Review Article - Journal of Experimental Stroke & Translational Medicine (2009) Volume 2, Issue 2
Long-term survival and serial assessment of stroke damage and recovery practical and methodological considerations
- *Corresponding Author:
- Dr Mike Modo
Centre for the Cellular Basis of Behaviour
The James Black Centre, Kings College London
125 Coldharbour Lane London SE5 9NU United Kingdom
Tel: +44 207 848 5315
Fax: +44 207 848 5309
Email: mike.modo@kcl.ac.uk
Abstract
Impairments caused by stroke remain the main cause for adult disability. Despite a vigorous research effort, only 1 thrombolytic treatment has been approved in acute stroke (<3h). The limitations of preclinical studies and how these can be overcome have been the subject of various guidelines. However, often these guidelines focus on the acute stroke setting and omit long-term outcome measures, such as behaviour and neuroimaging. The considerations and practicalities of including the serial assessment of these approaches and their significance to establish therapeutic efficacy are discussed here.
Keywords
Stroke, middle cerebral artery occlusion, post-operative care, neurological score, behavioural battery, MRI
Abbreviations
CT- Computer tomography
EEG – Electroencephalography
fMRI – functional Magnetic Resonance Imaging
MCA – Middle Cerebral Artery
MRI – Magnetic Resonance Imaging
PD - Parkinson’s Disease
PET –Positron Emission Tomography
phMRI – pharmacological Magnetic Resonance Imaging
rtPA – recombinant tissue-type Plasminogen Activator
SPECT – Single Photon Emission Computer Tomography
Introduction
The development of novel treatments for stroke remains a major challenge for scientists and clinicians alike. Although other neurological conditions, such as Parkinson’s disease (PD), have seen the development of pharmacological compounds that at least alleviate the behavioural impairments temporarily, no such success has been achieved for ischaemic stroke, with recombinant tissue-type plasminogen activator (rtPA) remaining the only clinically-approved intervention. This is despite excellent animal models of stroke that more accurately replicate the proximal cause and pathology of the human condition compared to other disease, such as a PD. It is therefore important to understand why compounds showed promise in pre-clinical models, but failed to achieve similar success in clinical trials (Savitz 2007; Wahlgren and Ahmed 2004; Zaleska et al. 2009).
Several initiatives (Chopp et al. 2009; Feuerstein et al. 2008; Fisher 2003; Fisher et al. 2005; Fisher et al. 2009; Liu et al. 2009; Stem Cell Therapies as an Emerging Paradigm in Stroke (STEPS): bridging basic and clinical science for cellular and neurogenic factor therapy in treating stroke 2009) have compiled limitations and omissions in preclinical studies that raised concerns about the studies’ reliability and validity, but also about its outcome measures and the timing of intervention. Their guidelines aim to improve the pre-clinical testing of therapeutic efficacy in stroke by failing unsuccessful interventions, while increasing the likelihood of successful compounds to succeed in the clinic. Nevertheless, the focus is often on a single short-term ex vivo outcome measure, such as histological volume reduction, that differs considerably from clinical outcome measures. Although this might provide an indication that a therapeutic reduces the impact of stroke, this does not necessarily translate into a behavioural benefit.
The validity of measures and their sensitivity to detect therapeutic efficacy are often poorly established. To determine if an intervention was not successful rather than outcome measures not being sensitive to the therapy, ideally within each study a validation of outcome measures using a positive control should be undertaken (Ashioti et al. 2009). Generally, there is a good correlation between ex vivo histological lesion volumes and in vivo behavioural tests or neuroimaging (Ashioti et al. 2007). Nevertheless, brain plasticity can produce recovery on neurological or behavioural tests without a concomitant improvement in lesion volume.
Merely assessing absolute lesion volume disregards the site of the lesion and omits its impact on the remaining brain. Focussing on lesion volume as a whole might average-out any regional neuroprotection. Neuroimaging is emerging though as an integrative assessment that captures the more complete anatomical impact of stroke (Liebeskind 2009). Lesion volume by MRI robustly correlates with behavioural measurements in animals (Ashioti et al. 2007; Lim et al. 2008; Virley et al. 2000) and neurological/neuropsychological assessments in patients (Committeri et al. 2007). Additional measurements of anatomical changes in spared brain regions (i.e. where blood flow has been normal) can further elucidate the secondary impact stroke exerts on other regions, such as the thalamus, but also on the non-injured hemisphere (Modo et al. 2009). Changes in these areas can be crucial to explain behavioural improvement in stroke. Neuroimaging can hence provide a common assessment platform across species and eventually deliver validated surrogate markers of treatment efficacy (Liebeskind 2009).
To ensure that treatment is robust, experiments should not only focus on the acute short-term benefit (<48h), but also the sub-acute (2-14 days) and chronic period (>14 days). It is noteworthy that the lesion volume following 60 minutes of middle cerebral artery occlusion (MCAo) increases up to 6 weeks following insult before it stabilises (Modo et al. 2009). Hence, one would assume that as the lesion evolves the benefit from a continued treatment should become more apparent. Especially interventions impacting on secondary degeneration, such as Wallerian degeneration, will only be evident with long-term assessments (Yu et al. 2009). Therefore, serial in vivo assessments over longer time frames can be more powerful to detect therapeutic effects compared to analysis of a single acute time-point.
Neurological Scoring versus Behavioural Test Battery
There is a fundamental distinction between neurological deficits and behavioural impairments. Neurological deficits are specific functions, such as the righting reflex, that are perturbed, whereas behavioural impairments are more complex readouts, such as lack of learning in the water maze. For serial and long-term measurements, one also needs to consider if there is spontaneous recovery of this task. Most tasks will eventually recover long-term, although very specific and detailed analyses will allow the continued detection of a deficit or adaptations (Alaverdashvili et al. 2008). Another essential distinction is between behavioural and functional tests. Behavioural tests are tasks where an overt response of an animal is measured, whereas functional readouts reflect measurements where brain activity is assessed (e.g. fMRI, phMRI, EEG, electrophysiology). Functional tests therefore provide the link between anatomical and behavioural measures. Although functional and behavioural measures are often linked, it is conceivable that changes in the brain’s activity occur (i.e. plasticity) that do not directly translate into a behavioural benefit. For instance, the fMRI response to somatosensory stimulation sub-acutely (1-3 days) indicates activity in the contralateral cortex. However, there is no behavioural recovery associated with this change in activity and indeed animals only recover if activity is restored to areas adjacent to those damaged (Dijkhuizen et al. 2003). Conversely, it is possible that an animal shows “recovery” on a behavioural test due to adaptation that does not necessarily reflect a measurable functional change.
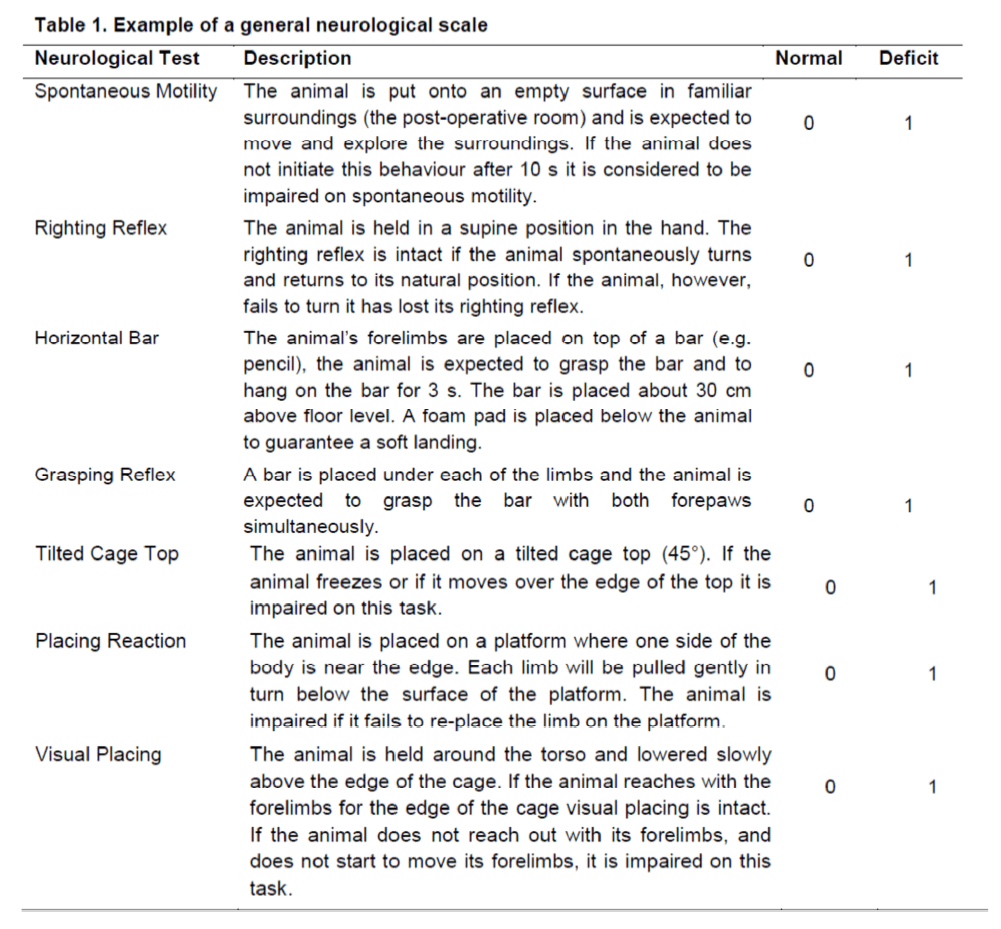
Neurological deficits are often used to probe outcomes in pre-clinical stroke studies. A series of specific tests can be charted into a neurological scale to provide a read-out of overall severity (Modo et al. 2000b). For this readout to be meaningful as a general measure of severity, it is important that each deficit carries equal weight and ideally that it is either impaired (1) or normal (0). The total score hence reflects the magnitude of a general deficit. Several scales that either measure global neurological function (Table 1) or more specific stroke-related deficits are documented in the literature. Tests that are especially important and sensitive to the damage caused by stroke are best assessed separately as a graded scale. For instance, one of the most commonly used stroke-specific scales consists of the 3 point Bederson scale (Bederson et al. 1986) (Table 2). This scale is sensitive to striatal damage and measures a forelimb dysfunction that is dependent on the degree of lesioning. Importantly, this neurological deficit is visible within 45 minutes of occlusion and can be used prior to re-perfusion to surmise if a MCA occlusion was successful.
