Research Article - International Journal of Clinical Rheumatology (2019) Volume 14, Issue 4
Study of prevalence of hypothyroidism in rheumatoid arthritis patients and its impact on disease severity
- Corresponding Author:
- Haitham A Azeem
Department of Internal Medicine, Al-Azhar University, Assiut, Egypt
E-mail:h_alashwal@azhar.edu.eg
Abstract
Objective: To establish the incidence and effect of hypothyroidism in Rheumatoid Arthritis (RA) patients and to analyze whether or not the presence of hypothyroidism related to RA disease severity or not. Methods: We reviewed information of all RA patients in period from March 2018 to August 2019 and chosen all those that had thyroid results recorded in their laboratory information (free thyroxine, free triiodothyronine and thyroid-stimulation hormone). we have a tendency to examined all laboratory results of the sample, to assess the incidence of hypothyroidism among those patients, in regards to the laboratory standard results. We have a tendency to studies the co-exciting between hypothyroidism and RA disease severity. Results: Our study include 1000 adult RA patients (850 female and 150 male) was previously diagnosed as RA patients recruited randomly from patients attending the Out-patient clinics and In-patients of Medicine Departments of Al-Sheikh Zayed Al-Nahian hospital and Azhar Assiut University hospital in the period from March 2018 to August 2019. Thyroid abnormalities were detected among 280 patients (28%) represented as Hypothyroidism 270 patients (Subclinical Hypothyroidism 210 patients - Clinical Hypothyroidism 60 patients) and Hyperthyroidism 10 patients (Subclinical Hyperthyroidism 8 patients - Clinical Hyperthyroidism 2 patients) While we couldn’t detect any case with Sick euthyroid Disease. While the RA patients with euthyroid stat was 720 patients. Conclusion: hypothyroidism is not uncommon in RA and it should be investigated since it is a risk factor for increasing the disease severity. This conclusion may help farther study to improve the patient's prognosis, and to establish public health protocols.
Introduction
RA may be a general autoimmune disorder characterized by joint affection and deformity, affecting a vast amount of individuals worldwide [1], but RA is more than a symmetrical joint inflammation, as growing evidence supports a higher risk of various autoimmune disorders. Involvement of genetic and environmental factor may provoke the establishment of RA with activation of the immune system [2]. Despite the fact that RA primarily affects joints, in about 40 percent of instances, extra- articular can be discovered [3]. In Egyptian RA instances such as interstitial pulmonary illness, kidney and h ematological diseases [4], various extra-articular manifestations were established.
Auto Immune Thyroid Diseases (AITD) is one of the most prevalent organ -specific autoimmune disorders characterized by endocrine dysfunction and increased existence of TAbs, such as TPOAbs and TgAbs antibodies. For that, for many researches, the RA and AITD were a topic of interest [5].
The two illnesses appear to coexist in each biology, such as HLA-DR B1, CTLA4 and PTPN22 and environmental risk factors such as vaccines and infection, endorsed their similarities [6]. The prevalence of AITD in RA patients analyzed in various research studies ranges across nations from 0.5% in Morocco to 27% in the European country [7]. In Egypt, the connection between hypothyroidism and RA had not been investigated. A better frequency of hypothyroidism or TAbs has been ascertained in RA patients compared with the overall population, and hypothyroidism and TAbs square measure related to RA disease activity [1]. Do RA patients with hypothyroidism have a risk of increasing the severity of RA than patients without? We aim of this study to asses and study the incidence of hypothyroidism in RA patients and its impact on the severity of RA.
Patients and methods
This retrospective cross-sectional study included 1000 adult RA Egyptian patients previously diagnosed as RA patients recruited randomly from patients attending the Out-patient clinics and In-patients of Medicine Departments of Al-Sheikh Zayed Al-Nahian hospital and Azhar Assiut University hospital in the period from March 2018 to August 2019. Group of control counted 200 individuals who are healthy similarly in age and sex to the patient's group.
The requirements for exclusion were as follows:
Patients on drugs known to cause thyroid abnormalities (e.g. interferon-alpha, lithium, etc.).
• Malignancy
• Current infection
• Other rheumatic disorder
• Pregnancy
• Chronic liver disorder or nephropathy
• Diabetic Mellitus
• Surgical removal of thyroid glands
All patients used the criteria for classification of ACR/EULAR RA in 2010 [8].
The RA patients were 850 (85%) females and 150 (15%) males while the control group were 160 (80%) women and 40 (20%) males. All individuals were oriented regarding the method and aim of the research and gives a verbal consent for agreeing to participate in our study. The following were presented to all patients:
• Full history including age, sex, patients' demographic data, duration of RA. Also, the medical files of all cases were checked and current medications were reported
• Anthropometric measurements: Bodyweight in Kg and height in meters.
