Research Article - Imaging in Medicine (2019) Volume 11, Issue 5
Retrospective Investigation Of The Relationship Between The Stylohyoid Ligament And Carotid Dissection
Halil İbrahim Özdemir1*, Celal Çınar2 & Murat Köylü31Department of Radiology, Ege University Faculty of Medicine, Bornova/Izmir, Turkey
2Department of Radiology (Interventional Radiology), Ege University Faculty of Medicine, Bornova/Izmir, Turkey
3Department of Radiation Oncology, Ege University Faculty of Medicine, Bornova/Izmir, Turkey
- Corresponding Author:
- Halil İbrahim Özdemir
Department of Radiology
Ege University Faculty of Medicine
Bornova/Izmir, Turkey
E-mail: halil.ibrahim.ozdemir@ege.edu.tr
Abstract
Purpose : Carotid dissection is the most important cause of ischemic stroke in young adults. Ischemic stroke rate up to 20% in people under 50 years. Recently, young patients under 50 years of age who were admitted to the emergency department with ischemic stroke complaints were diagnosed with carotid dissection.
Methods : For this purpose, cranio-cervical CT angio images of patients admitted to our clinic with suspected carotid artery dissection and diagnosed as ICA dissection were retrospectively reviewed. the characteristics of the patients with ICA dissection and normal patients undergoing cranio-cervical CT angiography were investigated using 3D anatomical programs, specifically the course of the stylohyoid ligament, the distance between stylohyoid ligament and the carotid artery, and contact between stylohyoid ligament and the carotid artery. Sectra and AW Server 2 archives and XIO 4.80 radiotherapy planning programs were used for 3D analysis of the images. The data obtained from the analyzes were evaluated using the SPSS statistical program.
Results : Descriptive statistics, Independent Samples T-Test, Pearson correlation analysis, multiple regression analysis, and graphical programs were used in statistical evaluations. According to Independent Samples T-Test results performed under SPSS, the distance between the ICA with stylohyoid ligament at 0.05 (sig.2-tailed) level (t = 5,950, sig = 0,000) and the difference between the age of the patients (t = 2,226, sig = 0,026) was significant. In addition, the axial (t = 2,938, sig = 0,005) and sagittal (t = 2,307, sig = 0,026) angles between the genders were different. In Pearson correlation analysis, styloid axial angles (r = -0.316, sig = 0.029), styloid sagittal angles (r = -0.333, sig = 0.020) and age of ICA dissection (r = -0.332, sig = 0.026) at 0,05 (2-tailed) level was found to be correlated. More importantly, in patients with ICA dissection, it was found that there was a strong correlation between stylohyoid ligament-carotid distance (r = -0.659, sig = 0.000) at the 0.01 (2-tailed) level.
Conclusion : As a result, the closure of the stylohyoid ligament closer than 3.9 mm to the carotid for any reason increases the risk of dissection in young patients. It is thought that the results of this research will constitute a step for radiological artificial intelligence studies.
Keywords
Stylohyoid Ligament (SHL) ■ Carotid Dissection (CD) ■ Computed Tomography Angiography (CTA)
Introduction
Carotid artery or vertebral artery dissections account for 2% of all ischemic strokes [1]. In young adults, carotid artery dissections are the most important causes of ischemic stroke; under the age of 50, this rate is increases 20% [2,3]. The main mechanism of development of carotid artery dissection is rupture of the carotid artery in the intima layer and the blood in that region progressing through the artery, causing an embolic or aneurysmatic pseudolumen in the subintimal or subadvential layers to disrupt the blood flow dynamics and cause ischemia [4-6]. Carotid artery dissections may be spontaneous (fibromuscular dysplasia, connective tissue diseases, hypertension, syphilis, migraine) or traumatic (blunt and penetrating head-neck traumas, treadmill running, vomiting, giving birth, coughing). Spontaneous dissections account for up to 60% of cases, while the rest are traumatic dissections. The most important findings of carotid artery dissections are ischemic (headache, neck pain, focal neurological symptoms, transient loss of vision, vertigo, pulsatile tinnitus, and Horner's syndrome) [7,8]. In addition, some investigators reported that styloid process elongation spontaneously, have caused carotid artery dissection. There are many studies reporting that dissection may occur as a result of the deviation and mechanical compression of 3 cm or more in an elongated styloid process on the external carotid artery (ECA) or internal carotid artery (ICA), also known as Eagle syndrome. There are also studies reporting that the proximity of styloid-hyoid bone may be a risk factor causing ICA dissection. The stylohyoid complex consists of the styloid process of the temporal bone, the stylohyoid ligament, and the lesser horn of the hyoid bone. This complex completes its formation in childhood and the adolescence. In adulthood, it may be calcified with age. Stylohyoid syndrome is also associated with abnormalities of these structures. These abnormalities include elongation of the styloid or hyoid bones, elongation of the carotid artery, developmental angulation, calcification of the stylohyoid ligament, or combinations of these pathologies [9-18]. Stylohyoid syndrome is thought to be caused by its association with structures around the stylohyoid complex or by irritation of neighboring structures with minimal neck movements. Recently, younger patients under 50 years of age who were admitted to the emergency department with complaints of ischemic stroke, diagnosed with carotid dissection without the above mentioned spontaneous or traumatic causes. In some of the patients, it was noteworthy that there was no cause of ICA dissection, and the majority of them were found to be young. It was thought that the stylohyoid ligament extending between the lesser horns of the hyoid bone of the same side with the styloid process was close to the carotid and might cause dissection by the rubbing of the carotid with neck movements. However, it is not possible to visualize the stylohyoid ligament through imaging methods used today (CT, MRI, US, etc.). However, on the volumetric CT data, the lower side of the segmented styloid bone can be overlapped with the same side of the hyoid bone lesser horns, and the distance of this superposition point to the carotid artery can be measured; or, again on the volumetric data, a line representing the stylohyoid ligament between the lower side of the styloid bone and the lesser horn of the same side can be drawn, and the closest distance to the carotid artery can be determined. It is also known that the anatomical course of the stylohyoid ligament between the styloid bone and the hyoid bone lesser horns is not in the form of a straight line and follows a curved path that faces the medial. This study was carried out to prove the hypothesis that a ligament might cause ICA dissection with friction caused by the rapid progression of the stylohyoid ligament and rapid neck movements.