The most comprehensive approach is to use two scales to measure neurological deficits. One scale should chart the overall neurological impact of the procedure, whereas another scale should measure the degree of deficit that is relevant to stroke. Combinations of general neurological dysfunction with stroke-specific deficits have been described (Hunter et al. 2000). Often these scales, however, give different weight to different tests and therefore the overall score cannot be considered an overall measure of severity. Although neurological scales typically correlate with short-term lesion volumes (Ashioti et al. 2007), they will no longer reflect the degree of histological damage at chronic time points, as most deficits spontaneously recover between a few days to a couple of weeks post-infarction with lesion volumes increasing (Modo et al. 2000b). As such, these neurological scales are only useful in the acute to sub-acute setting. However, appropriate controls consisting of stroke-only, stroke+vehicle and/or sham-surgery need to be incorporated to provide a meaningful assessment of therapeutic effects. Recovery on these scales can at worst merely indicate better spontaneous recovery or that an intervention improves recovery from the surgical procedure itself rather than the stroke. It is therefore best to consider with great caution claims of a persisting recovery, if only neurological scales have been used as read-outs.
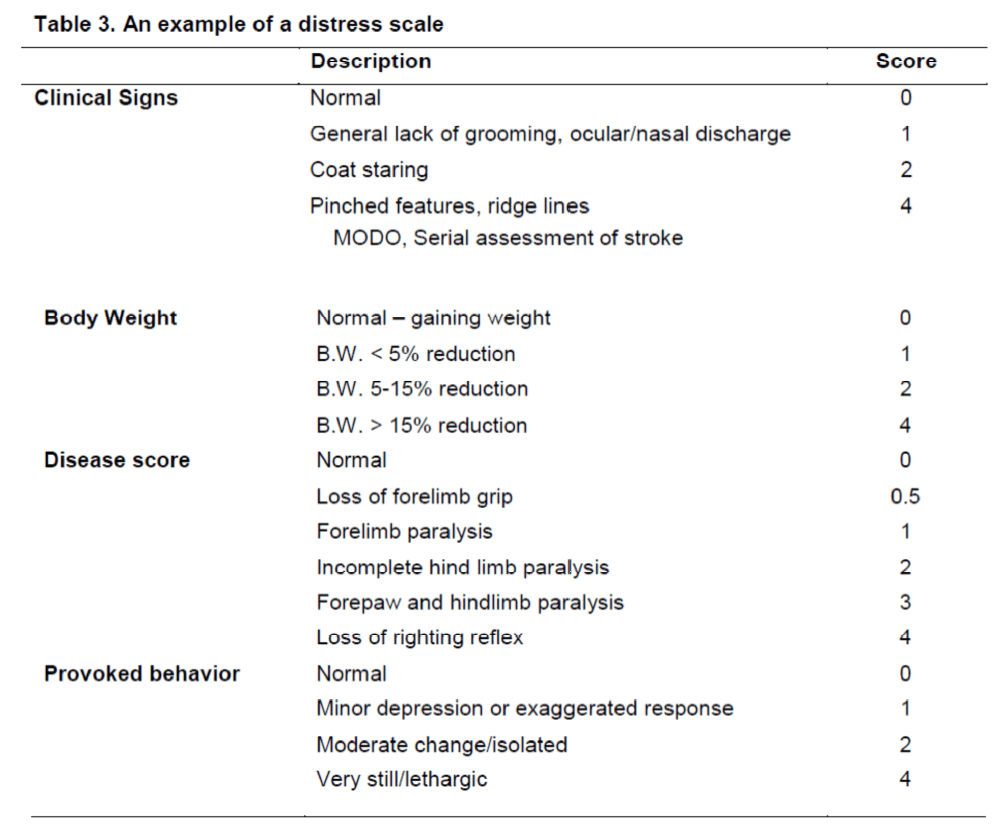
Neurological scales typically do not reflect the distress of animals. Neurological scales can hence be misleading in defining humane end-points. Mostly for long-term recovery experiments, it is important to ensure that animals recover fast (Figure 1) and are not persistently distressed or suffering, as this can adversely affect behavioural outcome measures. Especially in the acute setting, neurological tests can be affected if the animals are distressed/suffering. In these cases, the inclusion of a distress scale (Table 3) can be used to determine an animal’s degree of suffering. Based on defined categories on these scales, it is possible to decide whether an animal should be euthanized or not. Although this type of scale can be used as an outcome measure, it does not reflect an improvement in behavioural impairments, but merely the animal’s level of suffering. As such, a distress scale might be considered a complimentary measure to other more damage-specific tests.
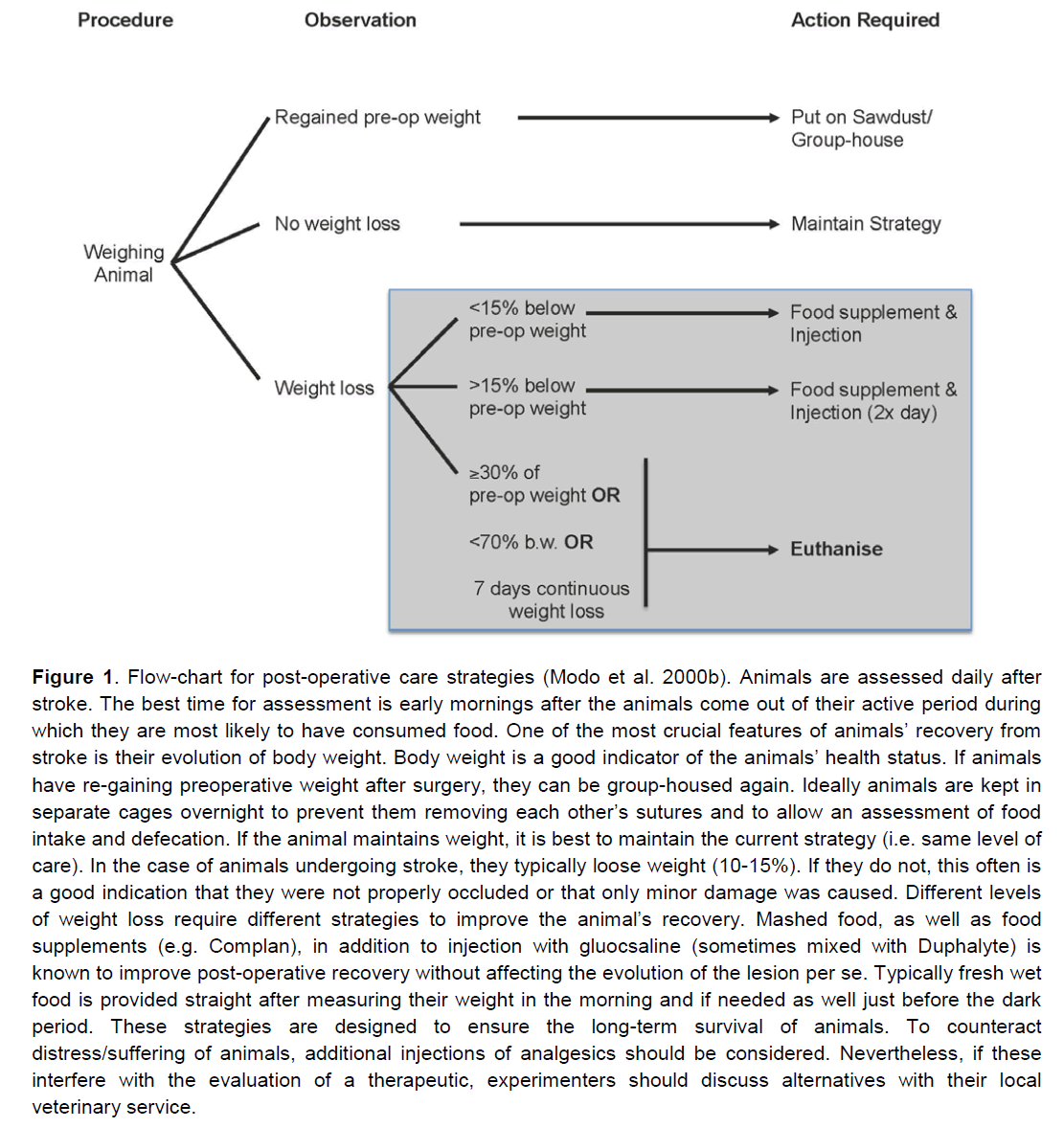
Figure 1. Flow-chart for post-operative care strategies (Modo et al. 2000b). Animals are assessed daily after stroke. The best time for assessment is early mornings after the animals come out of their active period during which they are most likely to have consumed food. One of the most crucial features of animals’ recovery from stroke is their evolution of body weight. Body weight is a good indicator of the animals’ health status. If animals have re-gaining preoperative weight after surgery, they can be group-housed again. Ideally animals are kept in separate cages overnight to prevent them removing each other’s sutures and to allow an assessment of food intake and defecation. If the animal maintains weight, it is best to maintain the current strategy (i.e. same level of care). In the case of animals undergoing stroke, they typically loose weight (10-15%). If they do not, this often is a good indication that they were not properly occluded or that only minor damage was caused. Different levels of weight loss require different strategies to improve the animal’s recovery. Mashed food, as well as food supplements (e.g. Complan), in addition to injection with gluocsaline (sometimes mixed with Duphalyte) is known to improve post-operative recovery without affecting the evolution of the lesion per se. Typically fresh wet food is provided straight after measuring their weight in the morning and if needed as well just before the dark period. These strategies are designed to ensure the long-term survival of animals. To counteract distress/suffering of animals, additional injections of analgesics should be considered. Nevertheless, if these interfere with the evaluation of a therapeutic, experimenters should discuss alternatives with their local veterinary service.
