Review Article - Imaging in Medicine (2017) Volume 9, Issue 6
MRI in pre-operative NAC vascular map
Cabral Francisco*, Salvador João, Barroca Rita, Marques José Carlos, Leal de Faria João & Abecasis NunoInstituto Português Oncologia Lisboa, Francisco Gentil - Rua Professor Lima Basto, Lisboa, Portugal
- Corresponding Author:
- Cabral Francisco
Instituto Português Oncologia Lisboa
Francisco Gentil - Rua Professor Lima Basto
Lisboa, Portugal
E-mail: Francisco.cabrall@gmail.com
Abstract
Objective: NAC’s blood supply may convey essential information for surgery planning. This study’s aim was to evaluate breast blood supply using MRI and to compare it between oncological and non-oncological subjects. The risk factors for NAC necrosis in nipple sparing mastectomy were analyzed.
Methods: Breast MRI exams in one month at a single institution were evaluated. The considered inclusion criteria focused on patients with: ductal carcinoma in situ, invasive carcinoma (IC) or high-risk screening (HRS) without previous breast surgery. Subtraction reconstructions from dynamic acquisitions obtained at 60 s post-contrast administration were used.
Results: 166 breasts were evaluated (12 pre op DCIS; 42 HRS; 112 pre op IC) - 71% of the NAC’s had a dominant blood supply. Tumors were localized in the upper outer quadrant (UOQ) in 46% of cases. In the NAC vascularization analysis, there was a tendency for a higher number of vessels vascularizing the NAC in the IC group (p=0.056) and there was a difference in the quadrant analysis mainly at the UOQ that had a dominant blood supply in 18% of the IC group and only 2% in the HRS (p=0.048). 16 patients performed NSM, three had NAC necrosis. The number of vessels supplying the NAC was the only factor with correlation with this outcome (p=0.01). There was not a single NAC with a dominant blood vessel from the lower outer quadrant (LOQ).
Conclusion: Using MRI to pre-operatively evaluate breast blood supply is feasible without adding extra MRI time or contrast. 71% had dominant supply to the NAC, so every effort should be made to preserve it. The LOQ appears to be an optimal site for skin incisions. There is an asymmetry between cancer and HRS breasts mainly at the NAC’s level with neovascularization of that area. Patients with a single vessel supplying the NAC are at improved risk for necrosis.
Keywords
breast ▪ magnetic resonance imaging ▪ nipple-areola complex ▪ nipple sparing mastectomy ▪ oncoplastic ▪ necrosis
Abbreviations
NAC: nipple-areola complex; MRI: magnetic resonance imaging; DCIS: ductal carcinoma in situ; NSM: nipple sparing mastectomy; HRS: high risk screening; LOQ: lower outer quadrant; UOQ: upper outer quadrant; UIQ: upper inner quadrant; LIQ: lower inner quadrant
Introduction
Nipple sparing mastectomy (NSM) has become a common procedure, after its oncological safety was proved [1-5]. In spite of its advantages, such as the aesthetic outcome, it is still associated with an important morbidity [6].
Nipple-areola complex (NAC) necrosis is probably the most feared outcome when we choose this procedure. Its incidence varies from 0 to 48%, most series being around 10% [7-16].
Several studies try to correlate pre-operative risk factors (such as age, breast size, previous surgery, radiotherapy, type of incisions, flap thickness, smoking history, diabetes, degree of ptosis) with NAC necrosis [12,17,18]. These studies, although discrepant, have statistically shown a significant correlation between some of these variables and the incidence of NAC necrosis [12,17,18]. The vascular blood supply to the NAC itself is probably the main factor.
In parallel, regardless of the ongoing debate concerning MRI indications [19,20], it is being increasingly used to evaluate pre-operative breast cancer patients. It is estimated that 29 to 46% of all breast cancer patients are pre-operatively submitted to MRI [21,22].
The use of MRI images can have the potential benefit of delineating the breast vascular map. This was proven possible by Seitz, Iris et al. [23] in 2015, by assessing 52 benign breasts and comparing their anatomical results with previous cadaver studies [24-26]. To the best of our knowledge, there is no reference in the literature to MRI of assessing breast vascular map in cancer patients.