• Calculating of BMI = BW in kg ÷ BH in m2.
• Thorough general clinical examination: including different body systems, tender joint count, swollen joint count and modified Health Assessment Questionnaire (MHAQ) was used to investigate the functional disabilities encountered the daily living activities. A questionnaire was used consisting of eight issues about dressing that arose from bed, eating (or drinking), walking, self-hygiene, reaching items, handgrip, and outdoor activities. For each object, one of four answers are registered: score 0 (without any trouble), score 1 (with some difficulty), score 2 (with a lot of difficulty) and score 3 (when impossible). The mean of complete results is the score of the patient [9].
• Laboratory investigations: each member's blood samples.
• The Westergren technique has been used to investigate the Erythrocyte Sedimentation Rate (ESR).
• The latex agglutination technique was used to investigate the C-Reactive Protein (CRP).
• The serum Rheumatoid Factor (RF) assay was explored using a latex agglutination map of the Biotec RA factor.
• ELISA investigated the anti-Cyclic Citrullinated Peptide (anti-CCP) assay.
• Total serum thyroxine level (T4) and triiodothyronine level (T3) radioimmunoassay were researched
• Immune-radiometric testing of the Thyroid-Stimulating Hormone (TSH) serum.
Patients split into the following groups, based on the definitions of thyroid dysfunctions shown in Table 1.
| Thyroid dysfunction | TSH level (mIU/L) | FT4 level (μg/dL) |
|---|---|---|
| Euthyroid | 0.45-4.49 (normal) | 4.5-12 (normal) |
| Subclinical hypothyroidism | >4.49 (increased) | 4.5-12 (normal) |
| Subclinical hyperthyroidism | <0.45 (decreased) | 4.5-12 (normal) |
| Overt hypothyroidism | >4.49 (increased) | <4.5 (decreased) |
| Overt hyperthyroidism | <0.45 (decreased) | >12 (increased) |
Table 1. Definitions of Thyroid Dysfunctions according to the FT4 and TSH serum levels.
Group (1): patients with rheumatoid arthritis and hypothyroidism.
Group (2): patients with rheumatoid arthritis without hypothyroidism.
The DAS-28 was assessed:
(DAS-28) = 0.56 × (28 TJC) + 0.28 × (28 SJC) + 0.70 × Ln (ESR) + 0.014 × VAS
The DAS-28 is a validated numerical scale from 0 to 10 indicating current RA (Table 2).
| Remission | DAS-28 | <2.6 |
|---|---|---|
| Low disease activity | DAS-28 | 2.6-3.1 |
| Moderate disease activity | DAS-28 | 3.2-5.1 |
| High disease activity | DAS-28 | >5.1 |
Table 2. Values of DAS-28 relate to clinical status.
All data were collected, tabulated, and statistically analyzed. Analysis of data was carried out by an IBM computer using Statistical Program for Social Science (SPSS) (software and services, North California, USA) version 18 as follows:
• Mean, SD, and number used to description of quantitative variables.
• Number and percentage used to description of qualitative variables.
• The χ2-test was used to compare groups in terms of qualitative variables.
• The Fisher exact test was used instead of χ2- test if one of the compared items (cell on excel) is less than digit 5.
• An unpaired t-test was used to compare two groups in terms of a quantitative variable.
• p value >0.05 non-significant, p value < 0.05 significant and p value <0.01 highly significant.
• Relationships between parameters were analyzed using the Pearson correlation coefficients (r) [10].
Results
Our study includes 1000 adult RA patients (850 female and 150 male) was previously diagnosed as RA patients recruited randomly from patients attending the Out-patient clinics and Inpatients of Medicine Departments of Al-Sheikh Zayed Al-Nahian hospital and Azhar Assiut University hospital in the period from March 2018 to August 2019. Thyroid abnormalities were detected among 280 patients (28%) represented as Hypothyroidism 270 patients (96.42%) (Subclinical Hypothyroidism 210 patients (77.77%), Clinical Hypothyroidism 60 patients (28.57%)) and Hyperthyroidism 10 patients (3.57%) (Subclinical Hyperthyroidism 8 patients (80%) - Clinical Hyperthyroidism 2 patients (20%)), While we couldn’t detect any case with Sick euthyroid Disease. While the RA patients with euthyroid was 720 patients (72%).