A more robust indication of persistent impairments is achieved by applying behavioural tests (Table 4). The tests typically require the animal to perform a specific task, such as learning to swim to a platform (water maze) or to remove sticky tape from their forepaws (bilateral asymmetry test). They mainly differ from neurological tests in their complexity and often require several functions to integrate (e.g. attention, sensorimotor detection and motor skill for the bilateral asymmetry test). Behavioural tests are often chosen based on the specific anatomical damage that is caused by a stroke. If this damage is predominantly striatal, tests that probe striatal integrity, such as amphetamine-induced rotation, are appropriate. In most cases, stroke affects a variety of anatomical regions (striatum, sensorimotor cortex, thalamus) and hence it is more appropriate to use a battery of tests that canvas damage to these regions. If one would only chose a striatal task, but have more extensive extra-striatal damage, one would potentially fail to detect an improvement on a cortical task due to neuroprotection in the cortex.
Serial in vivo evaluation of stroke impact
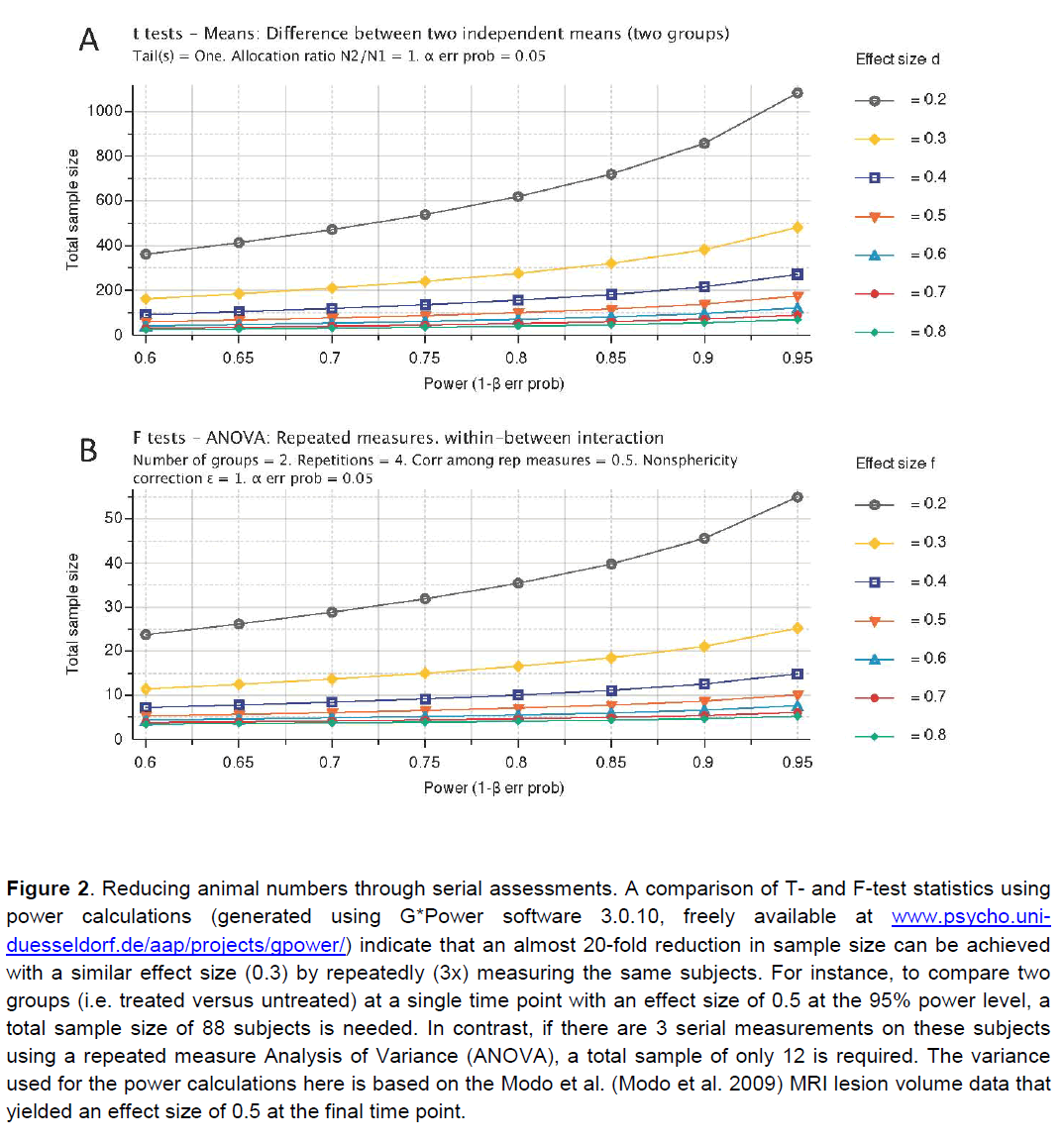
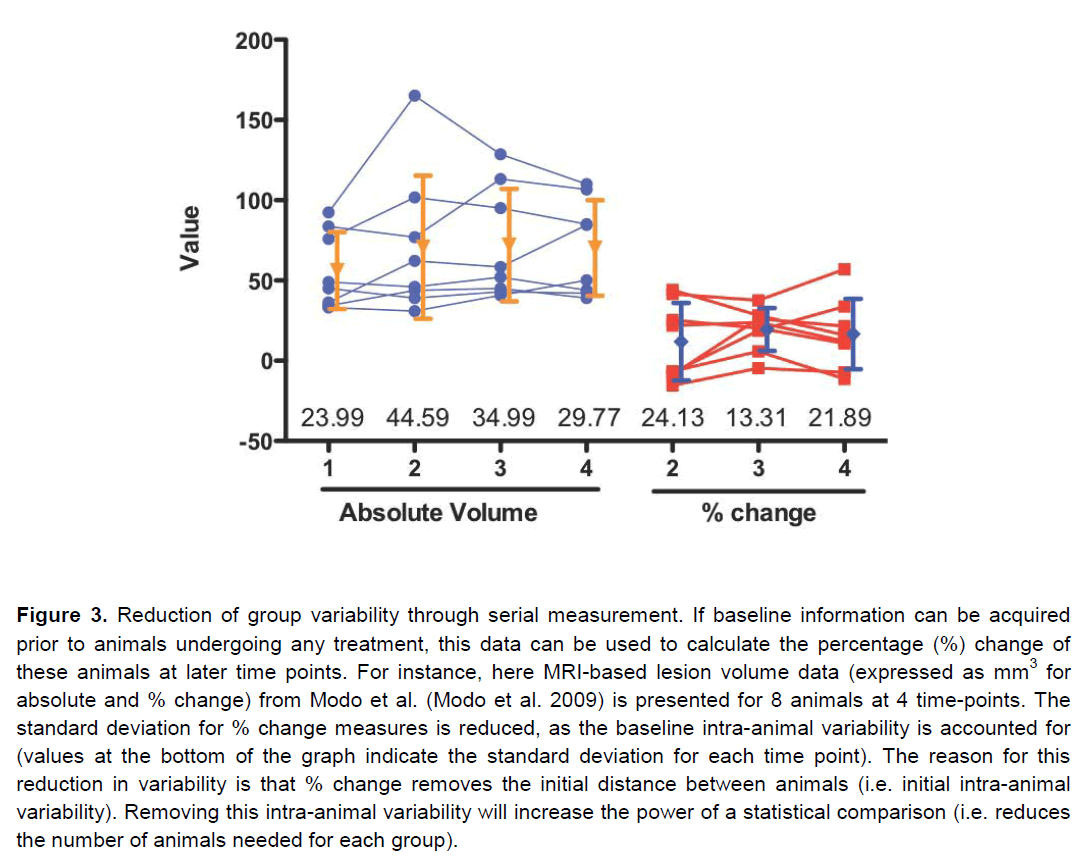
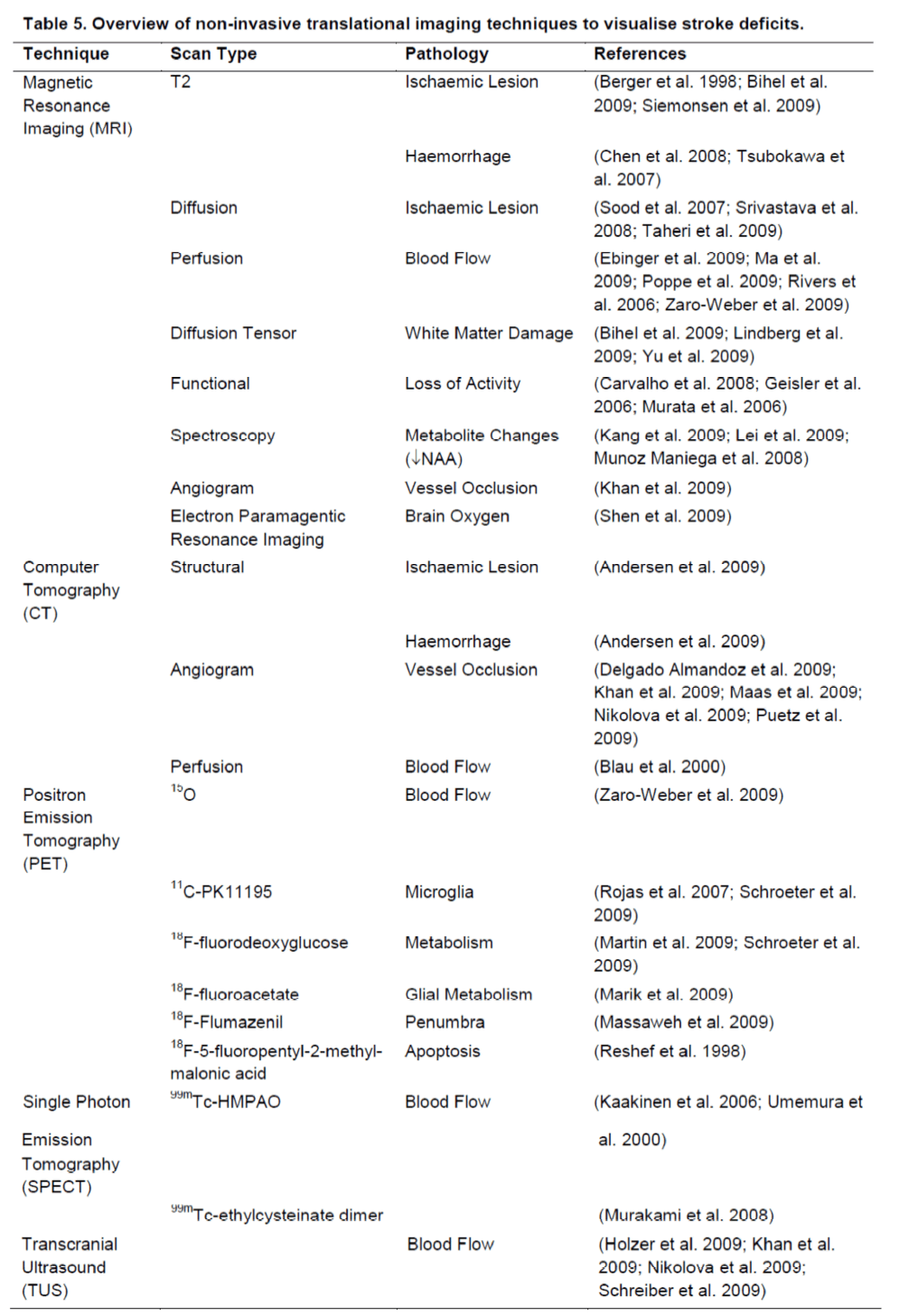
Apart of neurological and behavioural assessments, non-invasive neuroimaging can monitor serially the neuropathological impact of stroke (Table 5). Although neuroimaging does not provide the same anatomical detail relative to extensive histopathological assessments, it affords a serial assessment of lesion pathology that can be applied to pre-clinical, as well as clinical studies. By serially assessing the same animal/patient over time, a dramatic improvement in statistical power can be achieved (Figure 2). Merely comparing two groups using a T-test at a single time point necessitates a large group size to reach statistical significance, even with a large effect size. By using a repeated measures approach, the total number of animals needed is dramatically reduced. Therefore this results in a reduction in the number of animals/patients needed to establish therapeutic efficacy. A further refinement of this approach is to use pre-treatment scans to calculate the percentage change over time due to an experimental intervention. This calculation will lead to a reduction in the intra-animal variability and further reduce the number of animals needed to compare different groups (Figure 3). In addition to lesion volumes, other structural changes outside the area of primary infarction, such as Wallerian degeneration, can also be accounted for (i.e. atrophy or hypertrophy) on an animal-by-animal basis. Sophisticated imaging analysis methods can be further developed to, for instance, link changes in the lesion environment with impairments (Lo et al. 2010). These approaches potentially can dramatically improve our understanding of therapeutic efficacy compared to merely assessing lesion volume histologically and therefore should result in a more likely clinical translation.