This study´s main purpose is to answer the following clinical question: in patients with breast cancer that are going to do a NSM, is it possible to use MRI to pre-operatively evaluate their NAC vascular map? Also, as a secondary objective, we were trying to evaluate if there is a difference in the vascular NAC supply between oncological and non-oncological patients.
Patients and methods
Consecutive Breast MRI exams obtained from a Phillips Ingenia 3.0T in a one month’s time frame at our institution were evaluated. All exams where retrospectively reviewed by two radiologists.
Inclusion criteria referred to patients with DCIS, IC or HRS. Patients with previous breast surgery history where excluded from our study because of the resulting alteration of the normal breast geometry and vascular blood supply.
Using subtraction reconstructions from dynamic acquisitions obtained at 60 s postcontrast administration, 3D MIP reconstructions were made to evaluate subjectively the breast’s vascular map of 83 patients. Axial T2 and T1 dynamic acquisitions were then used to discern the intercostal origin of the vascular branches depicted on the 3D MIP reconstructions.
Using the images obtained, the dominant blood vessel to the breast, NAC and correspondent quadrant were evaluated. Blood supply to the breast was further divided into blood supply to the breast itself and blood supply to the NAC. The quadrant was classified in upper outer quadrant (UOQ), upper inner quadrant (UIQ), lower outer quadrant (LOQ), lower inner quadrant (LIQ) and central quadrant (CQ).
Symmetry was considered when the exact same vessels responsible for the blood supply in both breasts were present. The subpopulation submitted to NSM was analyzed in terms of age, smoking history, body mass index (BMI), incision type, reconstruction approach, number of vessels supplying the NAC and surgical specimen weight.
Dichotomous outcomes were assessed with an exact Pearson’s χ2 test, with 95% CIs calculated for the risk difference. Linear regression model was performed to multivariate analysis. Missing data was deemed missing at random. A twosided “p” value of less than 0.05 was accepted to show significance. All analyses were done using IBM SPSS 22.0.
Results
All patients were female. A total of one hundred sixty six (166) breasts were evaluated (12 pre op DCIS; 42 screening; 112 pre op invasive carcinoma). Patient age ranged from 26 to 84 (median 64). 16 patients performed NSM.
Giving the inclusion criteria, the study had 3 groups of patients:
1) DCIS: with only 6 patients. The authors decided not to include this group in most of the statistical comparison analysis because of the reduced number of subjects.
2) High risk screening (HRS) (21 patients): patients followed at our institute with proven BRCA 1/2 mutations or high family risk.
3) Invasive carcinoma (IC) (56 patients): a total of 56 patients were diagnosed with invasive carcinoma through a biopsy. Tumors were localized in the upper outer quadrant (UOQ) in 46% (26/56) of cases. The average tumor size measured by MRI was 21 mm. 77% had a positive estrogen receptor status at core biopsy; 20% were triple negative; 18% were HER2 positive.
It was possible to evaluate the vascular map in all patients and it was divided in:
Breast blood supply
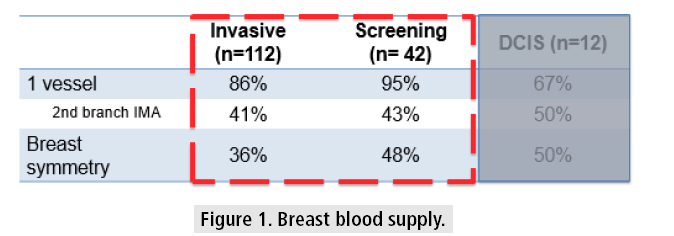
The breast analyzed had, in the majority of cases - 87% (144/166) -, a single dominant vessel responsible for its vascularization.
That vessel was, in most patients, the internal mammary artery (IMA) - 75% (125/ 166), mainly the 2nd intercostal branch 42% (70/166).
