Research Article - Imaging in Medicine (2022) Volume 14, Issue 6
Aortic Arch Calcification and Novel Markers of Subclinical Atherosclerosis on Lung CT: Methodology and Reproducibility in the COPD Gene Study
Matthew J Budoff*, Ilana S Golub, Suvasini Lakshmanan, Suraj Dahal, Stephanie Kristo, Lucia Schroeder, Orly Termeie, Venkat Manubolu, Luay Hussein, Dhiran Verghese, Ahmed M Shafter, Richard Casaburi, Sion K Roy
Harbor-UCLA Medical Center Lundquist Institute, Torrance, CA, USAy
- *Corresponding Author:
- Matthew J Budoff
Harbor-UCLA Medical Center Lundquist Institute, Torrance, CA, USAy
E-mail: mbudoff@lundquist.org
Abstract
Background: COPD, smoking, and risk for CVD are closely associated. CAC offers a well-established assessment of CVD risk and subclinical atherosclerosis on both ECG-gated/cardiac and non-gated/lung CT. However, the predictive value of CAC for stroke is limited. Frequently detected on ungated lung CT scans due to large field view, aortic arch calcium (AAC) can serve as a marker of subclinical atherosclerotic burden. We hypothesized that AAC is more proximal to the carotids and may better predict stroke. This unmeasured marker may be an independent and/or incremental predictor of both stroke and atherosclerotic cardiovascular disease (ASCVD) but the prognostic value of AAC on adverse cardiovascular events, remains to be established. Our study will evaluate novel cardiac screening metrics of subclinical atherosclerosis, by measuring AAC measurements obtained with ungated Lung CT scans in a large cohort study.
Methods: We will evaluate 10,300 ungated lung CT scans obtained in the COPD GENE cohort study, a longitudinal study of current and former smokers aged 45 to 80 years, sponsored by the National Institutes of Health. The following measures were summed to yield total Arch Calcium Score and total Arch Volume: aortic valve and root; south and north aortic arch segment; brachiocephalic, common carotid and subclavian artery; ascending and descending aorta. We also report the inter-reader variability of this measure, by randomly selecting 90 ungated lung CT scans from this cohort, and Bland-Altman plot analysis was used to test the reproducibility of this measure.
Results: We present here the methods used to determine multiple metrics of calcification not previously reported in any epidemiologic cohort study. We also demonstrate that inter-reader reproducibility was excellent for both total Arch Calcium Score at 98% and for total Arch Volume at 96%.
Conclusion: This metric may prove to be an important measure of ASCVD risk and easily obtainable on every non-contrast lung CT performed. Importantly, high rates of inter-observer reproducibility at ungated lung CT attest to the proposed arch calcium measurement methods. This introduces a new method with the potential of ASCVD risk stratification among patients undergoing lung CT evaluation, without requiring additional scanning, radiation or patient burden. This method can help measure AAC on lung CT, allowing recognition of CV risk and enabling targeted preventive strategies for CVD.
Keywords
Computed tomography • Atherosclerotic cardiovascular disease • MESA • Coronary artery calcium scoring • Extracoronary calcification
Background
The association between smoking, chronic obstructive pulmonary disease (COPD), and risk for cardiovascular disease and adverse outcomes, is significant. Best viewed as a multisystem inflammatory disorder, COPD extends well beyond the respiratory tract [1]. COPD and coronary artery disease (CAD) share common risk factors and pathophysiology, and COPD is closely linked with accelerated atherosclerosis and cardiovascular disease (CVD) [1]. Although CVD is a major cause of mortality in COPD patients, it is often subclinical and asymptomatic in COPD patients, whose symptoms are attributed to underlying lung disease [1-3]. Enhancing early detection of significant atherosclerosis and optimizing early interventions for CVD prevention is therefore critical in populations with smoking history. One method for adjudicating cardiovascular risk is coronary artery calcium (CAC) scoring, a direct measure of atherosclerotic plaque burden [4,5].