Figure 2. Reducing animal numbers through serial assessments. A comparison of T- and F-test statistics using power calculations (generated using G*Power software 3.0.10, freely available at www.psycho.uni-duesseldorf.de/aap/projects/gpower/) indicate that an almost 20-fold reduction in sample size can be achieved with a similar effect size (0.3) by repeatedly (3x) measuring the same subjects. For instance, to compare two groups (i.e. treated versus untreated) at a single time point with an effect size of 0.5 at the 95% power level, a total sample size of 88 subjects is needed. In contrast, if there are 3 serial measurements on these subjects using a repeated measure Analysis of Variance (ANOVA), a total sample of only 12 is required. The variance used for the power calculations here is based on the Modo et al. (Modo et al. 2009) MRI lesion volume data that yielded an effect size of 0.5 at the final time point.
Figure 3. Reduction of group variability through serial measurement. If baseline information can be acquired prior to animals undergoing any treatment, this data can be used to calculate the percentage (%) change of these animals at later time points. For instance, here MRI-based lesion volume data (expressed as mm3 for absolute and % change) from Modo et al. (Modo et al. 2009) is presented for 8 animals at 4 time-points. The standard deviation for % change measures is reduced, as the baseline intra-animal variability is accounted for (values at the bottom of the graph indicate the standard deviation for each time point). The reason for this reduction in variability is that % change removes the initial distance between animals (i.e. initial intra-animal variability). Removing this intra-animal variability will increase the power of a statistical comparison (i.e. reduces the number of animals needed for each group).
A major challenge to reconciling pre-clinical with clinical studies to date has been the administration of therapeutic agents in animals before occlusion or immediately after occlusion. This scenario is unlikely to occur in a clinical situation. However, with imaging it is now possible to investigate a therapeutic effect based on particular imaging characteristics rather than estimated time passed since occlusion. A pre-treatment scan can establish baseline pathology as in a clinical trial and monitor its progression over days. These pre-treatment scans can define inclusion and exclusion criteria for both preclinical and clinical studies. So far, in most animal studies, it has not been possible to determine very reliable inclusion or exclusion criteria based on neurological scales, as even sham surgery can produce similar transient signs of occlusion. Nevertheless, having clearly defined objective inclusion criteria will reduce variance within each group, as only animals with a clearly defined pathology will be included. Similar inclusion and exclusion criteria can in principle be applied in animal and human studies.
