Research Article - Imaging in Medicine (2018) Volume 10, Issue 2
Intracranial Venous Sinuses Characteristics Prior To And Following Resection Of Brain Tumors
Amit Azriel1*, Israel Melamed1, Yuval Sufaro1, Avi Cohen1, Mony Benifla2, Nitsan Mendelson3 & Ilan Shelef41Department of Neurosurgery, Soroka University Medical Center and Faculty of Health Sciences, Ben-Gurion University of the Negev, Beer Sheva, Israel
2Department of Pediatric Neurosurgery, Rambam Health Care Campus, Haifa, Israel
3Clinical Research Unit, Soroka University Medical Center, Beer-Sheva, Israel
4Department of Radiology, Soroka University Medical Center and Faculty of Health Sciences, Ben-Gurion University of the Negev, Beer Sheva, Israel
- Corresponding Author:
- Amit Azriel
Department of Neurosurgery
Soroka University Medical Center and Faculty of Health Sciences
Ben-Gurion University of the Negev, Beer Sheva, Israel
E-mail: azriela@post.bgu.ac.il
Abstract
Background: Intracranial pathologies often cause elevated intracranial pressure (ICP), which presence may be of great significance regarding the chosen treatment option as well as its timing. Radiography is routinely used to support the clinical diagnosis and to establish the etiology for the elevated ICP. The relation between increased ICP and cerebral venous sinuses dimensions has been previously demonstrated, mainly among patients diagnosed with idiopathic increased ICP. The relation between brain tumors and venous sinuses dimensions has not been established so far. The aim of this study was to investigate the relation between the presence of brain tumors and the characteristics of the large cerebral venous sinuses.
Methods: A single center retrospective study was conducted. Patients diagnosed with brain tumors who were surgically treated at our medical center were enrolled. MRI based cerebral venous sinuses measurements were performed, prior to the surgical resection and following it.
Results: The research group included 17 patients, primarily women (64%), aged 16-81 years. Brain pathologies included both benign and malignant tumors. The main cerebral venous sinuses were measured at 6 different locations before and after the resection procedure. Among all 6 measuring points there was a significant size increase following the operation. There was no significant difference in the results when addressing different types of tumors or anatomical location.
Conclusion: This study presents an additional indirect method of establishing the existence of increased ICP, and emphasizes the role of cerebral venous sinuses in ICP control. Using this method may assist in clinical and radiological evaluation of patients diagnosed with brain tumors.
Keywords
intracranial hypertension ▪ ICP ▪ brain tumors ▪ cerebral venous sinuses ▪ combined conduit score
Background
The cerebral venous sinuses system is complex and diverse, with great degree of anatomical variability. This system allows drainage of blood from the brain tissue, meninges and skull, as well as delivery of blood to the internal jugular veins [1]. Over the years, several radiological techniques have been developed to demonstrate the cerebral venous sinuses system. Imaging of the intracranial venous sinuses using magnetic intracranial venous imaging technique (MRV) technique assists with improving the anatomical and functional understanding of the venous sinuses [2-4]. Nowadays, MRV imaging studies are carried out for few different indications: MRV imaging may assist with the identification of possible causes to the Idiopathic Intracranial Hypertension (IIH) syndrome [5,6], with establishing the diagnosis of venous sinus thrombosis [7], as well as with pre-operative assessment of the proximity and invasion of brain tumor into the large intracranial venous sinuses [8].
Increase of intracranial pressure may be caused by several mechanisms. Idiopathic Intracranial Hypertension, and more specifically the Pseutotumor Cerebri (PTC) syndrome, has been investigated since late 19th century. During the last two decades there is increasing attention to those two diagnoses, in which the elevated intracranial pressure is referred to obstruction in brain venous blood drainage [9-12]. It has been previously shown that the dimensions of the large brain venous sinuses increase following lumbar puncture and drainage of cerebral-spinal fluid (CSF), when performed for patients diagnosed with PTC syndrome [13]. Moreover, the dimensions of the main cerebral venous sinuses are usually larger among patients diagnosed with intracranial hypotension [14].
Brain tumors may cause elevated intracranial pressure mainly due to the additional volume to the skull, as well as the vasogenic brain edema they might induce [15,16]. However, the effect of brain tumors related to elevated intracranial hypertension on the intracranial venous sinuses system (other than direct invasion) has not been properly described so far.