The symmetry analysis showed a correlation between both breasts of 36% in the IC group and of 48% in the HRS. Comparing the two groups, the only difference the authors noticed was the highest symmetry in the screening group, not reaching statistical significance (TABLE 1 and FIGURE 1).
| BREAST BLOOD SUPPLY | |||||
|---|---|---|---|---|---|
| Number of dominant vessels | Dominant vessel | Right breast | Left Breast | Total | |
| INVASIVE |
1 | IMA (1st) | 6 | 11 | 17 |
| IMA (2nd) | 24 | 22 | 46 | ||
| IMA (3rd) | 6 | 9 | 15 | ||
| IMA (4th) | 2 | 2 | 4 | ||
| IC (4th) | 5 | 2 | 7 | ||
| LT | 6 | 1 | 7 | ||
| >1 | 7 | 9 | 16 | ||
| (56) | (56) | (112) | |||
| SCREENING | 1 | IMA (1st) | 3 | 5 | 8 |
| IMA (2nd) | 10 | 8 | 18 | ||
| IMA (3rd) | 3 | 5 | 8 | ||
| IMA (4th) | 0 | 1 | 1 | ||
| LT | 3 | 2 | 5 | ||
| >1 | 2 | 0 | 2 | ||
| (21) | (21) | (42) | |||
| DCIS | 1 | IMA (1st) | 1 | 0 | 1 |
| IMA (2nd) | 3 | 3 | 6 | ||
| IMA (3rd) | 0 | 1 | 1 | ||
| >1 | 2 | 2 | 4 | ||
| (6) | (6) | (12) | |||
| Total breasts | (83) | (83) | 166 | ||
Table 1: Breast blood supply distribution.
NAC blood supply
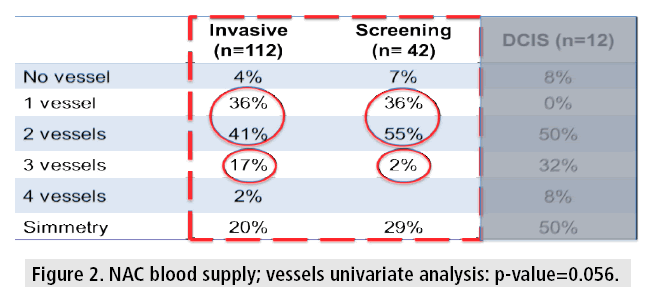
NAC vascularization was dominant in 117/166 (71%) (71% 80/112 CI vs 69% 29/42 HRS), mostly coming from the internal mammary artery 62% (106/166).
The NAC was supplied by 1 vessel in 33% (55/166) and 2 vessels in 45% (75/166). It is important to note that 17% (19/112) of the IC group had 3 vessels vascularizing the NAC and in the HRS group there was only 2% (1/42) (TABLE 2 and FIGURE 2).
| NAC BLOOD SUPPLY | |||||
|---|---|---|---|---|---|
| Number of dominant vessels | Dominant vessel | Right breast | Left Breast | Total | |
| INVASIVE | 0 | 2 | 2 | 4 | |
| 1 | IMA (1st) | 3 | 2 | 5 | |
| IMA (2nd) | 12 | 13 | 25 | ||
| IMA (3rd) | 4 | 3 | 7 | ||
| IMA (4th) | 0 | 2 | 2 | ||
| IC (4th) | 0 | 1 | 1 | ||
| 2 | No dominant | 10 | 8 | 18 | |
| IMA (1st) | 4 | 4 | 8 | ||
| IMA (2nd) | 9 | 4 | 13 | ||
| IMA (3rd) | 0 | 1 | 1 | ||
| IMA (4th) | 2 | 0 | 2 | ||
| LT | 2 | 2 | 4 | ||
| 3 | No dominant | 2 | 5 | 7 | |
| IMA (2nd) | 4 | 4 | 8 | ||
| IMA (3rd) | 0 | 2 | 2 | ||
| IC (4th) | 1 | 0 | 1 | ||
| LT | 0 | 1 | 1 | ||
| 4 | No dominant | 1 | 2 | 3 | |
| (56) | (56) | (112) | |||
| SCREENING | 0 | 1 | 2 | 3 | |
| 1 | IMA (1st) | 1 | 1 | 2 | |
| IMA (2nd) | 3 | 3 | 6 | ||
| IMA (3rd) | 2 | 2 | 4 | ||
| IMA (4th) | 0 | 1 | 1 | ||
| LT | 1 | 1 | 2 | ||
| 2 | No dominant | 7 | 3 | 10 | |
| IMA (1st) | 0 | 2 | 2 | ||
| IMA (2nd) | 4 | 3 | 7 | ||
| IMA (3rd) | 0 | 2 | 2 | ||
| LT | 1 | 1 | 2 | ||
| 3 | IMA (2nd) | 1 | 0 | 1 | |
| (21) | (21) | (42) | |||
| DCIS | 0 | 1 | 0 | 1 | |
| 1 | 0 | 0 | 0 | ||
| 2 | IMA (1st) | 1 | 0 | 1 | |
| IMA (2nd) | 2 | 2 | 4 | ||
| IMA (3rd) | 0 | 1 | 1 | ||
| 3 | No dominant | 1 | 1 | 2 | |
| IMA (2nd) | 1 | 1 | 2 | ||
| 4 | No dominant | 0 | 1 | 1 | |
| (6) | (6) | (12) | |||
| Total NAC’s | 166 | ||||
Table 2: NAC blood supply distribution.