Materials and Methods
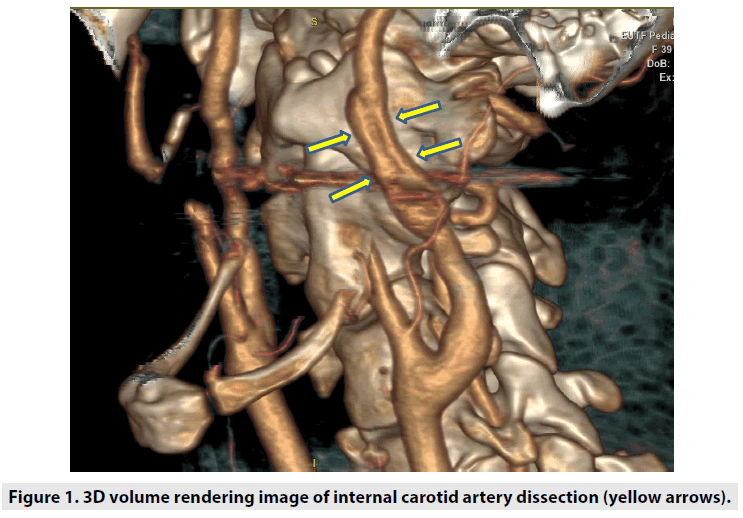
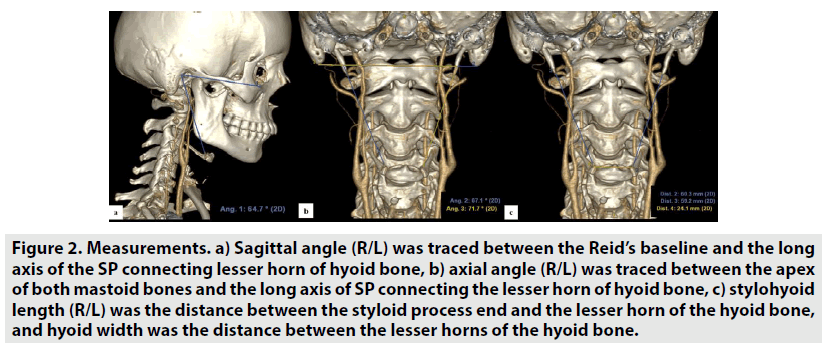
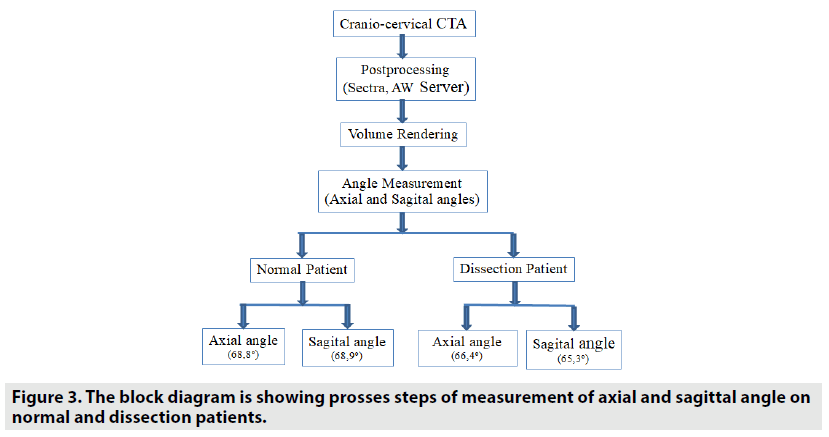
For this study, ethical committee approval was obtained. In order to prove the hypothesis, angiographic cranio-cervical CT images of the patients who were admitted to our clinic with the suspicion of carotid artery dissection and were diagnosed with ICA dissection were screened retrospectively (FIGURE 1). Patients with known causes of ICA dissections (Styloid elongation, eagle syndrome, mechanical compression due to anatomic variation of the styloid process, mechanical compression and stylohyoid ligament calcification as a result of contact with the carotid artery) were excluded from study not included in the. The data of ICA dissection patients having a styloid bone smaller than 3 cm and no mechanical compression or contact of the styloid bone on the carotid artery were used. The data were compared with the data of normal patients who subjected to cranial-cervical CT angiography for any reason. In this comparison, the relationship between the stylohyoid complex and the carotid artery was investigated in order to determine the characteristics of the stylohyoid ligament, its distance from the carotid artery, and its contact with the carotid artery. Angiographic craniocervical CT scans of the patients were performed with care dose CT with 128 slices (Somatom Definition, Siemens Healthcare, Erlangen, Germany). The scan parameters used for neck-brain CTA were kVp:120, mA: dose modulated, rotation time: 0.3 sec, thickness:0.6 