Patients and Methods
■ Patients
To examine the influence resection of brain tumors has on the dimensions of large intracranial venous sinuses; we reviewed brain imaging studies of patients diagnosed with different brain tumors who were surgically treated at the Soroka University Medical Center between the years 2013-2015. Inclusion criteria were age 16 years and older, new diagnosis of brain tumour with significant mass effect and reduction in mass effect following gross total surgical resection. We excluded cases with brain tumors directly invading the large intracranial venous sinuses, as well as cases in which the resection of the brain tumors included direct manipulations on the adjacent large intracranial venous sinuses. We also excluded cases of relatively small brain tumors, tumors that did not induce significant brain edema or mass effect and cases in which the resection of the brain tumors did not achieve an obvious decline of the intracranial mass effect.
■ Imaging
The data for this research was collected from TI weighted images (T1WI) after the injection of Gadolinium, in protocol T1 3D TFE SENSE performed in Phillips MRI scanner. The MRI scans were performed at the Imaging Institute of the Soroka University Medical Center.
■ Data analysis and statistics
For each patient measurements of the diameter of the large intracranial venous sinuses were performed prior to surgical resection of the brain tumor and following it. Measurements were performed at identical locations for all patients: right and left transverse sinuses in coronal sections, right and left sigmoid sinuses in coronal section, distal portion of the superior sagittal sinus in coronal section and superior sagittal sinus in axial section. All measurements were performed by a single examiner, using the PACS software.
Descriptive statistics of patient characteristics includes data summaries of main continuous variables in the form of medians and interquartile range (IQR), and categorical data is shown as counts and percentages. The preferred method of analysis for continuous variables utilized non-parametric procedures since parametric assumptions could not be satisfied. Coronal venous sinuses cross sections prior to and following resection of brain tumors were compared using Wilcoxon Signed-ranks test for related continuous variables. Change in size of coronal venous sinuses after brain tumor resection of patients with meningioma compared to other pathologies and change in venous sinuses size compared between supratentorial and infra-tentorial tumor resection were analyzed using Mann-Whitney test. All statistical tests were performed at α=0.05 (2-sided). The data were analyzed using IBM SPSS Statistics software (version 22).
Results
The examined group included 17 patients with different brain tumors, operated in our medical center. The age range was 16-81 years, with female majority (n=11, 64%). The tumor volumes ranged from 2.9 to 83.7 ml. Total skull volumes were also estimated for each patient. 8 patients (47%) were diagnosed with benign brain tumors (meningioma), 5 patients (29.4%) had brain metastases and 4 patients (23.5%) were diagnosed with primary brain tumors. Patient’s details are described in TABLE 1.
| Characteristics | Patients (n=17) |
|---|---|
| Age, years Median Range |
56 16-81 |
| Gender, n (%) Female |
11 (64.7%) |
| Tumor location, n (%) Right hemisphere Left hemisphere Bilateral Posterior fossa Left cerebellar+left frontal Pineal region |
7 (41.3%) 4 (23.5%) 1 (5.9%) 3 (17.7%) 1 (5.9%) 1 (5.9%) |
| Tumor volume (cc) Median Range |
20.4 2.9-83.7 |
| Skull volume (cc) Median Range |
1774 1052-2417 |
| Pathology, n (%) Dysgerminoma GBM Medulloblastoma Meningioma WHO-II Meningioma WHO-1 Metastatic tumors |
1 (5.6%) 2 (11.8%) 1 (5.9%) 2 (11.8%) 6 (35.3%) 5 (29.4%) |
| Drainage dominancy, n (%) Right |
12 (70.6%) |
Table 1: Clinical characteristics of study participants.
Following surgical resection of the brain tumors, significant increase in diameters of the large cerebral venous sinuses was noted in all the measured points (TABLE 2).
| Pre-operation (cc) | Post-operation (cc) | Wilcoxon | ||||
|---|---|---|---|---|---|---|
| Median | Quartile | Median | Quartile | Z | p | |
| Right transverse | 3.23 | 2.60-4.89 | 5.03 | 3.21-5.60 | -3.62 | <0.001 |
| Left transverse | 2.96 | 1.98-3.96 | 4.01 | 2.55-4.59 | -3.52 | <0.001 |
| Right sigmoid | 3.82 | 2.71-4.20 | 4.22 | 3.22-4.95 | -3.24 | 0.001 |
| Left sigmoid | 3.48 | 2.56-4.01 | 3.64 | 3.22-4.28 | -3.47 | 0.001 |
| Distal superior sagittal | 5.08 | 4.60-6.75 | 5.69 | 5.14-6.93 | -3.62 | <0.001 |
| Axial superior sagittal | 4.51 | 3.66-5.44 | 5.43 | 4.13-5.98 | -3.51 | <0.001 |
Table 2: Size comparison of six coronal venous sinuses before and after brain tumor resection.