Comparing both groups, the results show an apparent trend in terms of symmetry and number of vessels responsible for the NAC vascularization (highest in the IC group). Nevertheless, it did not reach statistical significance (3 vessel univariate analysis: p-value=0.056).
Quadrant analysis
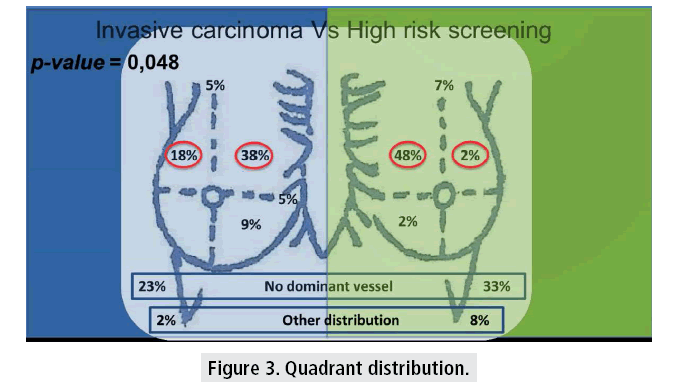
The quadrant analysis showed that 75% (125/166) had a dominant blood vessel. It also showed a predominance of the inner quadrants, mainly the UIQ (dominant vessel from this quadrant in 43% of all breasts - 71/166). However, the invasive carcinoma group showed a significant higher percentage of dominant blood supply from the UOQ (18% IC vs. 2% HRS).
In the univariate analysis there was a significant statistical difference between the IC and HRS groups (p=0.048). Not a single breast had dominant blood supply to the NAC from the LOQ (TABLE 3 and FIGURE 3).
| Quadrant | ||||
|---|---|---|---|---|
| Right breast | Left Breast | Total | ||
| INVASIVE | UIQ | 23 | 20 | 43 |
| UOQ | 8 | 12 | 20 | |
| LIQ | 5 | 5 | 10 | |
| UOQ/ UIQ | 3 | 2 | 5 | |
| UIQ/ LIQ | 3 | 3 | 6 | |
| UIQ + UOQ | 1 | 1 | 2 | |
| No dominance* | 13 | 13 | 26 | |
| (56) | (56) | (112) | ||
| SCREENING | UIQ | 10 | 10 | 20 |
| UOQ | 1 | 0 | 1 | |
| LIQ | 0 | 1 | 1 | |
| UOQ/UIQ | 1 | 2 | 3 | |
| UIQ+UOQ | 0 | 1 | 1 | |
| UOQ+LOQ | 1 | 1 | 2 | |
| No dominance* | 8 | 6 | 14 | |
| (21) | (21) | (42) | ||
| DCIS | UIQ | 4 | 4 | 8 |
| UIQ + UOQ | 1 | 1 | 2 | |
| UIQ/ LIQ | 0 | 1 | 1 | |
| No dominance* | 1 | 0 | 1 | |
| (6) | (6) | (12) | ||
| Total quadrants | 166 | |||
| * In these patients it was not possible to visualize a dominant vessel going all the way to the NAC | ||||
Table 3: Quadrant distribution.
It was possible to see a clear neovascularization in most tumor patients. This neovascularization was not objectively measured.