mm, pitch:0.8, coverage: 76.8 mm, kernel filter: 326f medium smooth, matrix:512*512 and FOV: 230 mm. In each patient, an intra-venous iodinated non-ionic contrast medium was used with the help of an automatic injector. Images were analyzed using 3D volume rendering analysis programs in Sectra and AW Server 2 archive systems. In the analyzes, the right and left styloid bone length, the axial and sagittal angles of the styloid bone with the base of the skull, the shortest distance to which the stylohyoid ligament plane approached the carotid, the distance between the stylohyoid ligament length and the hyoid bone lesser horn were measured. The styloid axial angle measurements, the line passing from the mastoid bone apex (parallel to the base of the head), and the long axis of the styloid bone (extending to the hyoid lesser horns) angle was used. In the sagittal angle measurement, the angle between the long axis of the styloid bone (extending to the hyoid lesser horns) and the Raid’s baseline (line between the infraorbital margin and the auricular point) were used (19,20) (FIGURE 2A-C). The block diagram showing the axial and sagittal angle measurement, presses of normal and dissected patients on volume rendering images is shown in FIGURE 3.
Figure 2: Measurements. a) Sagittal angle (R/L) was traced between the Reid’s baseline and the long axis of the SP connecting lesser horn of hyoid bone, b) axial angle (R/L) was traced between the apex of both mastoid bones and the long axis of SP connecting the lesser horn of hyoid bone, c) stylohyoid length (R/L) was the distance between the styloid process end and the lesser horn of the hyoid bone, and hyoid width was the distance between the lesser horns of the hyoid bone.