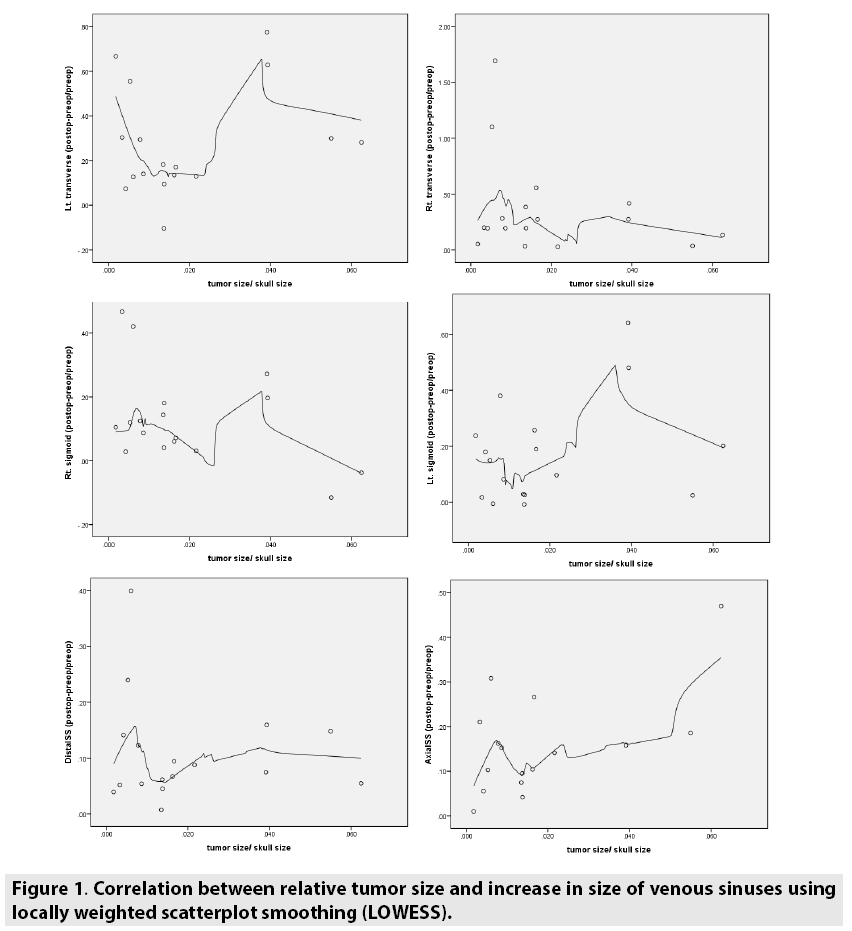
We compared the effect of surgical resection of brain tumors on the large cerebral venous sinuses dimensions between patients diagnosed with benign brain tumors (n=8) and patients diagnosed with malignant brain tumors (n=9). Among patients diagnosed with malignant brain tumors a higher increase in large cerebral venous sinuses diameters following tumor resection was noticed in most of the measured sinuses. A similar comparison was performed between patients with tumors located above the tentorium cerebelli (n=12) and patients with tumors in the posterior fossa (n=3). There was neither significant change nor trend in change in the large venous sinuses dimensions between these two groups. Standardization attempt for the results of this study was done by calculating the ratio between tumor volume and skull volume for each patient. Changes in dimensions of each cerebral venous sinus were calculated with this value, using Locally Weighted Scatterplot Smoothing (LOWESS) method (FIGURE 1).
Figure 1: Correlation between relative tumor size and increase in size of venous sinuses using locally weighted scatterplot smoothing (LOWESS).
Discussion
Brain tumors might cause an elevated ICP in several mechanisms other than the direct increase in pressure caused by the added mass to the skull volume. Some brain tumors induce vasogenic brain edema in the surrounding brain tissue, thus donating to the elevation in ICP [16,17]. A significant brain edema characterizes mainly rapidly growing brain tumors (e.g. metastases and primary brain tumors), but can also be caused in cases of slowly growing benign brain tumors like meningiomas [18]. Another mechanism in which brain tumors can cause elevation in ICP is interference in the natural CSF drainage resulting in obstructive hydrocephalus [19,20]. This mechanism characterizes mainly posterior fossa tumors obstructing the aqueduct of Sylvius, as well as the outlet of the fourth ventricle. The large cerebral venous sinuses also play an important role in maintaining normal ICP. Unlike the strong muscular arterial walls, those of the venous sinuses are susceptible to dilatation and compression [21]. Interfering with blood drainage from the brain might result in increased ICP, either by external compression, direct invasion of the tumor to the nearby large venous sinuses, or due to the hypercoagulable state induced by the tumor's presence causing formation of obstructing blood clots inside the venous sinuses [22].