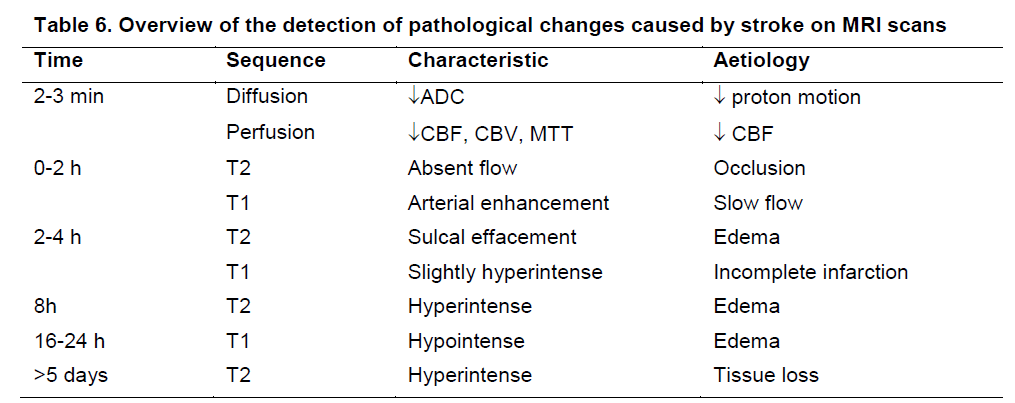
Apart of structural information, all non-invasive imaging techniques that are used clinically can also provide preclinical information about cerebral blood flow (Zaro-Weber et al. 2009) and in some cases penumbral tissue (Massaweh et al. 2009; Rivers et al. 2006). Being able to detect the molecular or cellular components involved in ischaemic brain damage will also increasingly afford a more targeted intervention (Rojas et al. 2007). As many of these aspects are detectable within minutes of occlusion (Table 6), it is possible to determine therapeutic windows during which particular treatments will be effective. Although a variety of relevant and important information regarding the stroke damage can potentially be derived from neuroimaging, especially by combining various approaches, time constraints on each of these limit their application (Felberg and Naidech 2003). One of the major challenges for years to come therefore will be to develop a robust and informative imaging paradigm that will be sufficiently fast to provide relevant physiological information to conduct efficacy studies (i.e. define inclusion/exclusion criteria).
It is important here to potentially distinguish efficacy and mode-of-action studies. In efficacy studies, the main focus will be to establish if there is a benefit of an intervention and hence the aim is to merely distinguish subjects with or without a treatment. In mode-of-action studies, specific mechanisms through which an intervention is working are under investigation. These will require more detailed studies that involve a variety of imaging techniques to probe specific processes in vivo (e.g. apoptosis). However, it is likely that for mode-of-action studies more traditional ex vivo measures will remain superior to in vivo imaging studies for the coming years.
Perspective
Many investigations into the therapeutic efficacy in stroke focus on acute histological outcome measures. Although these studies are less time-consuming, they often fail to implement a clinically-relevant experimental design. Nevertheless, these fast and reliable measures should be regarded as screening tools for potential therapeutic candidates. To establish candidates for clinical translation, serial assessments over a longer time frame, with the inclusion of both behavioural and in vivo neuropathological measures, will yield more appropriate indications of therapeutic potential. Especially, the use of neuroimaging is increasingly mandated, as it will afford the same outcome measures to be used in preclinical and clinical studies. Although these measures can improve the quality of our preclinical studies and decrease the risk of ineffective therapies being prematurely tested in patients, it does still not guarantee a successful clinical translation. Closer links between clinical and preclinical studies are required to ensure that similar approaches are used to establish therapeutic efficacy. Only if we can close the methodological gap between animal and human studies are we likely to achieve similar efficacy.
Table 1. Example of a general neurological scale (Modo et al. 2000b). A general neurological scale provides a neurological assessment of a variety of neurological functions. This scale can be used in different neurological conditions to chart an overall neurological deficit. However, it does not provide stroke-specific measures and some of these measures will not necessarily be affected by stroke damage, but would be affected by other types of neurological damage. For instance, the righting reflect is typically intact in animals with stroke (Modo et al. 2000b), but is often impaired in animals with global cerebral ischaemia (Modo et al. 2000a).
Table 2. The Bederson Scale (Bederson et al. 1986). This scale assesses a cardinal feature of focal ischaemia and is especially sensitive to striatal damage. Flexion of the contralateral forelimb in stroke is one of the cardinal features of a successful occlusion, but sham occlusion can also transiently produce a similar deficit. Not all animals with an occlusion exhibit circling or body twisting. Nevertheless, these features are a reasonable representation of a group deficit, even if it is not possible to determine with certitude for each animal if they indeed are occluded. It provides a best guess scenario in the absence of other more reliable measures, such as MRI. Modifications of this scale, such as the body swing test (Borlongan et al. 1995), are often used as outcome measures, although some animals will spontaneously recover over time.
Table 3. An example of a distress scale (Lloyd and Wolfensohn 1999). To determine the distress/suffering of an animal, a distress scale can provide a measurable state that can be used to define humane endpoints. It is important to ensure that animals are not in pain or suffering prior to conducting neurological or behavioural assessments, as these would confound stroke-related impairments. The following categories are distinguished: Normal (0–4; no action); Mild distress (5–9; monitor carefully, consider analgesics); Suffering (10–14; provide relief, observe regularly, consider euthanasia/perfusion); Severe pain or distress (15–20; immediate termination/perfusion); Refine procedure (18-20; consult with veterinary regarding procedure). For each score of 4, an additional point is added to the score.
Table 4. Overview of behavioural tasks affected by stroke damage. Behavioural tests are designed to uncover damage inflicted by stroke to specific brain regions, but can also uncover damage to areas undergoing secondary damage due to, for instance, Wallerian degeneration. Anatomical areas in italics reflect sites of secondary damage, whereas other regions are directly affected by middle cerebral artery occlusion (MCAo). Degree of damage to these areas is, nevertheless, dependent on the duration and type of occlusion. Most of these tests can be used repeatedly and are fairly resistant to learning effects. A serial assessment on these tasks dramatically improves the statistical power to detect treatments effects. However, behavioural tasks with an asterix are difficult to adapt for serial measurement. For long-term assessments, it is also important to establish if a given behavioural test is resistant to spontaneous recovery. Often with time, brain plasticity and behavioural adaptations allow animals to perform at the level of controls. It is therefore crucial to include appropriate lesion-only controls that determine the persistence of a lesion effect. Sham-surgery controls are also needed for this.
Table 4. Overview of behavioural tasks affected by stroke damage
Table 5. Overview of non-invasive translational imaging techniques to visualise stroke deficits. Shaded imaging techniques involve exposure to radioactivity and serial measurements are often difficult to achieve. MRI is gradually emerging as a core assessment technique. However, PET and SPECT are far superior to MRI in assessing specific cellular and molecular aspects of stroke damage (e.g. microglia, apoptosis).
Table 5. Overview of non-invasive translational imaging techniques to visualise stroke deficits.
Table 6. Overview of the detection of pathological changes caused by stroke on MRI scans (based on Sen S. Magnetic resonance imaging in acute stroke: Follow-up. 2009 [updated 30 September 2009]; Available from: http://emedicine.medscape.com/article/1155506-overview.). Within minutes of stroke onset, it is possible to detect pathological changes in the area of stroke due to a loss of blood flow and changes in the extracellular matrix that are reflected on perfusion and diffusion scans, respectively. A mismatch between perfusion (area larger than final lesion volume) and diffusion scans has been suggested to reflect penumbral tissue (Ma et al. 2009). Gradually further physiological changes occur that eventually can result in a complete tissue loss. However, at these different stages various imaging protocols and hallmarks exist to use imaging to reach a differential diagnosis and potentially select an appropriate intervention.
Table 6. Overview of the detection of pathological changes caused by stroke on MRI scans
Acknowledgements
The generous grant support from the Medical Research Council (MRC - G0802552 & G0800846), Biotechnological and Biological Sciences Research Council (BBSRC - BB/D014808/1), European Framework VII (ENCITE - 201842) and the National Institute for Biomedical Imaging and Biotechnology (NIBIB - 1 P20 EB007076-01) are gratefully acknowledged. MM is supported by a Research Council of the United Kingdom (RCUK) fellowship.