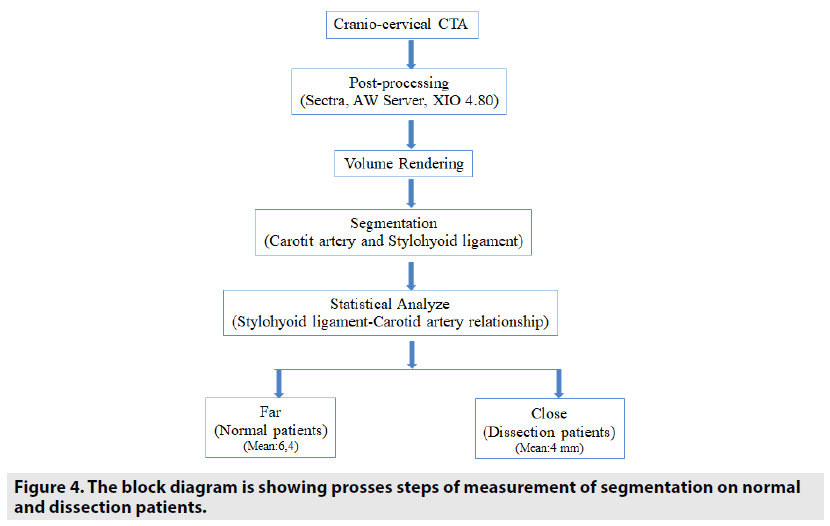
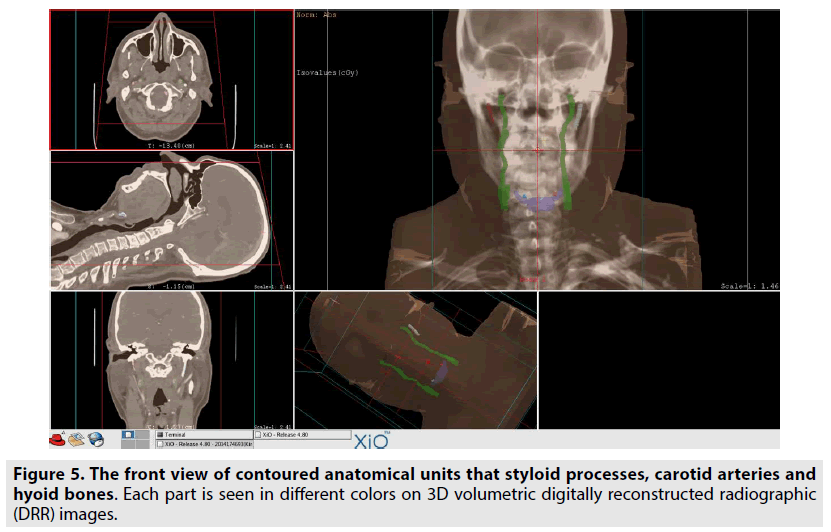
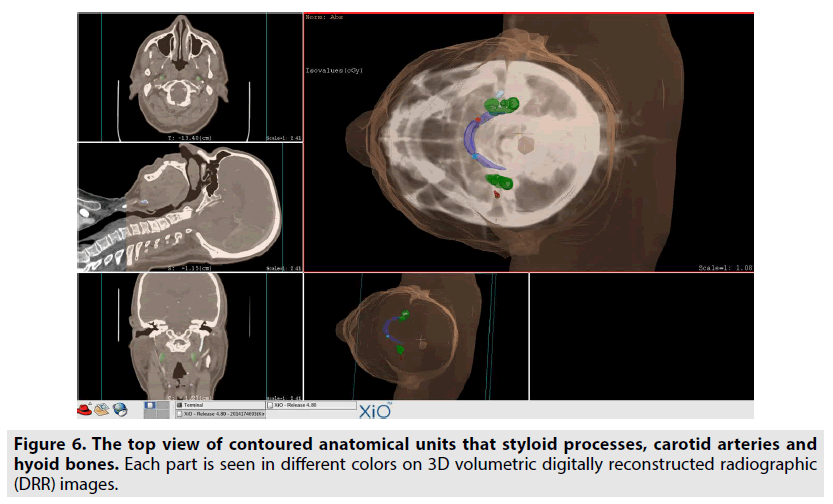
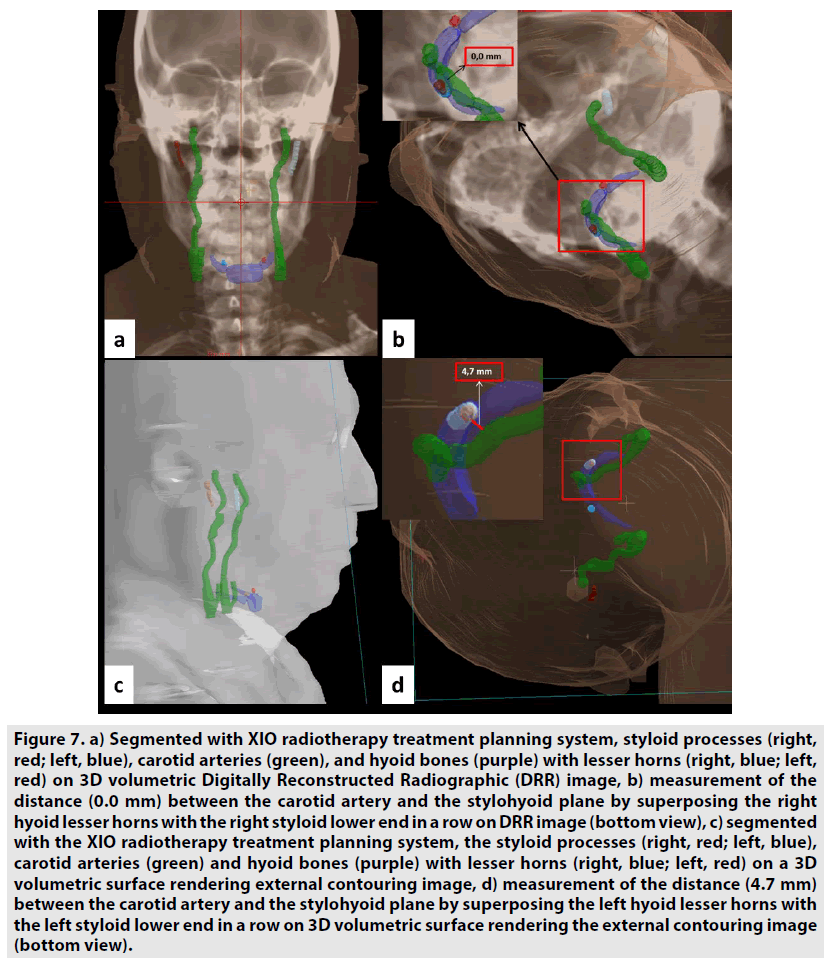
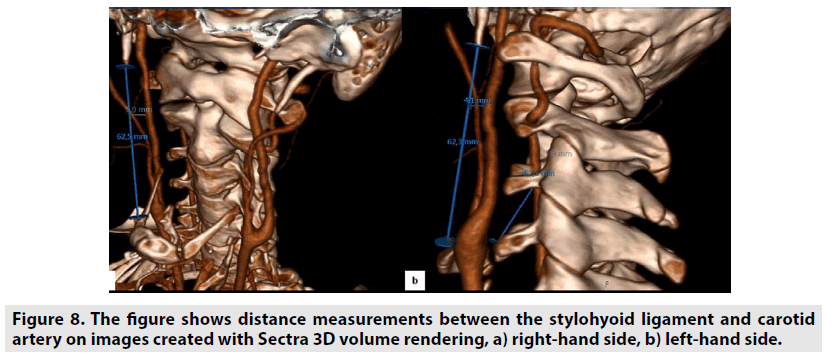
In addition, data of the same patients were segmented by contouring the styloid processes, the carotid arteries, and the hyoid bones in images using XIO 4.80 radiotherapy treatment planning systems. The block diagram showing the segmentation process steps is given in the FIGURE 4. Styloid processes, hyoid bones with anterior horns and carotid arteries were contoured on the images of both normal and dissected patients. The front and top view of these contoured anatomical units is also seen in FIGURES 5 & 6 on 3D volumetric digitally reconstructed radiographic (DRR) images. On volumetric contoured 3D images, the stylohyoid ligament extends, the lower end of the styloid bone, and the lesser horns of the same side hyoid bone were overlapped, and the distance from this point to the carotid artery was measured (FIGURE 7A-D). The same distance measurements were repeated by drawing a ligament line between the styloid bone lower end and the hyoid bone lesser horns by using the volumetric rendering and vascular analysis options of the AW Server 2 and Sectra image processing programs. Data we obtained with the AW Server 2 and Sectra programs were consistent with the data related to XIO radiotherapy treatment planning program (FIGURE 8A & B).