Anti-TPO antibodies were present in 56 (5.6%) RA patients, whereas anti-Tg antibodies were present in 42 (4.2%) RA patients compared with 4 (2%) and 2 (1%) at control participants respectively, with highly statistical difference (p<0.01) between both groups Accordingly, AITD was considered positive in 40 (14.81%) patients who showed hypothyroidism together with positive anti-TPO antibodies that was not observed in any of the controls.
Most of the randomly selected patients are rural (88%) and most of them are female (85%). In our study the mean ± SD of age at time of study was 40.75 ± 5, The mean ± SD of duration of disease at time of study was 12.2 ± 5.
In addition, we found that early morning stiffness, swelling joints and tender joints are the commonest manifestations among all studied patients, followed by Dryness of skin. While as regard the incidence of weight gain and joint deformity, swelling and tender joints was founded to be more in RA with Hypothyroidism. According to laboratory result we founded that the mean ± SD of ESR (mm/h) at time of study was 31.75 ± 4, while The mean ± SD of CRP (mg/l) at time of study was 11.7 ± 22 and also Anti CCP was founded positive in 655 patients (65.5%), while RF was founded positive in 585 patients (58.6%).
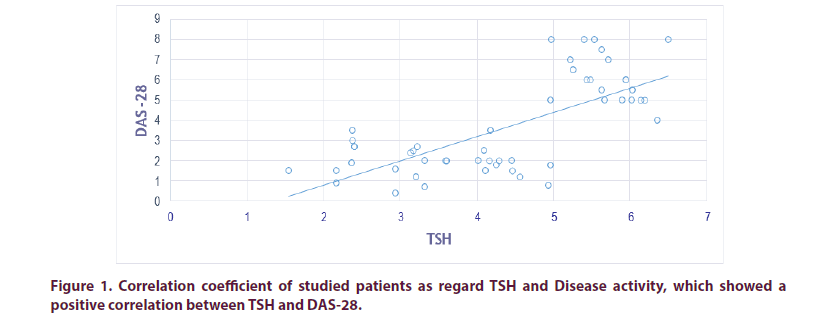
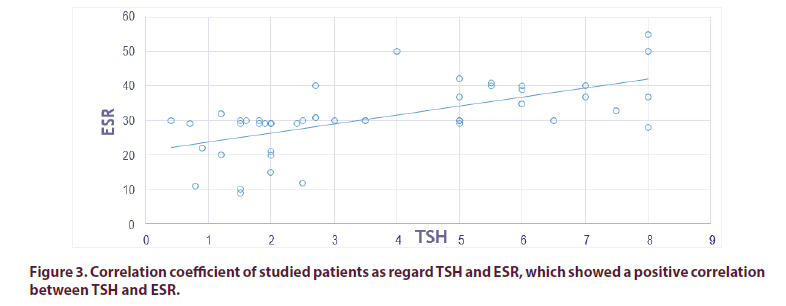
We found positive correlation between TSH and DAS-28 Score (r 0.895, p<0.01), also between TSH and ESR (r 0.7865, p<0.01), which indicate higher disease activity when TSH is high. According to DAS-28 scores: No patient founded in remission while low disease activity was discovered in 351 patients (27 in Group I and 324 in Group II), moderate disease activity was founded in 540 instances (162 in Group I and 378 in Group II), while high disease activity was identified in 72 instances (54 in Group I and 18 in Group II). High activity of the disease is greater in-group I compared to group II. Which showing that patient with Hypothyroidism has higher DAS-28 Score in compare with RA without Hypothyroidism. Anti CCP was not found to have a significant presence in coexisting RA and thyroid dysfunction (Tables 3-11 and Figures 1-3).
| Demographic characteristics | All patients | Group I | Group II | Control | P-value | |||
|---|---|---|---|---|---|---|---|---|
| Age (years) | 40.75 ± 5 | 41 ± 5 | 40.4 ± 5 | 43.7 ± 5 | > 0.05 | |||
| N (%) | N(270) | % | N(720) | % | N (%) | |||
| Sex | Male | 150 (15) | 70 | 25.9 | 80 | 11.11 | 40 (20) | > 0.05 |
| Female | 850 (85) | 200 | 74.1 | 640 | 88.88 | 160 (80) | > 0.05 | |
| Residence | Urban | 120 (12) | 54 | 20 | 61 | 8.47 | 60 (30) | > 0.05 |
| Rural | 880 (88) | 216 | 80 | 659 | 91.52 | 140 (70) | > 0.05 | |
| Weight (Kg) | 91.15 ± 15 | 92.8 ± 15 | 89.5 ± 15 | 85 ± 15 | > 0.05 | |||
| Body mass index (BMI) | 28.9 ± 5 | 28.8 ± 5 | 29.1 ± 5 | 22.1 ± 5 | > 0.05 | |||
| Duration of illness (years) | 12.2 ± 5 | 12.1 ± 5 | 12.3 ± 5 | > 0.05 |
Table 3. Mean ± SD of demographic characteristics of all studied patients versus control.