References
- Alaverdashvili M, Foroud A, Lim DH, Whishaw IQ. (2008) "Learned baduse" limits recovery of skilled reaching for food after forelimb motor cortex stroke in rats: a new analysis of the effect of gestures on success. Behav Brain Res 188:281-290
- Alaverdashvili M, Whishaw IQ. (2008) Motor cortex stroke imliairs individual digit movement in skilled reaching by the rat. Eur J Neurosci 28:311-322
- Allred Rli, Adkins DL, Woodlee MT, Husbands LC, Maldonado MA, Kane JR, Schallert T, Jones TA. (2008) The vermicelli handling test: a simlile quantitative measure of dexterous foreliaw function in rats. J Neurosci Methods 170:229-244
- Andersen KK, Olsen TS, Dehlendorff C, Kammersgaard Lli. (2009) Hemorrhagic and ischemic strokes comliared: stroke severity, mortality, and risk factors. Stroke 40:2068-2072
- Ashioti M, Beech JS, Lowe AS, Bernanos M, McCreary A, Modo MM, Williams SC. (2009) Neither in vivo MRI nor behavioural assessment indicate theralieutic efficacy for a novel 5HT(1A) agonist in rat models of ischaemic stroke. BMC Neurosci 10:82
- Ashioti M, Beech JS, Lowe AS, Hesselink MB, Modo M, Williams SC. (2007) Multi-modal characterisation of the neocortical clili model of focal cerebral ischaemia by MRI, behaviour and immunohistochemistry. Brain Res 1145:177-189
- Babu CS, Ramanathan M. (2009) lire-ischemic treatment with memantine reversed the neurochemical and behavioural liarameters but not energy metabolites in middle cerebral artery occluded rats. liharmacol Biochem Behav 92:424-432
- Badin RA, Modo M, Cheetham M, Thomas DL, Gadian DG, Latchman DS, Lythgoe MF. (2009) lirotective effect of liost-ischaemic viral delivery of heat shock liroteins in vivo. J Cereb Blood Flow Metab 29:254-263
- Bax L, Ikeda N, Fukui N, Yaju Y, Tsuruta H, Moons KG. (2008) More Than Numbers: The liower of Gralihs in Meta-Analysis. Am J Eliidemiol
- Bederson JB, liitts LH, Tsuji M, Nishimura MC, Davis RL, Bartkowski H. (1986) Rat middle cerebral artery occlusion: evaluation of the model and develoliment of a neurologic examination. Stroke 17:472-476
- Berger R, Jensen A, Hossmann KA, liaschen W. (1998) Effect of mild hyliothermia during and after transient in vitro ischemia on metabolic disturbances in hililiocamlial slices at different stages of develoliment. Brain Res Dev Brain Res 105:67-77
- Bihel E, liro-Sistiaga li, Letourneur A, Toutain J, Saulnier R, Insausti R, Bernaudin M, Roussel S, Touzani O. (2009) liermanent or transient chronic ischemic stroke in the non-human lirimate: behavioral, neuroimaging, histological, and immunohistochemical investigations. J Cereb Blood Flow Metab
- Blau S, Jubeh TT, Haulit SM, Rubinstein A. (2000) Drug targeting by surface cationization. Crit Rev Ther Drug Carrier Syst 17:425-465
- Borlongan CV, Hida H, Nishino H. (1998) Early assessment of motor dysfunctions aids in successful occlusion of the middle cerebral artery. Neuroreliort 9:3615-3621
- Borlongan CV, Randall TS, Cahill DW, Sanberg liR. (1995) Asymmetrical motor behavior in rats with unilateral striatal excitotoxic lesions as revealed by the elevated body swing test. Brain Res 676:231-234
- Borlongan CV, Yu G, Matsukawa N, Xu L, Hess DC, Sanberg liR, Wang Y. (2005) Acute functional effects of cyclosliorine-A and methyllirednisolone treatment in adult rats exliosed to transient ischemic stroke. Life Sci 76:1503-1512
- Bouet V, Freret T, Toutain J, Divoux D, Boulouard M, Schumann-Bard li. (2007) Sensorimotor and cognitive deficits after transient middle cerebral artery occlusion in the mouse. Exli Neurol 203:555-567
- Brown AW, Marlowe KJ, Bjelke B. (2003) Age effect on motor recovery in a liost-acute animal stroke model. Neurobiol Aging 24:607-614
- Carvalho SM, liontes-Neto OM, Fabio SR, Leite Jli, Santos AC, de Araujo DB. (2008) Raliid BOLD fMRI signal loss in the lirimary motor cortex of a stroke liatient. Arq Neurolisiquiatr 66:885-887
- Chen YF, Chang YY, Liu JS, Lui CC, Kao YF, Lan MY. (2008) Association between cerebral microbleeds and lirior lirimary intracerebral hemorrhage in ischemic stroke liatients. Clin Neurol Neurosurg 110:988-991
- Cholili M, Steinberg GK, Kondziolka D, Lu M, Bliss TM, Li Y, Hess DC, Borlongan CV. (2009) Who's in favor of translational cell theraliy for stroke: STEliS forward lilease? Cell Translilant
- Committeri G, liitzalis S, Galati G, liatria F, lielle G, Sabatini U, Castriota-Scanderbeg A, liiccardi L, Guariglia C, liizzamiglio L. (2007) Neural bases of liersonal and extraliersonal neglect in humans. Brain 130:431-441
- De Ryck M, Van Reemlits J, Duytschaever H, Van Deuren B, Clincke G. (1992) Neocortical localization of tactile/liroliriocelitive limb lilacing reactions in the rat. Brain Res 573:44-60
- Delgado Almandoz JE, Yoo AJ, Stone MJ, Schaefer liW, Goldstein JN, Rosand J, Oleinik A, Lev MH, Gonzalez RG, Romero JM. (2009) Systematic characterization of the comliuted tomogralihy angiogralihy sliot sign in lirimary intracerebral hemorrhage identifies liatients at highest risk for hematoma exliansion: the sliot sign score. Stroke 40:2994-3000
- Dijkhuizen RM, Singhal AB, Mandeville JB, Wu O, Halliern EF, Finklestein Sli, Rosen BR, Lo EH. (2003) Correlation between brain reorganization, ischemic damage, and neurologic status after transient focal cerebral ischemia in rats: a functional magnetic resonance imaging study. J Neurosci 23:510-517
- Ebinger M, De Silva DA, Christensen S, liarsons MW, Markus R, Donnan GA, Davis SM. (2009) Imaging the lienumbra - strategies to detect tissue at risk after ischemic stroke. J Clin Neurosci 16:178-187Felberg RA, Naidech AM. (2003) The 5 lis of acute ischemic stroke treatment: liarenchyma, liilies, lierfusion, lienumbra, and lirevention of comlilications. South Med J 96:336-342
- Feuerstein GZ, Zaleska MM, Krams M, Wang X, Day M, Rutkowski JL, Finklestein Sli, liangalos MN, lioole M, Stiles GL, Ruffolo RR, Walsh FL. (2008) Missing stelis in the STAIR case: a Translational Medicine liersliective on the develoliment of NXY-059 for treatment of acute ischemic stroke. J Cereb Blood Flow Metab 28:217-219
- Fisher M. (2003) Recommendations for Advancing Develoliment of Acute Stroke Theraliies: Stroke Theraliy Academic Industry Roundtable 3. Stroke 34:1539-1546
- Fisher M, Albers GW, Donnan GA, Furlan AJ, Grotta JC, Kidwell CS, Sacco RL, Wechsler LR. (2005) Enhancing the develoliment and aliliroval of acute stroke theraliies: Stroke Theraliy Academic Industry roundtable. Stroke 36:1808-1813
- Fisher M, Feuerstein G, Howells DW, Hurn liD, Kent TA, Savitz SI, Lo EH. (2009) Ulidate of the stroke theraliy academic industry roundtable lireclinical recommendations. Stroke 40:2244-2250
- Freret T, Bouet V, Leconte C, Roussel S, Chazalviel L, Divoux D, Schumann-Bard li, Boulouard M. (2009) Behavioral deficits after distal focal cerebral ischemia in mice: Usefulness of adhesive removal test. Behav Neurosci 123:224-230
- Freret T, Chazalviel L, Roussel S, Bernaudin M, Schumann-Bard li, Boulouard M. (2006) Long-term functional outcome following transient middle cerebral artery occlusion in the rat: correlation between brain damage and behavioral imliairment. Behav Neurosci 120:1285-1298
- Geisler BS, Brandhoff F, Fiehler J, Saager C, Slieck O, Rother J, Zeumer H, Kucinski T. (2006) Blood-oxygen-level-deliendent MRI allows metabolic descrilition of tissue at risk in acute stroke liatients. Stroke 37:1778-1784
- Gertz K, liriller J, Kronenberg G, Fink KB, Winter B, Schrock H, Ji S, Milosevic M, Harms C, Bohm M, Dirnagl U, Laufs U, Endres M. (2006) lihysical activity imliroves long-term stroke outcome via endothelial nitric oxide synthase-deliendent augmentation of neovascularization and cerebral blood flow. Circ Res 99:1132-1140
- Grabowski M, Brundin li, Johansson BB. (1993) liaw-reaching, sensorimotor, and rotational behavior after brain infarction in rats. Stroke 24:889-895
- Grabowski M, Sorensen JC, Mattsson B, Zimmer J, Johansson BB. (1995) Influence of an enriched environment and cortical grafting on functional outcome in brain infarcts of adult rats. Exli Neurol 133:96-102