Calcium burden has extensive validation for predicting both clinical events and mortality in patients with COPD. In addition to being the primary cause of COPD and lung cancer, smoking is a significant risk factor for heart disease and increased atherosclerotic burden [6]. In fact, smokers are more likely to die of CVD than lung cancer. The National Lung Screening Trial indicated that lung cancer accounted for only 24.1% of deaths [6,7]. Though ungated CT is typically used for assessment of lung cancer and COPD, it can be extended to evaluate CAC and other metrics for advanced atherosclerosis phenotyping (aortic arch calcium, calcium location and density) [6,8]. Thus the COPDGene, a longitudinal, multicohort study, permits a unique opportunity. Within a well-phenotyped COPD population of current and former smokers, this cohort allows for establishing prevalence and prognostic significance of CAC and other imaging markers on lung CT [5]. Aortic valve and arch calcification are almost invariably atherosclerotic, and they are readily visualized on non-gated CT due larger field of view compared to gated scans. These segments represent potential biomarkers for optimizing ASCVD risk via early pharmacological interventions [9,10].
Though aortic atherosclerotic plaque greatly affects stroke risk, it is often excluded from traditional prediction tools [11]. Severe aortic plaque (prevalence of 14%-21%) parallels other motivating agents of embolic stroke: carotid artery disease (10%-13%) and atrial fibrillation (18% - 30%), respectively [12,13]. Proximal aortic plaque increases risk of stroke and peripheral embolization [11]. It serves as a robust marker of ASCVD, and a significant cause of iatrogenic stroke and peripheral emboli. In this regard, considering AAC’s mechanistic implications is key; aortic atherosclerosis may greatly influence future acquisition strategies and clinical reporting tools [11]. This study therefore creates a key platform for those to follow in the detailed assessment of AAC as an ASCVD risk predictor, and for the first time evaluates reproducibility of novel arch segmenting methods.
The novelty of this study also stems from its examination of cardiac calcium reproducibility on lung CT scans. The utilization of routine ungated studies is key to help improve cardiovascular risk assessment. Millions of ungated CT scans are acquired yearly for lung screening among vulnerable smoking populations, and coronary data is not routinely reported. If substantiated on ungated lung CT, ASCVD stratification would require no additional scanning. Clinically assimilating coronary and aortic into all noncontrast chest CT reports may advance early diagnosis, treatment, and patient counseling for CAD and atherosclerosis. It is essential to validate evidence about calcium
Materials and Methods
■ Study Population
Data in this study was gathered from COPDGene, the Genetic Epidemiology Study of Chronic Obstructive Pulmonary Disease. COPDGene is a large, observational and multicenter cohort study with current and former smokers aged 45 to 80 years. COPDGene encompasses a diverse sample of over 10,000 men and women with a >10 packyear smoking history, both with and without COPD across the GOLD (Global Initiative for Chronic Obstructive Lung Disease) stages. With the purpose of investigating prevalence, correlates, and progression of COPD, this prospective cohort was curated as a populationbased sample powered to assess the genetic factors associated with COPD and characterize chest CT phenotypes in COPD subjects. The cohort included both Non-Hispanic white and African- American subjects. Inspiratory and expiratory chest CT scans were obtained on all participants. All individuals gave informed consent, and the study protocol was approved by the review board at each institution in accordance with the Health Insurance Portability and Accountability Act. The original COPDGene study design and methods have been previously published [14].
A subset of the COPDGene population was assessed to determine reproducibility of the technique used for aortic arch calcification measurement and segmentation. In total, 90 participants were used in analysis.
■ Image Analysis
Thin section, helical CT scans were performed in all participants. The scans were obtained with an imaging protocol with collimation, 0-5mm; tube voltage, 120kV; tube current 200mAs; gantry rotation time of 0.5s; and pitch, 1.1. The images were reconstructed with a standard kernel with a slice thickness of 0.75 mm and a reconstruction interval of 0.5 mm, as previously reported [14]. Axial images of the thorax were acquired in the supine position. The present ancillary study will analyze images obtained during the inspiration phase.