| Presenting symptoms | Group I | Group II | ||
|---|---|---|---|---|
| N (270) | % | N (720) | % | |
| Swelling joints | 203 | 75.18 | 720 | 100 |
| Tender joints | 229 | 84.81 | 720 | 100 |
| Joint deformity | 162 | 60 | 288 | 40 |
| Early morning stiffness | 270 | 100 | 720 | 100 |
| Weight gain | 189 | 70 | 216 | 30 |
| Dryness of skin | 150 | 55.55 | 324 | 45 |
| Manifestation of hypothyroidism | 60 | 22 | 0 | 0 |
| Palpitations | 81 | 30 | 0 | 0 |
Table 4. Clinical presenting symptoms of all studied patients.
| Item | All patients | Group I | Group II | Control | P value |
|---|---|---|---|---|---|
| HB (mg/dl) | 10.45 ± 2 | 10.2 ± 2 | 10.7 ± 2 | 13.5 ± 2 | > 0.05 |
| MCV (fl) | 79 ± 5.2 | 78.3 ± 5.2 | 79.7 ± 5.2 | 82 ± 5.2 | > 0.05 |
| PLT (_106/cmm) | 324.2 ± 50.6 | 321.7 ±50.6 | 326.7± 50.6 | 327 ± 50.7 | > 0.05 |
| TLC (_103/cmm) | 8.5 ± 2 | 8.9 ± 2 | 8.1 ± 2 | 6.6 ± 2 | > 0.05 |
| ESR (mm/h) | 31.75 ± 4.2 | 25.8 ± 4.2 | 37.7 ± 4.2 | 12.8 ± 4.2 | <0.01 |
| CRP (mg/l) | 11.7 ± 2.2 | 28 ± 4 | 24.9 ± 4 | 4.3 ± 2.2 | > 0.05 |
| Cholesterol (mg/dl) | 213.5 ±25.7 | 9.3 ± 2.2 | 14.3 ± 2.2 | 178 ±25.7 | <0.01 |
| Triglyceride (mg/dl) | 214.15 ± 33.7 | 31.2 ± 4.8 | 35.5 ± 4.8 | 110 ± 10 | <0.01 |
| LDL (mg/dl) | 119.2 ± 20.3 | 221.3 ±25.7 | 205.7± 25.7 | 81 ± 10 | > 0.05 |
| RBS (mg/dl) | 111.5 ± 15 | 229 ± 33.7 | 199.3± 33.7 | 100 ± 15 | > 0.05 |
| TSH (pmol/l) | 4.03 ± 0.5 | 1.92 ± 0.5 | 6.15 ± 0.5 | 2.18 ± 0.5 | <0.01 |
| FT3 (pg/ml) | 3.65 ± 0.7 | 3.5 ± 0.7 | 3.8 ± 0.7 | 3.55 ± 0.7 | > 0.05 |
| FT4 (ng/dl) | 1.59 ± 0.7 | 1.73 ± 0.7 | 1.45 ± 0.7 | 1.54 ± 0.7 | > 0.05 |
| Anti-TPO antibodies (IU/ml) | 93.435± 115.28 | 120.18 ± 122.23 | 68.69 ± 108.33 | 43.16 ± 132.23 | <0.01 |
| Anti-Tg antibodies (IU/ml) | 50.69± 60.36 | 54.19 ± 46.74 | 47.2 ± 73.98 | 24.19 ± 36.74 | <0.01 |
| RF +ve (%) | 585 (58.6%). | 150 (55.55) | 435 (60.41) | 0 | <0.05 |
| Anti-CCP +ve (%) | 655 (65.5%) | 200 (74.07) | 455 (63.19) | 0 | <0.05 |
NS: non-significant (p>0.05); S: significant (p<0.05); HS: Highly significant (p<0.01)
Table 5. Mean ± SD of hematological and rheumatic profiles among all studied patients versus control.