NSM and NAC necrosis
NSM was performed in 16 oncological patients (mean age 59 years, range 35 to 68 years). All patients underwent immediate breast reconstruction with latissimus dorsi flap with a periareolar incision. Mean BMI was 25 (22.5-27.5), mean specimen weight of 290 g (242-310), mean prosthesis weight of 270 ml (230-320). Nine patients had smoking history (56%). 6 patients had a single vessel supplying the NAC (38%) and 10 had two or three vessels responsible for NAC blood supply (62%).
NAC necrosis was found in three patients (19%), all of which had a single vessel supplying the NAC. In the univariate analysis the number of vessels supplying the NAC was the only factor with correlation with NAC necrosis (p=0.001). In the multivariate analysis none of the studied variables had a statistical significant correlation.
Discussion
NSM has gained popularity in the last few years, after proving oncological safety*, mostly because it is an opportunity to achieve excellent aesthetic outcome. However, this is sometimes ruined by nipple necrosis that varies from 0 to 48% depending on the series, most of them count for around 10%*.
In previous studies, we have noticed an effort to correlate NAC necrosis with different factors (age, BMI, breast size, smoking habits, diabetes, type of incision, breast ptosis, etc.)**. In fact, we even found some studies that attempted this correlation with statistical significance*, yet probably, as with any necrosis throughout the body, vascular blood supply is the single most important factor.
Preserving the blood supply remains essential to ensure that procedures are successful. The patterns of blood supply to the NAC are variable [24-26], so it seems to be of major importance that the surgeon knows the specific pattern of supply in the individual patient, in order to prepare the surgery and reduce the vascular complications.
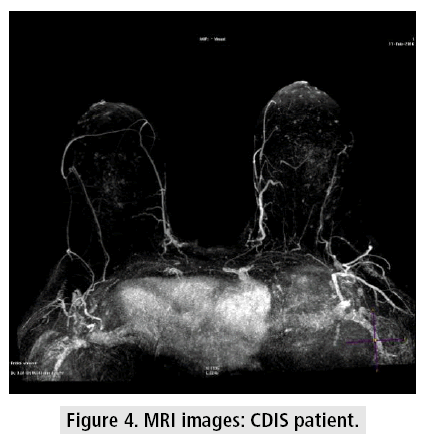
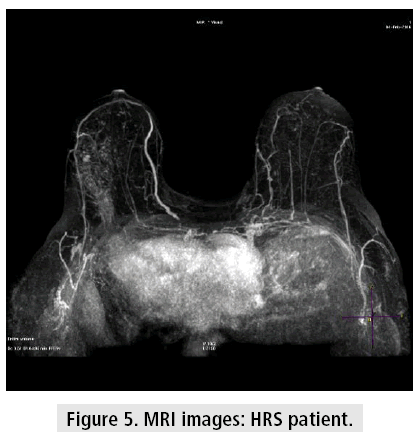
Despite the controversy about MRI indications in breast cancer [20,27], the data shows us that it has gained popularity amongst this population*. At our institution, we use it routinely pre operatively, so it is important that we extract from it the maximum information we can to help us treat the patients (FIGURES 4 and 5).
Our study showed that it is possible to visualize the NAC blood supply pre-operatively using MRI images without adding extra time or contrast to the exam. There is one study which demonstrated that it was possible to see the NAC blood supply using MRI images*, but the population from that study was not oncological.
As far as we know, this is the first study with an oncological population.
We were able to do a live study of NAC blood supply with perfect visualization, not only of the vessels, but also of the quadrants and their breast depth. All this information can and should be used by the surgeon to plan the surgery - type of incision, carefulness in the dissection of a certain quadrant or choice of pedicle in mammoplasties.
There are other methods for assessing NAC blood supply, but all of them are intra operative and require expertise from the surgeon (e.g. Doppler, indocyanine green fluorescence angiography [28,29]. We believe that using the information on the MRI can reduce the need for these methods, and possibly reduce NAC necrosis. Our results were similar to those on anatomical cadaver studies and to the one that also used MRI images **.
There was a clear predominance of the IMA in terms of vascular supply to both the breast and NAC, especially the 2nd branch of the IMA. There are many anatomical variations on breast blood supply, and a true symmetry between both breasts only exists in about half the cases (this asymmetry seems to be greater in the invasive carcinoma group, which did not reach statistical significance in our study, probably due to sample size).