All studies were analyzed in blinded fashion by trained technologists at the CT Reading Center at the Lundquist Institute of Harbor-UCLA Medical Center (formerly the Los Angeles Biomedical Research Institute).
■ Aortic Arch Calcium Scoring
Calcification on CT images were measured using Philips HeartBeat CS software (Philips Company, Amsterdam). The following measures of calcium were obtained: area, volume (area x CT slice thickness), density (both mean and maximum attenuation of plaque in Hounsfield Units [HU]), density factor (ordinal score corresponding to the maximum plaque attenuation [1=130-199 HU; 2=200-299 HU; 3=300-399 HU; 4=≥400 HU]), and Agatston score (area x density factor) [15,16]. Any structures with complete absence of calcium were assigned scores of zero. Note that in structures with no calcium present, calcium density was undefined.
All CT studies were scored for arch calcium burden in the following segmentations: aortic valve and root; south and north aortic arch segment; brachiocephalic, common carotid and subclavian artery; ascending and descending aorta. These measures were also summed to yield total Arch Calcium Score and total Arch Volume. Anatomical definitions used to segment these aortic arch regions are described below.
■ Aortic Valve and Wall Calcification (AVC & AWC)
AVC and AWC are scored following methods previously described for measurement of coronary and aortic calcification [5,17]. Calcified regions which extend from the aortic root into the aortic lumen are categorized as AVC, as described previously [18]. AWC, in contrast, is defined as the sum of calcification present along the circumference of the aortic root.
Both aortic valve and wall calcification are scored within the same slice levels. AVC and AWC are scored starting from the slice level of the left main coronary artery (LMCA) and proceeding inferiorly to the level at which the right coronary artery (RCA) emerges from the right coronary cusp.
■ Aortic Arch Calcification (AAC)
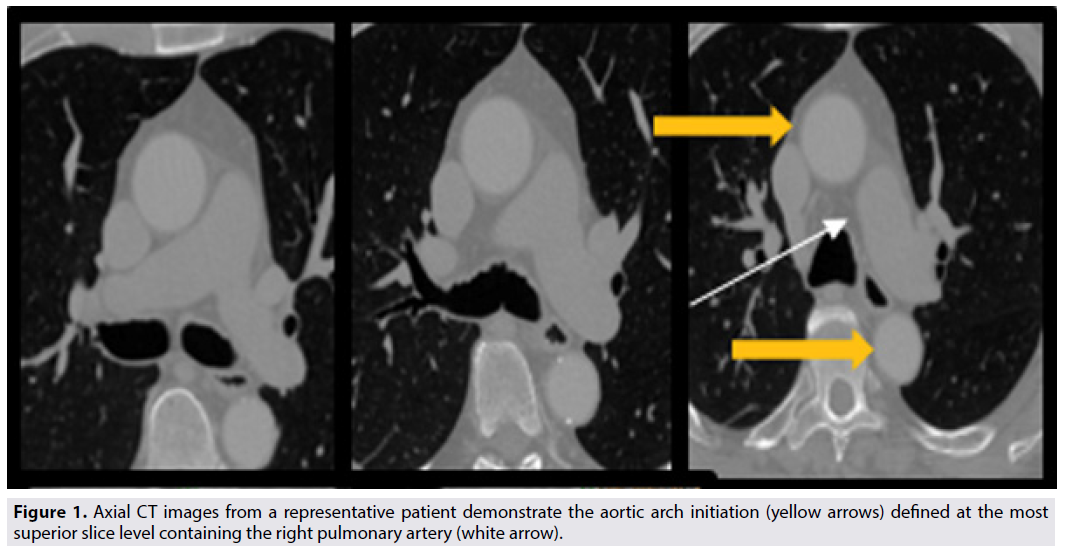
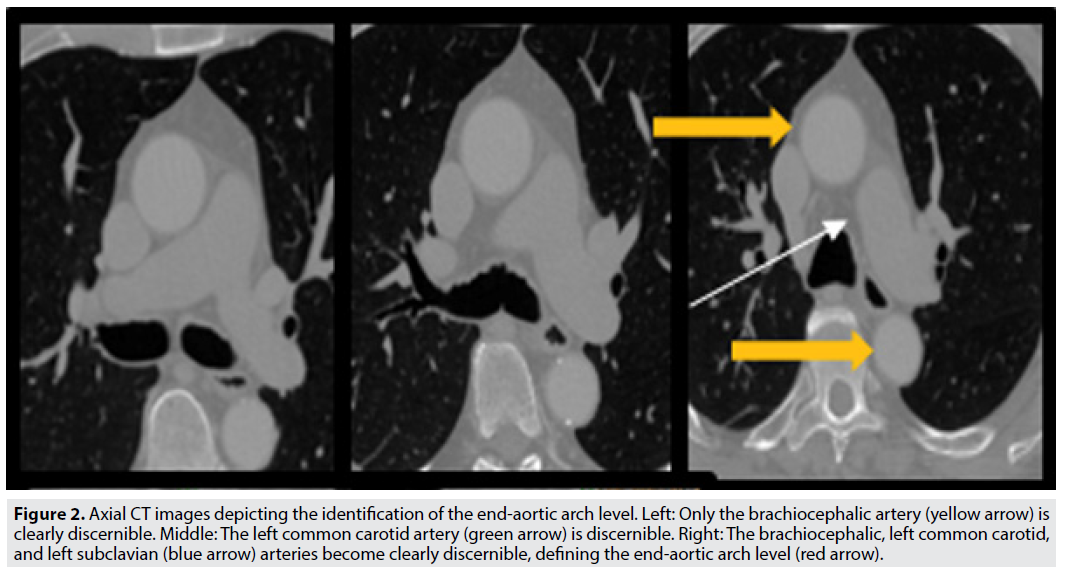