| Item | Group I | Group II | P value | Significance |
|---|---|---|---|---|
| TSH (pmol/l) | 6.15 ± 0.5 | 1.92 ± 0.5 | <0.01 | HS |
| FT3 (pg/ml) | 3.5 ± 0.7 | 3.8 ± 0.7 | > 0.05 | NS |
| FT4 (ng/dl) | 1.73 ± 0.7 | 1.45 ± 0.7 | > 0.05 | NS |
| Anti-TPO antibodies (IU/ml) +ve (%) | 40 (14.8%) | 12 (1.66%) | <0.01 | HS |
| Anti-Tg antibodies (IU/ml) +ve (%) | 32 (11.85%) | 8 (1.11) | <0.01 | HS |
NS: non-significant (p>0.05); S: significant (p<0.05); HS: Highly significant (p<0.01)
Table 6. Mean ± SD of thyroid profile in all studied patients.
| Item | RA patients | Controls | |||
|---|---|---|---|---|---|
| Male (%) | Female (%) | Male (%) | Female (%) | ||
| Total | 150 (15) | 850 (85) | 40 (20) | 160 (80) | |
| Hyperthyroidism | Subclinical Hyperthyroidism | 0 | 2 | 0 | 0 |
| Clinical Hyperthyroidism | 0 | 8 | 0 | 0 | |
| Euthyroidism | 80 | 640 | 40 (20) | 152 (98.5) | |
| Hypothyroidism | Subclinical Hypothyroidism | 60 (22.22) | 150 (55.55) | 0 | 8 (0.5) |
| Clinical Hypothyroidism | 10 (3.7) | 50 (18.51) | 0 | 0 | |
| Sick Euthyroid Disease | 0 | 0 | 0 | 0 | |
Table 7. Prevalence of thyroid dysfunction among studied patients.
| Item | Group I | Group II | P value | Significance | |||
|---|---|---|---|---|---|---|---|
| VAS | 6.95 ± 0.5 | 2.93 ± 0.5 | <0.01 | HS | |||
| TJC | 15.75 ± 0.5 | 3.9 ± 0.5 | <0.01 | HS | |||
| SJC | 9.9 ± 0.5 | 2.86 ± 0.5 | <0.01 | HS | |||
| DAS-28 | 5.7 ± 0.2 | 3.4 ± 0.2 | <0.01 | HS | |||
| N (270) | % | N (720) | % | ||||
| Disease activity | Remission | 0 | 0 | 0 | 0 | <0.01 | HS |
| Low | 27 | 10 | 324 | 45 | <0.01 | HS | |
| Moderate | 162 | 60 | 378 | 52.5 | <0.01 | HS | |
| Sever | 54 | 20 | 18 | 2.5 | <0.01 | HS | |
NS: non-significant (p>0.05); S: significant (p<0.05); HS: Highly significant (p<0.01)
Table 8. Mean ± SD of disease activity among studied patients.
| Item | TSH (pmol/l) | FT3 (pg/ml) | FT4 (ng/dl) | |||
|---|---|---|---|---|---|---|
| R | P value | R | P value | R | P value | |
| Disease duration | 0.2475 | > 0.05 | -0.2745 | > 0.05 | -0.1825 | > 0.05 |
| ESR | 0.7865 | <0.01 | -0.7495 | <0.01 | -0.34 | <0.01 |
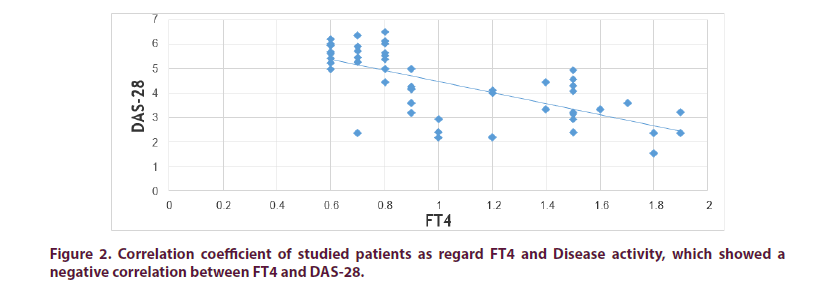
| DAS-28 | 0.895 | <0.01 | -0.824 | <0.01 | -0.4195 | <0.01 |
| BMI (kg/m2) | 0.7605 | <0.01 | -0.769 | <0.01 | -0.3705 | <0.01 |
| Cholesterol (mg/dl) | 0.24 | <0.05 | -0.239 | <0.05 | -0.139 | <0.05 |
| TG (mg/dl) | 0.6615 | <0.05 | -0.6795 | <0.05 | -0.3415 | <0.05 |
| LDL (mg/dl) | 0.203 | > 0.05 | -0.2895 | > 0.05 | -0.161 | > 0.05 |
NS: non-significant (p>0.05); S: significant (p <0.05); HS: Highly significant (p<0.01)
Table 9. Correlations of levels of thyroid-stimulating hormone, free T3 and free T4 with disease duration, activity score (DAS-28), BMI, cholesterol, Lipid profile among rheumatoid arthritis patients.