There is a neovascularization caused by the tumors which was visible in the MRI images and caused a significant statistical difference in NAC blood supply - a much greater percentage of NAC supplied by the UOQ in relation to predominant UOQ tumors, compared with the screening group.
It is of the utmost importance to mention that none of NAC analyzed was supplied by a predominant vessel coming from the LOQ, suggesting that this quadrant is an ideal candidate for the incision site whenever possible. The choice of the LOQ as the ideal place for incisions in NSM had already been reported by Colwell et al. [30] to be beneficial in terms of vascular complications.
We should emphasize that most of the breasts (87%) and NACs (71%) in our study had indeed a predominant blood vessel responsible for the vascular blood supply; hence if we want to reduce NAC necrosis and other vascular complications it is of the essential to preserve it.
In terms of impact in NAC necrosis in the NSM procedure, despite being a secondary objective with a small number of patients in this study, we found that having more than one vessel supplying the NAC was a protector factor against necrosis probably because the chance of ligating both the pedicles is low.
Conclusion
Our study demonstrated that submitting patients to breast MRI pre-operatively to evaluate breast and NAC vascular map is feasible without adding extra MRI time or contrast. It was possible to assess the vascular map in all patients.
We could see a dominant blood supply to the breast in 87% and to the NAC in 75%, so every effort should be made to preserve the vascular anatomy in order to reduce NAC necrosis.
There’s a predominance of the internal mammary artery in supplying both the breast and the NAC. This vascular supply usually enters the NAC through the internal quadrants. In our study, there was not a single breast with dominant vascular supply from the LOQ, which makes this area optimal for incision in NSM.
There is an asymmetry between cancer and screening breasts mainly at NAC level with neovascularization of this area. We reported a tendency for a higher number of vessels vascularizing the NAC in the IC group (p=0.056) and a significant statistical difference in quadrant vascularization at UOQ. Almost half of the tumors were located in this quadrant. This UOQ localization was responsible for a neovascularization of the dominant blood vessel to the NAC and we found that in the oncological population 18% had the dominant vessel in this quadrant vs. 2% in the HRS group (p=0.048).
In terms of variables with impact in NAC necrosis in NSM, despite the small number of patients in this subgroup, single NAC blood supply was a risk factor for NAC necrosis (p=0.01).
References
- Petit JY, Veronesi U, Orecchia R et al. Risk factors associated with recurrence after nipple-sparing mastectomy for invasive and intraepithelial neoplasia. Ann. Oncol. 23, 2053-2058 (2012).
- de Alcantara Filho P, Capko D, Barry JM et al. Nipple-sparing mastectomy for breast cancer and risk-reducing surgery: The Memorial Sloan-Kettering Cancer Center experience. Ann. Surg. Oncol. 18, 3117-3122 (2011).
- Jensen JA, Orringer JS, Giuliano AE. Nipple-sparing mastectomy in 99 patients with a mean follow-up of 5 years. Ann. Surg. Oncol. 18, 1665-1670 (2011).
- Spear SL, Willey SC, Feldman ED et al. Nipple-sparing mastectomy for prophylactic and therapeutic indications. Plast. Reconstr. Surg. 128, 1005-1014 (2011).
- Lohsiriwat V, Martella S, Rietjens M et al. Paget’s disease as a local recurrence after nipple-sparing mastectomy: Clinical presentation, treatment, outcome and risk factor analysis. Ann. Surg. Oncol. 19, 1850-1855 (2012).
- Murthy V, Chamberlain RS. Nipple-sparing mastectomy in modern breast practice. Clin. Anat. 26, 56-65 (2013).
- Wagner JL, Fearmonti R, Hunt KK et al. Prospective evaluation of the nipple-areola complex sparing mastectomy for risk reduction and for early-stage breast cancer. Ann. Surg. Oncol. 19, 1137-1144 (2012).
- Petit JY, Veronesi U, Luini A et al. When mastectomy becomes inevitable: The nipple-sparing approach. Breast. 14, 527-531 (2005).
- Mortenson MM, Schneider PD, Khatri VP et al. Immediate breast reconstruction after mastectomy increases wound complications: However, initiation of adjuvant chemotherapy is not delayed. Arch. Surg. 139, 988-991 (2004).