Figure 7: a) Segmented with XIO radiotherapy treatment planning system, styloid processes (right, red; left, blue), carotid arteries (green), and hyoid bones (purple) with lesser horns (right, blue; left, red) on 3D volumetric Digitally Reconstructed Radiographic (DRR) image, b) measurement of the distance (0.0 mm) between the carotid artery and the stylohyoid plane by superposing the right hyoid lesser horns with the right styloid lower end in a row on DRR image (bottom view), c) segmented with the XIO radiotherapy treatment planning system, the styloid processes (right, red; left, blue), carotid arteries (green) and hyoid bones (purple) with lesser horns (right, blue; left, red) on a 3D volumetric surface rendering external contouring image, d) measurement of the distance (4.7 mm) between the carotid artery and the stylohyoid plane by superposing the left hyoid lesser horns with the left styloid lower end in a row on 3D volumetric surface rendering the external contouring image (bottom view).
These data obtained from patients with normal and dissection diagnosis were compared and evaluated by using descriptive statistics, Independent Samples T-Test, Anova test, Pearson correlation analysis, multiple regression analysis, and graphical programs under SPSS.
Results
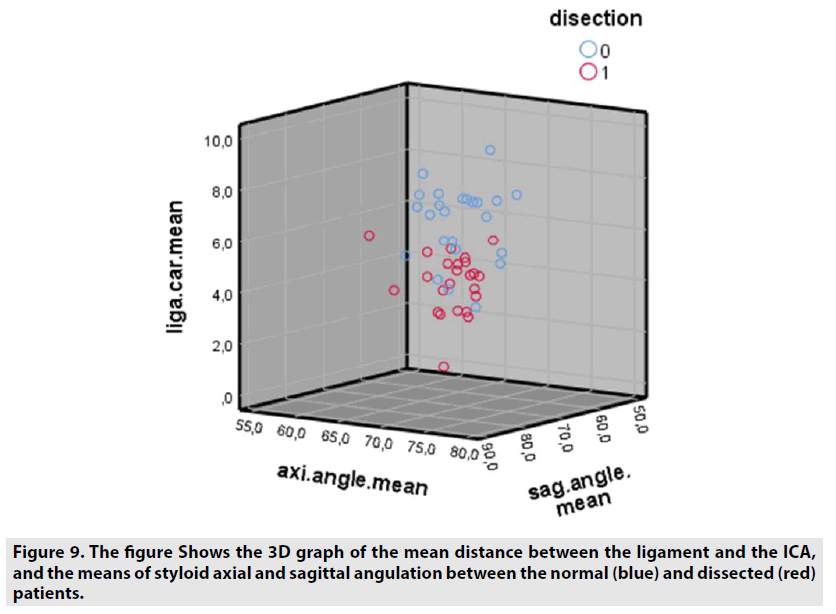
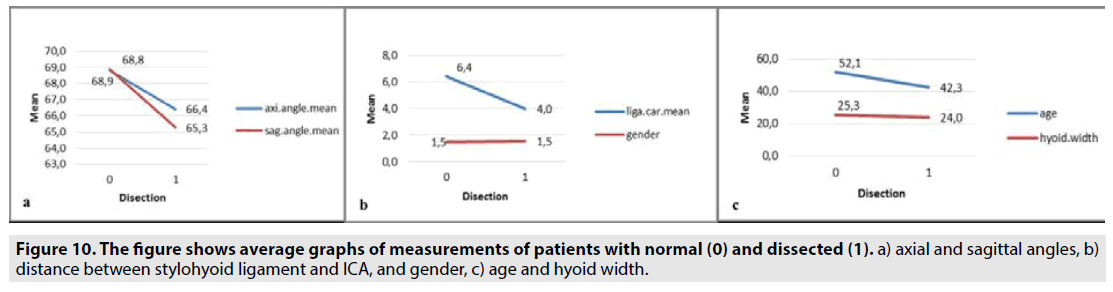
A total of 48 patients were included in the study, 24 of them were normal (13M, 11F) and 24 had ICA dissection (11M, 13F). Of the 24 patients with ICA dissection, 18 had right ICA dissection and 6 had left ICA dissection. It was observed that right-side ICA dissection was more common. In normal patients, the distances between the right-left side stylohyoid ligament and the carotid artery were not different. Descriptive information and group statistics of the study are given in TABLES 1 & 2. Comparative graphs of ligament-carotid distance, styloid axial, and sagittal angulation, hyoid width, age, and sex averages in patients and also the 3D graph of the data of patients with normal and ICA dissection are given below (FIGURE 9). In patients with dissection, the stylohyoid ligament was markedly close to the carotid, and the axial and sagittal styloid angles were narrowed. According to Independent Samples T-Test results performed under SPSS; the distance between ICA and stylohyoid ligament at 0.05 level (sig. 2-tailed) (t = 5.950, sig = 0,000), and the difference between the age of the patients was statistically significant (t = 2.226, sig = 0.026). In addition, the axial (t = 2.938, sig = 0.005) and sagittal (t = 2.307, sig = 0.026) angles between the genders were different. In other words, the axial and sagittal opening of styloid bones in females were found to be narrower than in males. However, no difference was found between styloid bone lengths (0.266), hyoid lesser horns (hyoid width) distances (0,052), stylohyoid ligament lengths (0.065) and genders (0,695). FIGURE 10A-C shows the graphs of the average measurements of patients with normal (0) and dissection.