| TSH (pmol/l) | P value | ||
|---|---|---|---|
| Item | Group I | Group II | |
| R | R | ||
| Disease duration | 0.245 | 0.25 | > 0.05 |
| ESR | 0.791 | 0.782 | <0.01 |
| DAS-28 | 0.901 | 0.889 | <0.01 |
| BMI (kg/m2) | 0.762 | 0.759 | <0.01 |
| Cholesterol (mg/dl) | 0.242 | 0.238 | <0.05 |
| TG (mg/dl) | 0.667 | 0.656 | <0.05 |
| LDL (mg/dl) | 0.208 | 0.198 | > 0.05 |
NS: non-significant (p>0.05); S: significant (p<0.05); HS: Highly significant (p<0.01)
Table 10. Correlations of levels of thyroid stimulating hormone with disease duration, activity score (DAS-28), BMI, cholesterol, Lipid profile among Group1 versus Group 2.
NS: non-significant (p>0.05); S: significant (p<0.05); HS: Highly significant (p<0.01)
Table 11. Correlations of levels of FT4 and FT3 with disease duration, activity score (DAS-28), BMI, cholesterol, Lipid profile among Group1 versus Group 2.
Discussion
Our study includes 1000 adult RA patients (850 female and 150 male) was previously diagnosed as RA patients recruited randomly. Thyroid abnormalities were detected among 280 patients (28%) represented as Hypothyroidism 270 patients (Subclinical Hypothyroidism 210 patients - Clinical Hypothyroidism 60 patients) and Hyperthyroidism 10 patients (Subclinical Hyperthyroidism 8 patients - Clinical Hyperthyroidism 2 patients) While we couldn’t detect any case with Sick euthyroid Disease. While the RA patients with euthyroid stat was 720 patients, this result is in agreement with the study of Saqre et al. [6] who found that 29 (30%) RA patients had evidence of thyroid dysfunction, although another Egyptian study Gheita and Eesa [3] were reported less frequent thyroid dysfunction (8.3%).
We try to exclude other diseases, which can also affect the RA disease activity such as chronic liver or renal diseases and Diabetic patients. Moreover, to prove the relationship between hypothyroidism with RA disease activity.
In our research, we describe TSH-based hypothyroidism as we deemed 4.1 IU/mL to be the upper limit, TSH between 4.1-10 IU/mL plus ordinary FT4 reference range, and symptoms of hypothyroidism to define subclinical hypothyroidism, while TSH to describe clinical hypothyroidism more than 10 IU/mL [11].
We found no statistically significant differences founded between groups studded as regard to Demographic characteristics. The autoimmune disease tends to affect females more than Male with varying frequency according to disease and organ/body system. Elattar et al. [1] found that RA was more prevalent in females than men, with females accounting for 66-86 percent. In our research we discovered that RA and hypothyroidism are more prevalent in women in both groups of our survey randomly chosen patients (76.66 Female: 23.33 Male) AITD was also more prevalent in women than men. These results are in agreement with the study of Mahagna et al. [12] who believe the cause of RA and hypothyroidism is autoimmunity so it is more common in females.
Also, we showed that swelling joints, tender joints, and early morning stiffness are the commonest manifestations among all studied patients, followed by Dryness of skin and weight gain. It also showed that patients with hypothyroidism showed that weight gain and joint deformity are more common than patients with RA without hypothyroidism are. These results are in agreement with Marcucci et al. [13].