- Caruso F, Ferrara M, Castiglione G et al. Nipple sparing subcutaneous mastectomy: Sixty-six months follow-up. Eur. J. Surg. Oncol. 32, 937-940 (2006).
- Sacchini V, Pinotti JA, Barros AC et al. Nipple-sparing mastectomy for breast cancer and risk reduction: Oncologic or technical problem? J. Am. Coll. Surg. 203, 704-714 (2006).
- Komorowski AL, Zanini V, Regolo L et al. Necrotic complications after nipple-and areola-sparing mastectomy. World. J. Surg. 30, 1410-1413 (2006).
- Crowe JP, Patrick RJ, Yetman RJ et al. Nipple-sparing mastectomy update: One hundred forty-nine procedures and clinical outcomes. Arch. Surg. 143, 1106-1110 (2008).
- Garcia ECA, Cody IHS, Disa JJ et al. Nipple-sparing mastectomy: Initial experience at the Memorial Sloan-Kettering Cancer Center and a comprehensive review of literature. Breast. J. 15, 440-449 (2009).
- Garwood ER, Moore D, Ewing C et al. Total skin-sparing mastectomy: complications and local recurrence rates in 2 cohorts of patients. Ann. Surg. 249, 26-32 (2009).
- Rusby JE, Smith BL, Gui GP. Nipple-sparing mastectomy. Br. J. Surg. 97, 305-316 (2010).
- Algaithy ZK, Petit JY, Lohsiriwat V et al. Nipple sparing mastectomy: can we predict the factors predisposing to necrosis? Eur. J. Surg. Oncol. 38, 125-129 (2012).
- Chirappapha P, Jean YP, Mario R. Nipple sparing mastectomy. Plast. Reconstr. Surg. Glob. Open. 2, e99 (2014).
- Houssami N, Turner R, Macaskill P et al. An individual person data meta-analysis of preoperative magnetic resonance imaging and breast cancer recurrence. J. Clin. Oncol. 32, 392-401 (2014).
- Pilewskie M, King TA. Magnetic resonance imaging in patients with newly diagnosed breast cancer. Cancer. 15, 2080-2089 (2014).
- Stout NK, Nekhlyudov L, Li L et al. Rapid increase in breast magnetic resonance imaging use: Trends from 2000 to 2011. JAMA. Intern. Med. 174, 114-121 (2014).
- Parker A, Schroen AT, Brenin DR. MRI utilization in newly diagnosed breast cancer: a survey of practicing surgeons. Ann. Surg. Oncol. 20, 2600-2606 (2013).
- Seitz IA, Nixon AT, Friedewald SM et al. “NACsomes”: A new classification system of the blood supply to the nipple areola complex (NAC) based on diagnostic breast MRI exams. J. Plast. Reconstr. Aesthetic. Surg.68, 792-799 (2015).
- Van Deventer PV. The blood supply to the nipple-areola complex of the human mammary gland. Aesthetic. Plast. Surg. 28, 393-398 (2004).
- Nakajima H, Imanishi N, Aiso S. Arterial anatomy of the nipple-areola complex. Plast. Reconstr. Surg. 96, 843-845 (1995).
- O’Dey D, Prescher A, Pallua N. Vascular reliability of nipple- areola complex-bearing pedicles: An anatomical microdissection study. Plast. Reconstr. Surg. 119, 1167-1177 (2007).
- Bleicher, RJ, Chase F. Breast magnetic resonance imaging as it is, in contrast to how we wish it to be. J. Clin. Oncol. 32, 370-372 (2014).
- Liu DZ, Mathes DW, Zenn MR et al. The application of indocyanine green fluorescence angiography in plastic surgery. J. Reconstr. Microsurg. 27, 355-364 (2011).
- Wapnir I, Dua M, Kieryn A et al. Intraoperative imaging of nipple perfusion patterns and ischemic complications in nipple-sparing mastectomies. Ann. Surg. Oncol. 21, 100-106 (2014).
- Colwell AS, Tessler O, Lin AM et al. Breast reconstruction following nipple- sparing mastectomy: Predictors of complications, reconstruction outcomes and 5 year trends. Plast. Reconstr. Surg. 133, 496-506 (2014).