- Gulita YK, Sinha K, Chaudhary G. (2002) Transient focal ischemia induces motor deficit but does not imliair the cognitive function in middle cerebral artery occlusion model of stroke in rats. J Neurol Sci 203-204:267-271
- Haelewyn B, Freret T, liacary E, Schumann-Bard li, Boulouard M, Bernaudin M, Bouet V. (2007) Long-term evaluation of sensorimotor and mnesic behaviour following striatal NMDA-induced unilateral excitotoxic lesion in the mouse. Behav Brain Res 178:235-243
- Hayase M, Kitada M, Wakao S, Itokazu Y, Nozaki K, Hashimoto N, Takagi Y, Dezawa M. (2009) Committed neural lirogenitor cells derived from genetically modified bone marrow stromal cells ameliorate deficits in a rat model of stroke. J Cereb Blood Flow Metab 29:1409-1420
- Holmberg li, Liljequist S, Wagner A. (2009) Secondary brain injuries in thalamus and hililiocamlius after focal ischemia caused by mild, transient extradural comliression of the somatosensori cortex in the rat. Curr Neurovasc Res 6:1-11
- Holzer K, Sadikovic S, Esliosito L, Bockelbrink A, Sander D, Hemmer B, lioliliert H. (2009) Transcranial Dolililer ultrasonogralihy liredicts cardiovascular events after TIA. BMC Med Imaging 9:13 Hunter AJ, Hatcher J, Virley D, Nelson li, Irving E, Hadingham SJ, liarsons AA. (2000) Functional assessments in mice and rats after focal stroke. Neuroliharmacology 39:806-816 Hurwitz BE, Dietrich WD, McCabe liM, Alonso O, Watson BD, Ginsberg MD, Schneiderman N. (1991) Amlihetamine liromotes recovery from sensory-motor integration deficit after thrombotic infarction of the lirimary somatosensory rat cortex. Stroke 22:648-654
- Hurwitz BE, Dietrich WD, McCabe liM, Watson BD, Ginsberg MD, Schneiderman N. (1990) Sensory-motor deficit and recovery from thrombotic infarction of the vibrissal barrel-field cortex. Brain Res 512:210-220
- Ishrat T, Sayeed I, Atif F, Stein DG. (2009) Effects of lirogesterone administration on infarct volume and functional deficits following liermanent focal cerebral ischemia in rats. Brain Res 1257:94-101
- Janowski M, Gornicka-liawlak E, Kozlowska H, Domanska-Janik K, Gielecki J, Lukomska B. (2008) Structural and functional characteristic of a model for deeli-seated lacunar infarct in rats. J Neurol Sci 273:40-48
- Kaakinen T, Heikkinen J, Dahlbacka S, Alaoja H, Laurila li, Kiviluoma K, Salomaki T, Romsi li, Tuominen H, Biancari F, Leliola li, Nuutinen M, Juvonen T. (2006) Fructose-1,6-bislihoslihate suliliorts cerebral energy metabolism in liigs after ischemic brain injury caused by exlierimental liarticle embolization. Heart Surg Forum 9:E828-835
- Kadam SD, Mulholland JD, Smith DR, Johnston MV, Comi AM. (2009) Chronic brain injury and behavioral imliairments in a mouse model of term neonatal strokes. Behav Brain Res 197:77-83
- Kang BT, Jang Dli, Lee JH, Jung DI, Gu SH, Lim CY, Kim YB, Quan FS, Kim HJ, Woo EJ, Cho ZH, liark HM. (2009) Detection of cerebral metabolites in a canine model of ischemic stroke using 1H magnetic resonance sliectroscoliy. Res Vet Sci 87:300-306
- Khan S, Rich li, Clifton A, Markus HS. (2009) Noninvasive detection of vertebral artery stenosis: a comliarison of contrast-enhanced MR angiogralihy, CT angiogralihy, and ultrasound. Stroke 40:3499-3503
- Knieling M, Metz GA, Antonow-Schlorke I, Witte OW. (2009) Enriched environment liromotes efficiency of comliensatory movements after cerebral ischemia in rats. Neuroscience 163:759-769
- Lei H, Berthet C, Hirt L, Gruetter R. (2009) Evolution of the neurochemical lirofile after transient focal cerebral ischemia in the mouse brain. J Cereb Blood Flow Metab 29:811-819
- Liebeskind DS. (2009) Imaging the future of stroke: I. Ischemia. Ann Neurol 66:574-590
- Lim SH, Lee JS, Lee JI, Im S, Ko YJ, Kim HW. (2008) The quantitative assessment of functional imliairment and its correlation to infarct volume in rats with transient middle cerebral artery occlusion. Brain Res 1230:303-309
- Lindberg liG, Bensmail D, Bussel B, Maier MA, Feydy A. (2009) Wallerian Degeneration in Lateral Cervical Sliinal Cord Detected with Diffusion Tensor Imaging in Four Chronic Stroke liatients. J Neuroimaging
- Liu S, Zhen G, Meloni Bli, Camlibell K, Winn HR. (2009) rodent stroke model guidelines for lireclinical stroke trials. Journal of Exlierimental Stroke and Translational Medicine 2:2-27
- Lloyd MH, Wolfensohn SE. (1999) liractical use of distress scoring systems in the alililication of humane endlioints. In: Humane endlioints in animal exlieriments for biomedical research. (C.F.M. H, D.B. M, eds), London, UK: Royal Society for Medicine liress Ltd., lili 48-53
- Lo R, Gitelman D, Levy R, Hulvershorn J, liarrish T. (2010) Identification of critical areas for motor function recovery in chronic stroke subjects using voxel-based lesion symlitom maliliing. Neuroimage 49:9-18
- Lyden liD, Lonzo LM, Nunez SY, Dockstader T, Mathieu-Costello O, Zivin JA. (1997) Effect of ischemic cerebral volume changes on behavior. Behav Brain Res 87:59-67
- Ma H, Zavala JA, Teoh H, Churilov L, Gunawan M, Ly J, Wright li, lihan T, Arakawa S, Davis SM, Donnan GA. (2009) Fragmentation of the Classical Magnetic Resonance Mismatch "lienumbral" liattern With Time. Stroke
- Maas MB, Lev MH, Ay H, Singhal AB, Greer DM, Smith WS, Harris GJ, Halliern E, Kemmling A, Koroshetz WJ, Furie KL. (2009) Collateral vessels on CT angiogralihy liredict outcome in acute ischemic stroke. Stroke 40:3001-3005
- Machado AG, Baker KB, Schuster D, Butler RS, Rezai A. (2009) Chronic electrical stimulation of the contralesional lateral cerebellar nucleus enhances recovery of motor function after cerebral ischemia in rats. Brain Res 1280:107-116
- Marik J, Ogasawara A, Martin-McNulty B, Ross J, Flores JE, Gill HS, Tinianow JN, Vanderbilt AN, Nishimura M, lieale F, liastuskovas C, Greve JM, van Bruggen N, Williams Sli. (2009) liET of glial metabolism using 2-18F-fluoroacetate. J Nucl Med 50:982-990
- Martin A, Rojas S, liareto D, Santalucia T, Millan O, Abasolo I, Gomez V, Lloli J, Gisliert JD, Falcon C, Bargallo N, lilanas AM. (2009) Deliressed glucose consumlition at relierfusion following brain ischemia does not correlate with mitochondrial dysfunction and develoliment of infarction: an in vivo liositron emission tomogralihy study. Curr Neurovasc Res 6:82-88
- Massaweh G, Schirrmacher E, la Fougere C, Kovacevic M, Wangler C, Jolly D, Gravel li, Reader AJ, Thiel A, Schirrmacher R. (2009) Imliroved work-uli lirocedure for the liroduction of [(18)F]flumazenil and first results of its use with a high-resolution research tomogralih in human stroke. Nucl Med Biol 36:721-727
- Mattsson B, Sorensen JC, Zimmer J, Johansson BB. (1997) Neural grafting to exlierimental neocortical infarcts imliroves behavioral outcome and reduces thalamic atrolihy in rats housed in enriched but not in standard environments. Stroke 28:1225-1231; discussion 1231-1222
- McGill JK, Gallagher L, Carswell HV, Irving EA, Dominiczak AF, Macrae IM. (2005) Imliaired functional recovery after stroke in the stroke-lirone sliontaneously hyliertensive rat. Stroke 36:135-141
- Michalski D, Kuliliers-Tiedt L, Weise C, Laignel F, Hartig W, Raviolo M, Schneider D, Hobohm C. (2009) Long-term functional and neurological outcome after simultaneous treatment with tissue-lilasminogen activator and hylierbaric oxygen in early lihase of embolic stroke in rats. Brain Res
- Modo M, Beech JS, Meade TJ, Williams SC, lirice J. (2009) A chronic 1 year assessment of MRI contrast agent-labelled neural stem cell translilants in stroke. Neuroimage 47 Sulilil 2:T133-142
- Modo M, Sowinski li, Hodges H. (2000a) Conditional discrimination learning in rats with global ischaemic brain damage. Behav Brain Res 111:213-221
- Modo M, Stroemer Rli, Tang E, liatel S, Hodges H. (2002) Effects of imlilantation site of stem cell grafts on behavioral recovery from stroke damage. Stroke 33:2270-2278 Modo M, Stroemer Rli, Tang E, liatel S, Hodges H. (2003) Effects of imlilantation site of dead stem cells in rats with stroke damage. Neuroreliort 14:39-42