Several imaging techniques exist in order to demonstrate the presence of elevated intracranial pressure. These non-invasive methods rely on indirect signs suggesting elevated ICP, and include typical characteristics in computed tomography (CT), magnetic resonance imaging (MRI), ophthalmological ultrasound, transcranial Doppler, direct fundoscopy and optic nerves electrical potentials measurements [23]. During the last few years, several studies described narrowing of the large cerebral venous sinuses among patients suffering from increased ICP, mainly following the diagnosis of Idiopathic Intracranial Hypertension (IIH). The purpose of our study was to examine the size of the large cerebral venous sinuses among patients diagnosed with brain tumors likely causing elevated ICP, whether by the actual size of the tumor, by the vasogenic brain edema it induced, or by the enlargement of brain ventricles related to the specific location of the tumor. The population evaluated in this study had more diverse characterizations in comparison with other studies among patients diagnosed with IIH, a diagnosis commonly made among young women. A significant increase was noticed in the dimensions off all examined venous sinuses following surgical resection of the brain tumors, a finding that matches previously described results for different populations and different pathologies.
According to the cerebral autoregulation theory, elevated intracranial volume can be compensated until a certain limit is reached [24]. Presumably, Rapid addition of significant mass to the skull volume might cause the breakage of the cerebral autoregulation earlier than slower addition of the same mass. However, the results of this study failed to show more than a general trend in dimensions change of the cerebral venous sinuses following surgical resection of malignant brain tumors in comparison to benign tumors. A larger sample size is probably needed in order to achieve more decisive results. Nonetheless, it is possible that at the time of diagnosis the benign brain tumors were large enough to result in significant increased intracranial pressure and breakage of cerebral autoregulation, despite their slow growth rate.
When we examined the change in sinuses dimensions following surgical resection of tumors located over the tentorium cerebelli in comparison to infra-tentorial tumors, no significant difference was noted. Presumably, many of the infra-tentorial tumors cause an indirect enlargement of the brain ventricles (i.e. obstructive hydrocephalus) which results in a significant elevation of pressure in the supratentorial compartment as well, thus eliminating the anatomical difference in terms of the source for elevated pressure between tumors located over the tentorium cerebelli and under it.
In attempting to create standardization, a relative parameter was calculated by dividing the tumor's volume with the volume of the skull for each patient. When examining the relation between this calculated value and the change in dimensions of the cerebral venous sinuses, a relatively stable trend is noted in most cases. This trend may indicate the existence of a constant relation between the standardized volume of the tumor and the expected change in the dimensions of venous sinus following its resection, but clearly similar analysis should be performed again for a larger sample size.
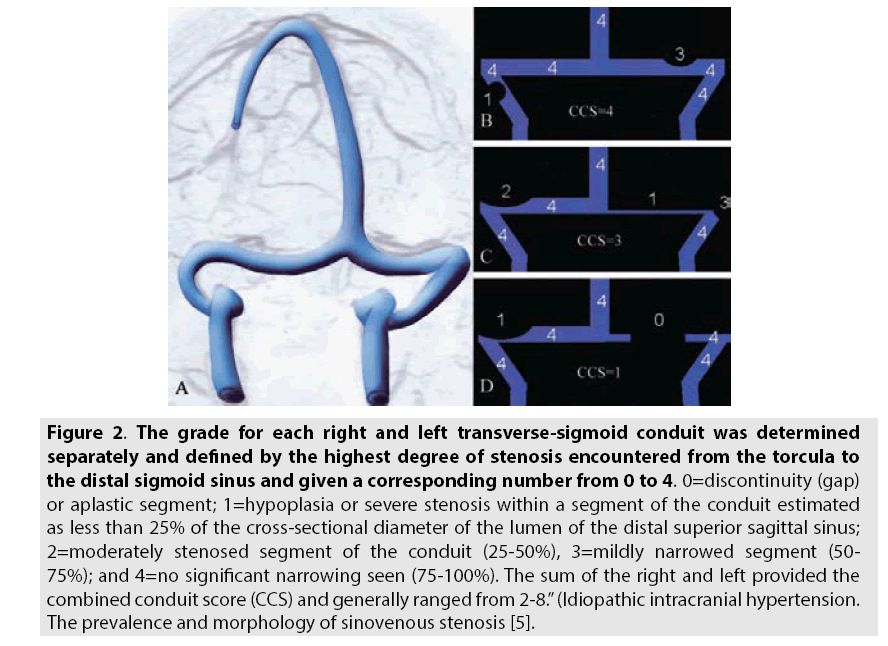
In estimating the degree of brain venous sinuses narrowing among patients diagnosed with IIH, Farb et al. examined the dimensions of the sinuses among IIH patients in comparison to healthy population [5]. In order to compare these two groups a unique grading system was used (Combined Conduit Score, CCS(, based on the ratio between large brain venous sinuses as were demonstrated in brain MRI scans (FIGURE 2). Venous sinuses constriction was demonstrated among 90% of patients with IIH, compared to 7% in healthy population. The researchers concluded that this imaging tool can be utilized as a diagnostic tool for IIH syndrome. Horev et al. [13] quantified the change in brain venous sinuses dimensions following lumbar puncture among 13 patients diagnosed with IIH. Significant increase was noted in the dimensions of all brain venous sinuses examined. The data for patients in our study was also analyzed using Farb's scoring system. Significant increase in CCS was noted following resection of the brain tumors.