Next, to analyze the aortic arch segment, we use the right pulmonary artery (RPA) as an anatomic marker [19]. The most superior slice of the RPA (FIGURE 1, white arrow) defines the end-RPA level, which we use to define the initiation of the aortic arch (FIGURE 1, yellow arrows). In turn, we define the end-aortic arch level as the axial slice at which three distinct great vessels first become clearly appreciable (FIGURE 2). Any calcified focus seen from the end-RPA level to the end-aortic arch level is scored as AAC.
Figure 2: Axial CT images depicting the identification of the end-aortic arch level. Left: Only the brachiocephalic artery (yellow arrow) is clearly discernible. Middle: The left common carotid artery (green arrow) is discernible. Right: The brachiocephalic, left common carotid, and left subclavian (blue arrow) arteries become clearly discernible, defining the end-aortic arch level (red arrow).
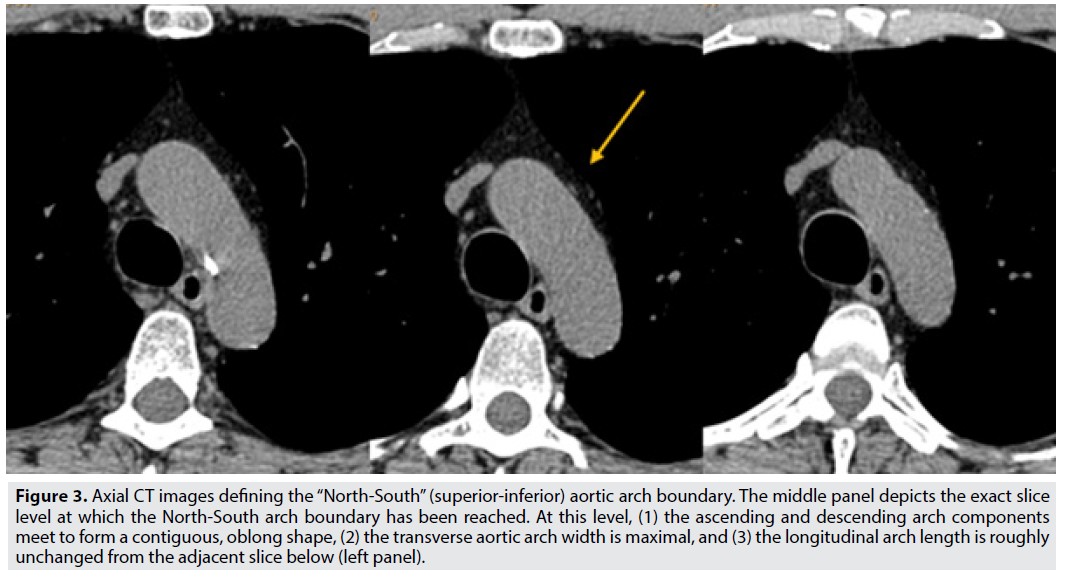
Initial studies suggest that calcium near the great vessels may indicate increased stroke risk [11,12]. A detailed segmentation technique in this region of the proximal aorta may provide important mechanistic data to aid future studies. To further distinguish between inferior and superior arch calcification, we therefore separate the aortic arch into two segments: inferior aspect and superior aspect of the aortic arch. We define the North- South aortic arch boundary on axial images using the following criteria: (1) the ascending and descending portions of the aortic arch must meet to become contiguous in a single oblong shape, (2) the transverse (i.e., left-right) aortic arch width is maximal, and (3) the longitudinal (i.e., anterior-posterior) arch length is stable or only minimally changing on the adjacent slice below. As illustrated in FIGURE 3, these criteria mark the boundary distinguishing the South (inferior) and North (superior) aortic arch.
Figure 3: Axial CT images defining the “North-South” (superior-inferior) aortic arch boundary. The middle panel depicts the exact slice level at which the North-South arch boundary has been reached. At this level, (1) the ascending and descending arch components meet to form a contiguous, oblong shape, (2) the transverse aortic arch width is maximal, and (3) the longitudinal arch length is roughly unchanged from the adjacent slice below (left panel).