| Group Statistics | |||||
|---|---|---|---|---|---|
| disection | N | Mean | Std. Deviation | Std. Error Mean | |
| liga.car.mean | 0 | 24 | 6,4 | 1,6336 | ,3335 |
| 1 | 24 | 4,0 | 1,1660 | ,2380 | |
| axi.angle.mean | 0 | 24 | 68,8 | 3,7707 | ,7697 |
| 1 | 24 | 66,5 | 3,5963 | ,7341 | |
| sag.angle.mean | 0 | 24 | 68,9 | 5,3520 | 1,0925 |
| 1 | 24 | 65,1 | 4,7127 | ,9620 | |
| age | 0 | 24 | 52,1 | 15,046 | 3,071 |
| 1 | 24 | 42,3 | 14,484 | 2,957 | |
Table 1. Shows descriptive and informative group statstics.
| Group Statistics | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| N | Min. | Max. | Mean | Gender | N | Mean | Disection | N | Mean | Side | Mean | |
| liga.car.mean | 48 | 1,0 | 9,4 | 5,2 | M | 23 | 5,1 | 0 | 24 | 6,4 | R | 5,0 |
| F | 25 | 5,3 | 1 | 24 | 4,0 | L | 5,3 | |||||
| axi.angle.mean | 48 | 57,6 | 76,1 | 67,6 | M | 23 | 69,2 | 0 | 24 | 68,8 | R | 67,7 |
| F | 25 | 66,2 | 1 | 24 | 66,1 | L | 67,5 | |||||
| sag.angle.mean | 48 | 52,4 | 82,9 | 67,1 | M | 23 | 68,8 | 0 | 24 | 68,8 | R | 66,6 |
| F | 25 | 65,4 | 1 | 24 | 65,2 | L | 67,4 | |||||
| age | 48 | 13 | 73 | 47,2 | M | 23 | 48,7 | 0 | 24 | 52,1 | ||
| F | 25 | 45,8 | 1 | 24 | 42,3 | |||||||
Table 2. Shows descriptive and informative data.
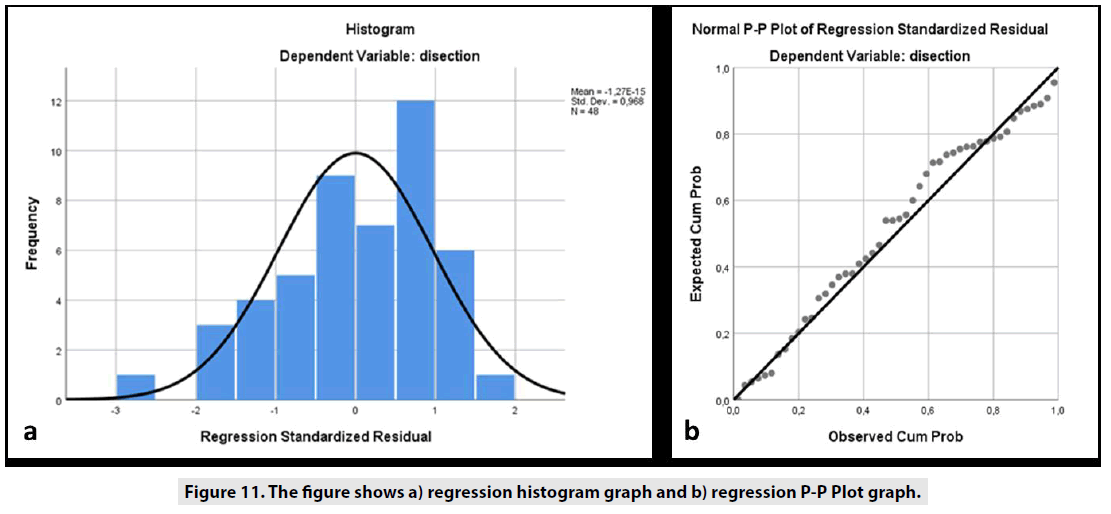
In Pearson correlation analysis under SPSS, styloid axial angles (r = -0.316, sig = 0.029), styloid sagittal angles (r = -0.333, sig = 0.020) and age of ICA dissection (r = -0.332, sig = 0.026) at 0,05 (2-tailed) level was found to be correlated. More importantly, in patients with ICA dissection, it was found that there was a strong correlation between stylohyoid ligamentcarotid distance (r = -0.659, sig = 0.000) at the 0.01 (2-tailed) level. In addition, the Anova test (sig = 0,000) was found to be significant in the multiple regression analysis, and Adjust square R (R = 0.492, 49.2%) results showed that the stylohyoid ligament-carotid distance, the axial styloid angle, and the sagittal styloid angle were the causes of ICA dissection with 49.2%. The distance between the stylohyoid ligament and the carotid was found to be significantly effective in the ICA dissection (B = -0.617, t = -5.870 sig = 0.000). The non-standardized constant value was found to be as B = 3.925. In other words, the critical distance between the stylohyoid ligament and the carotid is 3.9 mm, with the risk of for developing ICA dissection with the neck movements of the stylohyoid ligament at values below this distance. According to statistical data, the fact that this distance increased by 1 unit (about on unit increase in this distance) means that the ICA dissection risk has reduced by 0.167 units. The histogram and P-P Plot graphs of the regression analyzes are shown in FIGURE 11A & B).