Figure 2: The grade for each right and left transverse-sigmoid conduit was determined separately and defined by the highest degree of stenosis encountered from the torcula to the distal sigmoid sinus and given a corresponding number from 0 to 4. 0=discontinuity (gap) or aplastic segment; 1=hypoplasia or severe stenosis within a segment of the conduit estimated as less than 25% of the cross-sectional diameter of the lumen of the distal superior sagittal sinus; 2=moderately stenosed segment of the conduit (25-50%), 3=mildly narrowed segment (50- 75%); and 4=no significant narrowing seen (75-100%). The sum of the right and left provided the combined conduit score (CCS) and generally ranged from 2-8.” (Idiopathic intracranial hypertension. The prevalence and morphology of sinovenous stenosis [5].
Possible explanation for the differences in cerebral venous sinuses characteristics following resection of brain tumors may be related to the changes in direct pressure on the sinuses walls, allowing them to expand following the surgical reduction in ICP. Furthermore, it can be argued that the expansion of the large cerebral venous sinuses may itself lead to more efficient blood drainage from the brain, hence to a secondary reduction in ICP.
The results of this study and its methodological design have several limitations. Intentional selection bias exists, since the study included patients diagnosed with large brain tumors causing obvious mass effect on the surrounding brain tissue. Furthermore, the study group is clearly not large enough for significant conclusions. However, since the aim of this study was to evaluate the feasibility of an imaging tool designed to identify the presence of elevated intracranial pressure, the study population included cases where this condition was likely to exist in advance. The results of this study allow a more thorough examination of cases where the probability for an elevated intracranial pressure is not necessarily high. Moreover, an effort should be made to determine the threshold for using this tool, based on the tumor's size, nature and effect on the surrounding brain tissue. Second, the dimensions of the large brain venous sinuses were measured by a single investigator in a manual technique, and therefore might reduce the reliability of this study's results. However, this measuring technique (and specifically the CCS index) was already validated in Farb's research. Thus, it is unlikely to assume that the general trend of this study's results will change significantly if the dimensions of the large brain venous sinuses would be measured using independent automatic methods. Obviously, this kind of methods should be developed in order to achieve more precise analysis of larger population’s in future similar studies.
Conclusion
This study presents another indirect radiological tool for identification of elevated ICP. The use of this tool may assist with the clinical and radiological surveillance of patients undergoing surgical resection of brain tumors, prior to the surgical intervention and following it. In order to strengthen the reliability of this study's results and the imaging tool it presents, there is an obvious need to expand its scope and to collect data from a larger cohort of patients diagnosed with brain tumors or with intracranial hypertension.
Funding
No funding was received for this research.
Conflict of Interest
All authors certify that they have no affiliations with or involvement in any organization or entity with any financial interest (such as honoraria; educational grants; participation in speaker's bureaus; membership, employment, consultancies, stock ownership or other equity interest; and expert testimony or patentlicensing arrangements) or non-financial interest (such as personal or professional relationships, affiliations, knowledge or beliefs) in the subject matter or materials discussed in this manuscript.