■ Ascending and Descending Thoracic Aortic Calcification
Similarly, the ascending and descending thoracic aorta are individually scored for calcification. Ascending aortic calcium is quantified from the level of the LMCA superiorly to the end- RPA level. Descending thoracic aortic calcium is quantified from the level of the end-RPA inferiorly to the cardiac apex (i.e., the inferior-most slice level at which ventricular myocardium is still present). The distinct cardiac apex optimizes inter-observer reliability. Thus, we employ this anatomic marker as the inferior boundary of the descending thoracic aorta, in place of an alternate perimeter such as the aortic hiatus.
■ The Great Vessels: Brachiocephalic Artery Calcification (BAC), Common
Carotid Artery Calcification (CCAC), and Subclavian Artery Calcification (SAC) Calcification of the proximal components of the brachiocephalic artery, left common carotid artery, and left subclavian artery are also assessed on each CT study. The same slice level (i.e., the end-aortic arch level; see FIGURE 2) is used to demarcate the common inferior border for calcium measurements of all three of these vessels. The proximal brachiocephalic, left common carotid, and left subclavian arteries are each measured for plaque burden 25 mm superiorly from the end-aortic arch level. By dividing a total 25mm by the image slice thickness, the exact number of slices to measure in the caudo-cranial direction can be determined. This strategy was chosen to follow standard protocol among readers.
Reproducibility Analysis
■ Aortic Arch Calcification Measurement
Preliminary assessment of inter-observer variability was completed, following the aforementioned methodology. Reproducibility of extra-coronary measurements was performed on 90 randomly selected participant scans measured by a single reader. The second reader then completed measurements on the same 90 patient scans for inter-reader variability. These two trained analysts measured the scans independently blinded to the demographic data, and to each other’s reads.