Subclinical hypothyroidism was found more female patients with RA than general population. Waldenlind et al. [14] who believe that many causes for hypothyroidism established, the communist one, especially in elder female, is autoimmune thyroiditis (Hashimoto’s thyroiditis). In our study, 200 (74.1%) female RA patients compared with 70 (25.9%) men had hypothyroidism and this also maybe for the autoimmune cause of hypothyroidism. Many genes, including PTPN22, CTLA4, CD40, TSH receptor gene, HLA gene complex, and thyroglobulin gene, discovered to boost the danger of creating AITD. Part of those like PTPN22, CTLA4, CD40, and the complex of HLA genes have a part to play in RA advancement. This mechanism can explain RA and hypothyroidism harmony [6]. The prevalent etiology of both hypothyroidism and RA makes their mixture sound logical and even anticipated, but there remains confusion about the sure manner they evolve. Many studies attempt to clarify this mix; one of them showed that auto-reactive cells that are accountable for the underlying pathology in autoimmune thyroiditis that causes Hashimoto’s thyroiditis could also cause RA. A number of research studies have also explored the co-excitation of hypothyroidism with RA disease activity as environmental variables can accelerate this outcome [15]. A prevalence of AITD in 9.8 percent of their patients with RA research; however, others discovered an elevated incidence of 16 % in their instances [12], in our research the Anti-TPO antibodies were present in 56 (5.6%) RA patients, whereas anti-Tg antibodies were present in 42 (4.2%) RA patients compared with 4 (2%) and 2 (1%) at control participants respectively, with highly statistical difference (p<0.01) between both groups Accordingly, AITD was considered positive in 40 (14.81%) patients who showed hypothyroidism together with positive anti-TPO antibodies that was not observed in any of the controls. These results are almost in agreement with the results of Elattar et al. [1], who found positive anti-TPO and anti-Tg antibodies in 10 and 6% of Egyptian RA patients. However, our data were different from those of other populations, where these antibodies were present in 15.9 and 12.3% of Turkish RA patients [16]. Also, a higher percentage of thyroid antibodies was recorded in Polish RA patients (15 and 12%, respectively) [12] as well as in Colombian RA patients (37.8 and 20.8%) [17]. These variations in the percentage of antithyroid antibodies can be attributed to ethnic and environmental differences of the studied populations. Common etiological factors of RA and hypothyroidism, such as the use of medication to treat RA (as NSAIDs or corticosteroids) leading to thyroid dysfunction, have been explored [18].
For this purpose, the pathogenesis of hypothyroidism in RA patients may have the same pathway and it has been suggested that hypothyroidism is the result of the antithyroid activity of one of the RA produced antibodies [19].
Moreover, one possible response to the foundation of two or more autoimmune disorders in one patient is a genetic predisposition formed by a certain form of HLA, most often HLA-DR [20]. More proof was suggested when anti-TNF-α was used in RA patients and thyroid function was improved [12].
Our results showed that RA and subclinical hypothyroidism are surely linked but, it is difficult to improve retrospectively which disorder anticipate the other, or if in fact, this relationship is a temporally.
Our results showed positive significant correlations between RA activity parameters (ESR and DAS-28) and TSH level indicating that higher levels of TSH are accompanied with high RA disease activity as did the study by Elattar et al. [1] they, in common with our study and Bliddal et al. [21] likewise showed no correlation of hypothyroidism with anti-CCP and positive correlation of RF, CRP; but this result was contradicted by Gheita and Eesa [3].
We believe that as hypothyroidism is not uncommon in RA patients, it is recommended to investigate their thyroid function.
Also, we found negative correlations between serum levels of FT4 and RA activity parameters (ESR and DAS-28), indicating that lower FT4 level is linked to high RA disease activity.
Hypothyroidism was discovered by Marcucci et al. [13] to correlate with the number of swollen joints in RA patients. Where others like Tarhan et al. [18] discovered elevated CRP in RA patients with hypothyroidism, which is agree with our findings.
However, other trials like Gheita and Eesa [3] have not been able to create a connection between hypothyroidism and RA activity parameter and have found that hypothyroidism coexists with RA duration and not severity, although it is incompatible with our outcomes; this could be due to the brief mean duration of our RA (12.2 ± 5 years) compared to theirs.
Symptoms of hypothyroidism should be explored as usual in all RA patients, and more frequent thyroid disease screening should be performed if symptoms of thyroid dysfunction are found. On the other side, due to this elevated incidence of hypothyroidism, the symptoms of hypothyroidism should be systematically and thoroughly researched in all RA patients throughout their follow-up, given this high prevalence of hypothyroidism established by our study. Fatigue, for instance, is a common symptom in RA patients and also hypothyroidism; for that, hypothyroidism screening should be regarded, particularly if other causes of fatigue have been excluded.
However, the impact of treating hypothyroidism on RA disease activity was not tested in our research. Looking ahead, look at the impact of hypothyroidism and other thyroid dysfunctions.
Limitation of the study
• The sample was drawn in small size.
• Impact of treatment of hypothyroidism on disease activity not addressed in our study.
• Correlation of anti-rheumatic agents on disease activity not addressed in our study.
Conclusion
• Thyroid dysfunction is prevalent in rheumatoid arthritis patients.
• Hypothyroidism is the most prevalent disorder in RA patients.
• Hypothyroidism and illness activity are correlated.
Acknowledgments
None.
Patient consent
Obtained.
Ethics approval
The local ethical committee (Al-Azhar University Hospitals -Assiut) approved the study.