Discussion
The lateral deviation of in the styloid projection causes compression in the external carotid artery, and medial deviation causes damage to the internal carotid artery and the tonsils. Posterior deviation causes irritation of in the last four cranial nerves, while anterior deviation leads to mucosal irritation, and tonsils cause stress in vital structures [19-22]. It is possible that the Medial deviation of in styloid can cause stylohyoid ligament to converge on the internal carotid artery and also dissection with neck movements. As a matter of fact, Dimitri et al. emphasized that the styloid or hyoid bone proximity is the most important environmental risk factor for carotid artery dissections [23]. The results obtained in our study showed that the sagittal angulation of the styloid bones was medial, and the axial angulation was narrowed by anterior deviation. This suggests that the styloid processes of patients with unresolved ICA dissection are malposed [24]. In a similar study by De Paz et al., of 835 craniums in anatomy museum, they found the axial styloid angulation as 53° and the sagittal angulation as 65° [25]. Again, Andrei et al., found no difference between the right and left angles of the axial (66.7) and sagittal (53.9) angle measurements of in 54 patients on their dental CT images. In the same study, they reported that the sagittal opening was due to increased styloid process length; and the axial angulation was higher in males [18]. Thanuja et al., carried out a study with the same number of patients but with using a different method, found that the sagittal angulation was 80.6 degrees in males and 82.4 degrees in females, and the axial angulation was 25.2 degrees in males and 31.1 degrees in females. As a result, no statistically significant differences were found between the genders and the right / left side [26]. Crohn et al., found the mean styloid sagittal angle to be 56.7 degrees and a significant difference between males (63.4) and females (54.9) [27]. In our study, sagittal (M: 68,8, F: 65,4) and axial (M: 69,2, F: 66,2) angulation were found to be statistically significant in the male / female comparison. Patil et al., found a significant difference between sagittal angles, 52.4 - 60.1 degrees in study patients and 61.0 - 69.0 degrees in healthy people [28]. Bashekim et al., in their study found the axial angle between 60.6 and 84.1 ° (mean 69.4°) among age groups [29]. In a study conducted by Buyuk et al., they measured 1,000 patients with different reference points, and found significant in the sagittal angle between females (70.4) and males (74.0) and in the axial angle between females (70.3) and males (72.2). In our measurements, there was no difference between the sides in the axial angulation (right: 6.,7 and left: 67.5; mean: 67.6) and sagittal angulation (right: 66.6 and left: 67.4; mean: 67.1). However, the axial angulation (N: 69.8, dis: 66.4) and sagittal angulation (N: 68.5, dis: 65.5) were found to be different between normal patients and patients with carotid dissection [30].
Conclusion
The tendency of the stylohyoid ligament to be closer than 3.9 mm to any segment of the carotid artery (especially ICA) due to the narrowing of the axial-sagittal recessions of the styloid process and carotid elongation will increase the contact and friction between the stylohyoid ligament and the carotid artery and it causes carotid dissection. Younger age and a life with active neck movement can support this process. As a result, the closure of the stylohyoid ligament closer than 3.9 mm to the carotid for any reason, may increase the risk for dissection in younger patients. It is thought that the results of this study and the approach of this research will be a step towards the current radiological artificial intelligence studies.
Highlights
1. Increased carotid dissection in young patients presenting to the emergency department in recent years.
2. Diagnosis of unspecified carotid dissection except for trauma or mechanical pressure.
3. The majority of patients with carotid dissection are young.
References
- Bray F, Ferlay J, Soerjomataram I et al. Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA. Cancer. J. Clin. 68: 394-424, (2018).
- Vergouwen MD, Intravenous thrombolysis in ischaemic stroke secondary to cervical artery dissection: safe but not effective? Eur. J. Neurol. 19: 1155-1156, (2012).