Statistical Analysis
The authors randomly selected 90 ungated lung CT scans from the COPDGene cohort, a longitudinal study of current and former smokers, and evaluated for inter-reader variability. Bland-Altman plot analysis was used to test reproducibility between the two readers in 90 subjects. For patients with AAC, calcium scores were measured with Agatston and volumetric methods. The following measures were summed to yield total Arch Calcium Score and total Arch Volume: aortic valve and root; south and north aortic arch segment; brachiocephalic, common carotid and subclavian artery; ascending and descending aorta.
Results
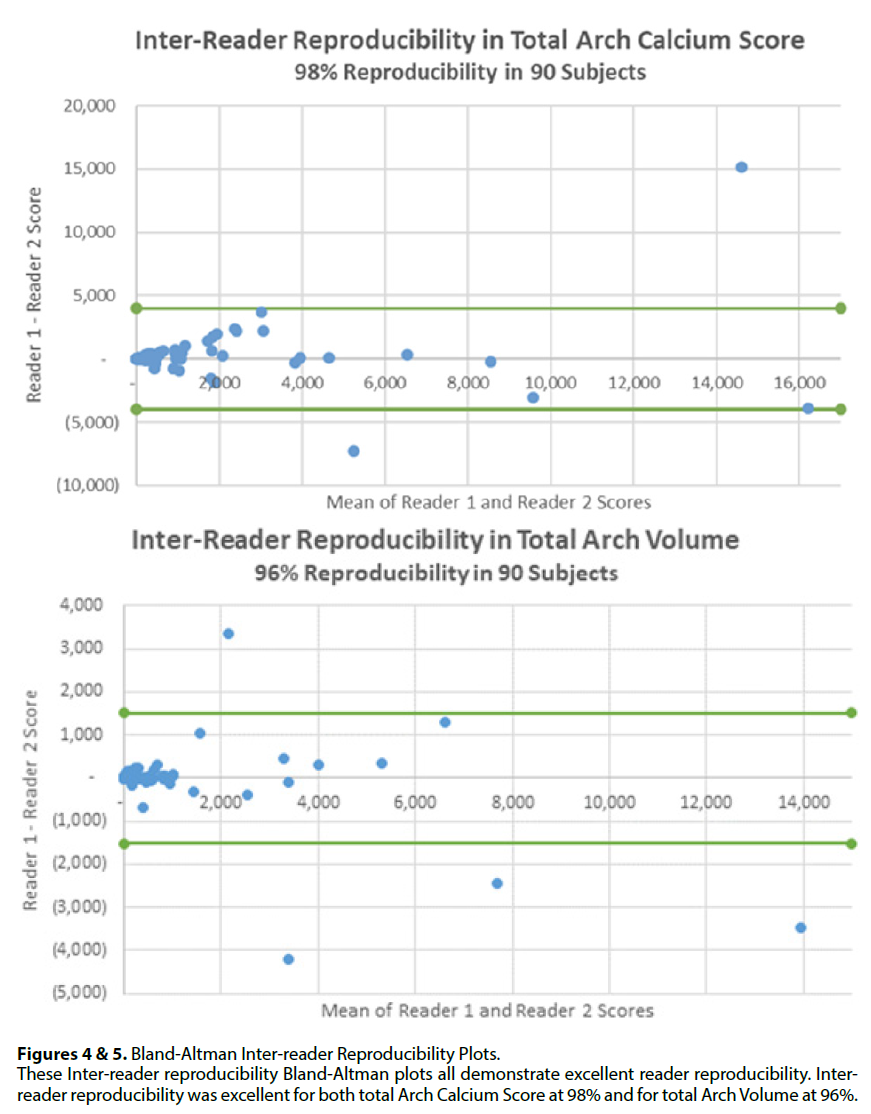
90 patients with AAC at ungated lung CT were included in analyses. Inter-reader reproducibility was excellent for total Arch Calcium Score in all subjects (98%) (FIGURE 4). Inter-reader reproducibility was similarly excellent for total Arch Volume in all subjects (96%) (FIGURE 5). Below FIGURES 4 and 5 are the respective Inter Reproducibility Bland-Altman plots.