Conflict of interest
None.
Funding
This research did not receive any specific grant from funding agencies in the public, commercial, or not-forprofit sectors.
References
- Elattar EA, Younes TB, Mobasher SA. Hypothyroidism in patients with rheumatoid arthritis and its relation to disease activity. Egypt. Rheumatol. Rehabil. 41(2), 58–65 (2014).
- Li Y, Xu J, Gu C et al. Carvacrol suppresses inflammatory responses in rheumatoid arthritis fibroblastâ€Âlike synoviocytes. J. Cell. Biochem. 120(5), 8169–8176 (2019).
- Gheita T and Eesa N. Rheumatology in Egypt back to the future. Rheumatol. Int. 39(1), 1–12 (2019).
- Fadda S, Khairy N, Fayed H et al. Interstitial lung disease in Egyptian patients with rheumatoid arthritis. Frequency, pattern, and correlation with clinical manifestations and anti- citrullinated peptide antibodies level. Egypt. Rheumatol. 40(3),155–160 (2018).
- Tagoe, Sheth, Golub et al. Rheumatic associations of autoimmune thyroid disease: a systematic review. Clin. Rheumatol. 38, 1–9 (2019).
- Saqre I, El-Bahnasawy A, Farag S et al. Autoimmune thyroid disease in Egyptian patients with rheumatoid arthritis. Egypt. Rheumatol. 4, 5–10 (2019).
- Abd-Elhafeez H, El-Meghawry E, Al-Azhary S et al. Frequency of rheumatoid arthritis in patients with autoimmune thyroid disease. a case-control study. Egypt. J. Obesity. Diabetes. Endocrinology. 4(1), 5 (2018).
- Choi H, Kim K, Jin S et al. Decreased Expression of Sphingosine-1-Phosphate Receptor 1 in the Blood Leukocyte of Rheumatoid Arthritis Patients. Immune. Netw. 18(5) (2018).
- Voshaar, Vonkeman, Courvoisier et al. Towards standardized patient reported physical function outcome reporting: linking ten commonly used questionnaires to a common metric. Qual. Life Res. 28(1), 187–197 (2019).
- Kocher Mininder S, David Zurakowski. Clinical epidemiology and biostatistics: a primer for orthopaedic surgeons. J. Bone. Joint. Surg. 86(3), 607–620 (2004).
- Razvi Salman, Robin Peeters, Simon HS et al. Thyroid Hormone Therapy for Subclinical Hypothyroidism. Jama. 321(8), 804 (2019).
- Mahagna H, Caplan A, Watad A et al. Rheumatoid arthritis and thyroid dysfunction: A cross-sectional study and a review of the literature. Best. Pract. Res. Clin. Rheumatol. 32(5), 683–691 (2018).
- Marcucci E, Bartoloni E, Alunno A et al. Extra-articular rheumatoid arthritis. Reumatism. 70(4), 212–224 (2018).
- Waldenlind K, Saevarsdottir S, Bengtsson C et al. Risk of Thyroxine-Treated Autoimmune Thyroid Disease Associated With Disease Onset in Patients With Rheumatoid Arthritis. JAMA. Netw. Open. 1(6), 183567 (2018).
- Rayman M. Multiple nutritional factors and thyroid disease, with particular reference to autoimmune thyroid disease. Proceedings of the Nutrition Society. 78(1), 34–44 (2019).
- Fröhlich E, Wahl R. Thyroid autoimmunity: role of anti-thyroid antibodies in thyroid and extra- thyroidal diseases. Front. Immunol. 8, 521 (2017).
- Molano-González N, Rojas M, Monsalve DM et al. Cluster analysis of autoimmune rheumatic diseases based on autoantibodies. New insights for polyautoimmunity. J. Autoimmun. 98, 24–32 (2019).
- Tarhan F, Oruk G, Niflioglu O et al. Thyroid involvement in ankylosing spondylitis and relationship of thyroid dysfunction with anti-TNFa treatment. Rheumatol. Int. 33(4), 853–857 (2013).
- McInnes I, Schett G. Pathogenetic insights from the treatment of rheumatoid arthritis. Lancet. 389(10086), 2328–2337 (2017).
- Tagoe C, Sheth T, Golub E et al. Rheumatic associations of autoimmune thyroid disease: a systematic review. Clin. Rheumatol. 38(7), 1801–1809 (2019).
- Bliddal S, Borresen S, Feldt-Rasmussen U. Thyroid autoimmunity and function after treatment with biological ant rheumatic agents in rheumatoid arthritis. Front. Endocrinol. 8, 179 (2017).