir G, Sidhu P. Clinical and ultrasound features of segmental testicular infarction: six-year experience from a single centre. European Radiology 17, 2810–2818 (2007).
- Cook JL, Dewbury K. The changes seen on high-resolution ultrasound in orchitis. Clin. Radiol. 55, 13–18 (2000).
- Das KM, Prasad K, Szmigielski W, Noorani N. Intratesticular varicocele: evaluation using conventional and Doppler sonography. Am. J. Roentgenol. 173, 1079–1083 (1999).
- Stewart VR , Sidhu PS. The testis: the unusual, the rare and the bizarre. Clin. Radiol. 62, 289–302 (2007).
- Purushothaman H, Sellars ME, Clarke JL, Sidhu PS. Intratesticular hematoma: differentiation from tumor on clinical history and ultrasound appearances in two cases. Br. J. Radiol. 80, E184–E187 (2007).
- Avila NA, Premkumar A, Shawker TH, Jones JV, Laue L, Cutler GB. Testicular adrenal rest tissue in congenital adrenal hyperplasia: findings at gray-scale and color Doppler US. Radiology 198, 99–104 (1996).
- Sellars MEK, Sidhu PS. Pictorial review: ultrasound appearances of the rete testis. Eur. J. Ultrasound 14, 115–120 (2001).
- Watanabe Y, Dohke M, Ohkubo K et al. Scrotal disorders: evaluation of testicular enhancement patterns at dynamic contrastenhanced subtraction MRI. Radiology 271, 219–227 (2000).
- Sidhu PS, Allan PL, Cattin F et al. Diagnostic efficacy of SonoVue(R), a second generation contrast agent, in the assessment of extracranial carotid or peripheral arteries using colour and spectral Doppler ultrasound: a multicentre study. Br. J. Radiol. 79, 44–51 (2006).
- Claudon M, Cosgrove D, Albrecht T et al. Guidelines and good clinical practice recommendations for contrast enhanced ultrasound (CEUS) – update 2008. Ultraschall in Med. 29, 28–44 (2008).
- Bertolotto M, Derchi LE, Sidhu PS et al. Acute segmental testicular infarction at contrast-enhanced ultrasound: early features and changes during follow-up. AJR Am. J. Roentgenol. 196, 834–841 (2011).
- Li Y, Snedeker JG. Elastography: modalityspecific approaches, clinical applications, and research horizons. Skeletal Radiol. 40, 389–397 (2010).
• Good review of the ultrasound findings of testicular tumors with histological correlation.
• Good review of the multitude of abnormalities that affect the scrotum, with their clinical presentation and ultrasound appearances well described.
• Good review of the ultrasound findings of testicular microlithiasis and the significance of this abnormality.
• Important overview of the prevalance of all types of testicular microlithiasis.
• Review of the color Doppler findings in segmental infarction with a good review of the literature.
• Excellent review of unusual testicular findings.
• Details the use of contrast-enhanced ultrasound in the evaluation of absence of vascularity in the testis.