- Putaala J, Metso AJ, Metso TM et al. Analysis of 1008 consecutive patients aged 15 to 49 with first-ever ischemic stroke: the Helsinki young stroke registry. Stroke. 40: 1195-1203, (2009).
- Schievink WI, Spontaneous dissection of the carotid and vertebral arteries. N. Engl. J. Med. 344: 898-906, (2001).
- Poppe AY, Minuk J, Glikstein R et al. Fibromuscular dysplasia with carotid artery dissection presenting as an isolated hemianopsia. J. Stroke. Cerebrovasc. Dis. 16:130-134, (2007).
- Macdonald DJ, McKillop EC. Carotid artery dissection after treadmill running. Br. J. Sports. Med. 40: 10, (2006).
- Caplan LR. Dissections of brain-supplying arteries. Nat. Clin. Pract. Neurol. 2008; 4:34-42.
- Eagle WW. Elongated styloid process; further observations and a new syndrome. Arch. Otolaryngol. 47: 630-640, (1948).
- Eagle WW. Symptomatic elongated styloid process; report of two cases of styloid process-carotid artery syndrome with operation. Arch. Otolaryngol. 49: 490-503, (1949).
- David J, Lieb M, Rahimi SA. Stylocarotid artery syndrome. J. Vasc. Surg. 60: 1661-1663, (2014).
- Soo OY, Chan YL, Wong KS. Carotid artery dissection after prolonged head tilting while holding a newborn baby to sleep. Neurology. 2004; 62:1647-1648.
- Zuber M, Meder JF, Mas JL. Carotid artery dissection due to elongated styloid process. Neurology. 53: 1886-1887, (1999).
- Chuang WC, Short JH, McKinney AM et al. Reversible left hemispheric ischemia secondary to carotid compression in Eagle syndrome: surgical and CT angiographic correlation. AJNR. Am. J. Neuroradiol. 28: 143-145, (2007).
- Basekim CC, Mutlu H, Gungor A et al. Evaluation of styloid process by three-dimensional computed tomography. Eur. Radiol. 15: 134-139, (2005).
- Piagkou M, Anagnostopoulou S, Kouladouros K et al. Eagle’s syndrome: a review of the literature. Clin. Anat. 22: 545-558, (2009).
- Faivre A, Abdelfettah Z, Rodriguez S et al. Bilateral internal carotid artery dissection due to elongated styloid processes and shaking dancing. J. Neurol. Neurosurg. Psychiatry. 80: 1154-1155, (2009).
- Raser JM, Mullen MT, Kasner SE et al. Cervical carotid artery dissection is associated with styloid process length. Neurology. 77: 2061-2066, (2011).
- Renard D, Azakri S, Arquizan C et al. Styloid and Hyoid Bone Proximity Is a Risk Factor for Cervical Carotid Artery Dissection.Stroke. 44: 2475-2479, (2013).
- Andrei F, Motoc AG, Didilescu AC et al. A 3D cone beam computed tomography study of the styloid process of the temporal bone. Folia. Morphol. 72: 29-35, (2013).
- Onbas O, Kantarci M, Murat KR, et al. Angulation, length and morphology of the styloid process of the temporal bone analyzed by multidetector computed tomography. Acta. Radiol. 46: 881-886, (2005).
- Lengele BG, Dhem AJ. Length of the styloid process of the temporal bone. Arch. Otolaryngol. Head. Neck. Surg. 114: 1003-1006, (1988).
- Piagkou M, Anagnostopoulou S, Kouladouros K et al. Eagle’s syndrome: A review of the literature. Clin. Anat. 22: 545-558, (2009).
- Mazzetto MO, Andrade KM, Magri LV et al. Anterior and medial angulations of the styloid process in subjects with TMD: clinical and radiographic findings. Braz. Dent. J. 24: 80-84, (2013).
- Renard D, Azakri S, Arquizan C et al. Styloid and Hyoid Bone Proximity Is a Risk Factor for Cervical Carotid Artery Dissection.Stroke. 44: 2475-2479, (2013).
- Frommer JJ. Anatomic variations in the stylohyoid chain and their possible clinical significance. Oral. Surg. Oral. Med. Oral. Pathol. 38: 659-667, (1974).
- De-Paz FJ, Rueda C, Barbosa M et al. Biometry and statistical analysis of the styloid process. Anat. Rec. 295: 742-747, (2012).
- Thanuja R, Karan S. Assessment of the Styloid Process by Cone Beam Computed Tomography. Int. J. Radiol. Radiat. Ther. 2: 123‒127, (2017).
- Krohn S, Brockmeyer P, Kubein-Meesenburg D et al. Elongated styloid process in patients with temporomandibular disorders - Is there a link? Ann. Anat. 217: 118-124, (2018).
- Patil S, Ghosh S, Vasudeva N. Morphometric study of the styloid process of temporal bone. Clin. Diagn. Res. 8: 04-06, (2014).
- Buyuk C, Gunduz K, Avsever H. Morphological assessment of the stylohyoid complex variations with cone beam computed tomography in a Turkish population. Folia. Morphol. 77: 79-89, (2018).