Ethical Approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the Soroka University Medical Center research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards. For this type of study formal consent is not required.
References
- Cure JK, Van TP, Smith MT. Normal and variant anatomy of the dural venous sinuses. Semin. Ultrasound. CT. MR. 15, 499-519 (1994).
- Ayanzen RH, Bird CR, Keller PJ et al. Cerebral MR venography: Normal anatomy and potential diagnostic pitfalls. Am. J. Neuroradiol. 21, 74-78 (2000).
- Agid R, Shelef I, Scott JN et al. Imaging of the intracranial venous system. Neurologist. 14, 12-22 (2008).
- Liauw L, Van Buchem MA, Spilt A. MR angiography of the intracranial venous system. Radiology. 214, 678-682 (2000).
- Farb RI, Vanek I, Scott JN et al. Idiopathic intracranial hypertension. The prevalence and morphology of sinovenous stenosis. Neurol. 60, 1418-1424 (2003).
- Widjaja E, Griffiths PD. Intracranial MR Venography in children: Normal anatomy and variations. Am. J. Neuroradiol. 25, 1557-1562 (2004).
- Meckel S, Reisinger C, Bremerich J et al. Cerebral venous thrombosis: diagnostic accuracy of combined, dynamic and static, contrast-enhanced 4D MR venography. Am. J. Neuroradiol. 31, 527-535 (2010).
- Bozzao A, Finocchi V, Romano A et al. Role of contrast-enhanced MR venography in the preoperative evaluation of parasagittal meningiomas. Eur. Radiol. 15, 1790-1796 (2005).
- Degnan AJ, Levy LM. Pseudotumor cerebri: Brief review of clinical syndrome and imaging findings. Am. J. Neuroradiol. 32, 1986-1993 (2011).
- Higgins JNP, Gillard JH, Owler BK et al. MR venography in idiopathic intracranial hypertension: Unappreciated and misunderstood. J. Neurol. Neurosurg. Psychiatry. 75, 621-625 (2004).
- Johnston I, Kollar C, Dunkley S et al. Cranial venous outflow obstruction in the pseudotumour syndrome: Incidence, nature and relevance. J. Clin. Neurosci. 9, 273-278 (2002).
- Spennato P, Ruggiero C, Parlato RS et al. Pseudotumor cerebri. Child. Nerv. Syst. 27, 215-235 (2011).
- Horev A, Hallevy H, Plakht Y et al. Changes in cerebral venous sinuses diameter after lumbar puncture in idiopathic intracranial hypertension: A prospective MRI study. J. Neuroimaging.20, 1-4 (2012).
- Farb RI, Forghani R, Lee SK et al. The venous distension sign: A diagnostic sign of intracranial hypotension at MR imaging of the brain. Am. J. Neuroradiol. 28, 1489-93 (2007).
- Klatzo I. Evolution of brain edema concepts. Acta. Neurochir. Suppl. 60, 3-6 (1994).
- Papadopoulos MP, Saadoun S, Binder DK et al. Molecular mechanisms of brain tumor edema. Neurosci.129, 1011-1020 (2004).
- Murayi R, Chittiboina P. Glucocorticoids in the management of peritumoral brain edema: A review of molecular mechanisms. Child. Nerv. Syst. 32, 2293-2302 (2016).
- Hou J, Kshettry VR, Selman WR et al. Peritumoral brain edema in intracranial meningiomas: The emergence of vascular endothelial growth factor-directed therapy. Neurosurg. Focus. 35, 2 (2013).
- Lin CT, Riva-Cambrin JK. Management of posterior fossa tumors and hydrocephalus in children: A review. Child. Nerv. Syst. 31, 1781-1789 (2015).
- Raimondi AJ, Tomita T. Hydrocephalus and infratentorial tumors. J. Neurosurg. 55, 174-182 (1981).
- Wilson MH. Monro K. The dynamic vascular and venous pathophysiological components of intracranial pressure. J. Cereb. Blood Flow. Metab. 36, 1338-1350 (2016).
- Schiff D, Lee EQ, Nayak L et al. Medical management of brain tumors and the sequelae of treatment. Neuro. Oncol 17, 488-504 (2015).
- Xu W, Gerety P, Aleman T et al. Noninvasive methods of detecting increased intracranial pressure. Child. Nerv. Syst. 32, 1371-1386 (2016).
- Rangel CL, Gasco J, Nauta HJW et al Cerebral pressure autoregulation in traumatic brain injury. Neurosurg. Focus. 25, 7 (2008).