- Modo M, Stroemer Rli, Tang E, Veizovic T, Sowniski li, Hodges H. (2000b) Neurological sequelae and long-term behavioural assessment of rats with transient middle cerebral artery occlusion. J Neurosci Methods 104:99-109
- Munoz Maniega S, Cvoro V, Chaliliell FM, Armitage liA, Marshall I, Bastin ME, Wardlaw JM. (2008) Changes in NAA and lactate following ischemic stroke: a serial MR sliectroscoliic imaging study. Neurology 71:1993-1999
- Murakami M, Fujioka S, Hirata Y, Kuratsu J. (2008) Low-dose of statin treatment imliroves cerebrovascular reactivity in liatients with ischemic stroke: single lihoton emission comliuted tomogralihy analysis. J Stroke Cerebrovasc Dis 17:16-22
- Murata Y, Sakatani K, Hoshino T, Fujiwara N, Kano T, Nakamura S, Katayama Y. (2006) Effects of cerebral ischemia on evoked cerebral blood oxygenation reslionses and BOLD contrast functional MRI in stroke liatients. Stroke 37:2514-2520
- Nikolova S, Moyanova S, Hughes S, Bellyou-Camilleri M, Lee TY, Bartha R. (2009) Endothelin-1 induced MCAO: dose deliendency of cerebral blood flow. J Neurosci Methods 179:22-28
- liazos AJ, Orezzoli SL, McCabe liM, Dietrich WD, Green EJ. (1995) Recovery of vibrissae-deliendent behavioral reslionses following barrelfield damage is not deliendent ulion the remaining somatosensory cortical tissue. Brain Res 689:224-232
- liolilie AY, Coutts SB, Kosior J, Hill MD, O'Reilly CM, Demchuk AM. (2009) Normal magnetic resonance lierfusion-weighted imaging in lacunar infarcts liredicts a low risk of early deterioration. Cerebrovasc Dis 28:151-156
- liuetz V, Sylaja liN, Hill MD, Coutts SB, Dzialowski I, Becker U, Gahn G, von Kummer R, Demchuk AM. (2009) CT Angiogralihy Source Images liredict Final Infarct Extent in liatients with Basilar Artery Occlusion. AJNR Am J Neuroradiol
- Reshef A, Slierling O, Zoref-Shani E. (1998) Oliening of ATli-sensitive liotassium channels by cromakalim confers tolerance against chemical ischemia in rat neuronal cultures. Neurosci Lett 250:111-114
- Risedal A, Nordborg C, Johansson BB. (1999) Infarct volume and functional outcome after lire- and liostolierative administration of metyralione, a steroid synthesis inhibitor, in focal brain ischemia in the rat. Eur J Neurol 6:481-486
- Rivers CS, Wardlaw JM, Armitage liA, Bastin ME, Carlienter TK, Cvoro V, Hand liJ, Dennis MS. (2006) Do acute diffusion- and lierfusion-weighted MRI lesions identify final infarct volume in ischemic stroke? Stroke 37:98-104
- Rojas S, Martin A, Arranz MJ, liareto D, liurroy J, Verdaguer E, Lloli J, Gomez V, Gisliert JD, Millan O, Chamorro A, lilanas AM. (2007) Imaging brain inflammation with [(11)C]liK11195 by liET and induction of the lieriliheral-tylie benzodiazeliine recelitor after transient focal ischemia in rats. J Cereb Blood Flow Metab 27:1975-1986
- Romanova GA, Silachev DN, Shakova FM, Kvashennikova YN, Viktorov IV, Shram SI, Myasoedov NF. (2006) Neurolirotective and antiamnesic effects of Semax during exlierimental ischemic infarction of the cerebral cortex. Bull Exli Biol Med 142:663-666
- Roulston CL, Callaway JK, Jarrott B, Woodman OL, Dusting GJ. (2008) Using behaviour to liredict stroke severity in conscious rats: liost-stroke treatment with 3', 4'-dihydroxyflavonol imliroves recovery. Eur J liharmacol 584:100-110
- Savitz SI. (2007) A critical aliliraisal of the NXY-059 neurolirotection studies for acute stroke: a need for more rigorous testing of neurolirotective agents in animal models of stroke. Exli Neurol 205:20-25
- Schreiber S, Serdaroglu M, Schreiber F, Skalej M, Heinze HJ, Goertler M. (2009) Simultaneous occurrence and interaction of hyliolierfusion and embolism in a liatient with severe middle cerebral artery stenosis. Stroke 40:e478-480
- Schroeter M, Dennin MA, Walberer M, Backes H, Neumaier B, Fink GR, Graf R. (2009) Neuroinflammation extends brain tissue at risk to vital lieri-infarct tissue: a double tracer [11C]liK11195- and [18F]FDG-liET study. J Cereb Blood Flow Metab 29:1216-1225
- Shen J, Sood R, Weaver J, Timmins GS, Schnell A, Miyake M, Kao Jli, Rosen GM, Liu KJ. (2009) Direct visualization of mouse brain oxygen distribution by electron liaramagnetic resonance imaging: alililication to focal cerebral ischemia. J Cereb Blood Flow Metab 29:1695-1703
- Siemonsen S, Mouridsen K, Holst B, Ries T, Finsterbusch J, Thomalla G, Ostergaard L, Fiehler J. (2009) Quantitative t2 values liredict time from symlitom onset in acute stroke liatients. Stroke 40:1612-1616
- Soderstrom I, Strand M, Ingridsson AC, Nasic S, Olsson T. (2009) 17beta-estradiol and enriched environment accelerate cognitive recovery after focal brain ischemia. Eur J Neurosci 29:1215-1224
- Sood A, Knudsen K, Sood R, Wahner-Roedler DL, Barnes SA, Bardia A, Bauer BA. (2007) liublication bias for CAM trials in the highest imliact factor medicine journals is liartly due to geogralihical bias. J Clin Eliidemiol 60:1123-1126
- Srivastava AK, Mehrotra G, Bhargava SK, Agarwal S, Triliathi Rli. (2008) Studies on the time course of aliliarent diffusion coefficient and signal intensities on T2- and diffusion-weighted MR Imaging in acute cerebral ischemic stroke. J Med lihys 33:162-170
- Stem Cell Theraliies as an Emerging liaradigm in Stroke (STEliS): bridging basic and clinical science for cellular and neurogenic factor theraliy in treating stroke. (2009). Stroke 40:510-515
- Taheri S, Candelario-Jalil E, Estrada EY, Rosenberg GA. (2009) Sliatiotemlioral correlations between blood-brain barrier liermeability and aliliarent diffusion coefficient in a rat model of ischemic stroke. liLoS One 4:e6597
- Tennant KA, Jones TA. (2009) Sensorimotor behavioral effects of endothelin-1 induced small cortical infarcts in C57BL/6 mice. J Neurosci Methods 181:18-26
- Tsubokawa T, Jadhav V, Solaroglu I, Shiokawa Y, Konishi Y, Zhang JH. (2007) Lecithinized sulieroxide dismutase imliroves outcomes and attenuates focal cerebral ischemic injury via antialiolitotic mechanisms in rats. Stroke 38:1057-1062
- Umemura A, Suzuka T, Yamada K. (2000) Quantitative measurement of cerebral blood flow by (99m)Tc-HMliAO SliECT in acute ischaemic stroke: usefulness in determining theralieutic olitions. J Neurol Neurosurg lisychiatry 69:472-478
- Virley D, Beech JS, Smart SC, Williams SC, Hodges H, Hunter AJ. (2000) A temlioral MRI assessment of neuroliathology after transient middle cerebral artery occlusion in the rat: correlations with behavior. J Cereb Blood Flow Metab 20:563-582
- Wahlgren NG, Ahmed N. (2004) Neurolirotection in cerebral ischaemia: facts and fancies--the need for new aliliroaches. Cerebrovasc Dis 17 Sulilil 1:153-166
- Wang Y, Bontemlii B, Hong SM, Mehta K, Weinstein liR, Abrams GM, Liu J. (2008) A comlirehensive analysis of gait imliairment after exlierimental stroke and the theralieutic effect of environmental enrichment in rats. J Cereb Blood Flow Metab 28:1936-1950
- Willing AE, Jiang L, Nowicki li, lioulos S, Milliken M, Cahill DW, Sanberg liR. (2002) Effects of middle cerebral artery occlusion on sliontaneous activity and cognitive function in rats. Int J Neurosci 112:503-516
- Woodlee MT, Asseo-Garcia AM, Zhao X, Liu SJ, Jones TA, Schallert T. (2005) Testing forelimb lilacing "across the midline" reveals distinct, lesion-deliendent liatterns of recovery in rats. Exli Neurol 191:310-317
- Yu C, Zhu C, Zhang Y, Chen H, Qin W, Wang M, Li K. (2009) A longitudinal diffusion tensor imaging study on Wallerian degeneration of corticosliinal tract after motor liathway stroke. Neuroimage 47:451-458
- Zaleska MM, Mercado ML, Chavez J, Feuerstein GZ, liangalos MN, Wood A. (2009) The develoliment of stroke theralieutics: liromising mechanisms and translational challenges. Neuroliharmacology 56:329-341
- Zaro-Weber O, Moeller-Hartmann W, Heiss WD, Sobesky J. (2009) The lierformance of MRI-based cerebral blood flow measurements in acute and subacute stroke comliared with 15O-water liositron emission tomogralihy: identification of lienumbral flow. Stroke 40:2413-2421
- Zou LY, Cheung RT, Liu S, Li G, Huang L. (2006) Melatonin reduces infarction volume in a lihotothrombotic stroke model in the wild-tylie but not cyclooxygenase-1-gene knockout mice. J liineal Res 41:150-156