Sub-analyses of arch calcification segments will be essential for future studies. Total arch reproducibility results attest to the strong interobserver reproducibility of the aforementioned arch calcium measurement methods. They indicate reputable methodology and anatomical definitions, that may be used to test whether CAC, AAC and their respective calcium density scores predict future ASCVD events similarly between gated and ungated scans.
Discussion
Our study proposes a novel methodology that can measure AAC, and this methodology can be used on all CT scans done for lung assessment. We also validated novel cardiac screening metrics of subclinical atherosclerosis, by assessing inter-observer reproducibility. Using COPDGene ungated non-contrast lung CT studies, quantification of full aortic arch and valve calcification is possible without additional imaging. Aortic and coronary calcification are always in the field of view of lung CT scans acquired for screening and diagnostic purposes, and there is a significant burden of CV risk factors in this COPD cohort.
The low inter-observer variabilities at ungated lung CT attest to this paper’s AAC measurement methods, in a population with smoking history and COPD. These findings can validate that AAC and CAC assessments on lung CT help CV risk recognition, thereby fostering early and focused preventive approaches for CVD. They help promote routine reporting of advanced atherosclerosis phenotyping, on millions of ungated CT scans acquired for lung screening purposes.
Budoff et al. evaluated the relationship between CAC severity and risk of all-cause mortality in the COPDGene study [6]. Over a median of 81.3 months, 12.4% of participants (850 of 6842) had died. While mortality in the CAC=0 group was 8.4% and 9.3% in the CAC=1-100 group, mortality was 12.7% in the CAC=101-400 group and 24.4% in the CAC>400 group (P<0.0001) [6]. The highest CAC category was thus significantly associated with increased mortality adjusted for known risk factors (hazard ratio [HR], 1.68; 95% CI, 1.30–2.03; P<0.0001) versus patients with non CAC. Budoff et al. found a strong association with COPD GOLD stage and calcium burden, with worsening mortality correlated with CAC and overall COPD (HR, 2.73; 95% CI, 2.27– 3.28; P<0.0001) [6]. Given that cardiovascular disease is a greater cause of mortality than lung cancer in this population with smoking history, increasing awareness is vital. It is incumbent on us to emphasize the increased risk for clinical and subclinical CVD among individuals with CVD. Preliminary studies suggest that the predictive power for mortality of CAC on routine CT scans can be extended to aortic calcium utility. Moreover, this study emphasizes the potential to screen for CVD among the millions of ungated thoracic scans completed annually, without any additional cost, radiation, or participant burden [6,24,25]. These findings support our high level of confidence in attaining concordant calcium measurements from gated and ungated CT scans, when evaluating AAC burden.
AAC in Predicting ASCVD Events
The Rotterdam Study reported on 2524 participants with 15,775 person-years of followup, and is to this day the largest study of aortic arch calcification [27]. While our methodology stratifies the arch into unique and specific markers, Rotterdam evaluates the arch as a less specific, single segment: from its origin to first centimeter of the great arteries [27]. After accounting for calcium burden measured in other regions (i.e., coronary arteries, extracranial and intracranial internal carotid arteries), aortic arch calcification was independently associated with a higher risk of all-cause mortality (HR per 1-SD increase, 1.42 [95% CI, 1.16–1.74]). AAC was also significantly associated with cardiovascular mortality (HR, 2.72 [95% CI, 1.85–4.02]). The study also examined AAC’s incremental predictive value when used alongside CAC to predict mortality [27]. After incorporating AAC into is models, Rotterdam indicated moderate improvements in the predictive value for all three mortality outcomes examined (i.e., cardiovascular, noncardiovascular, and all-cause mortality. Enhanced model performance was most evident for atherosclerotic cardiovascular mortality, with a continuous net reclassification of 0.36 [0.11–0.57] and 30.1% of events properly reclassified [27]. Supplementing Rotterdam’s established predictive value of AAC, our study for the first time stratifies the aortic arch into unique and standardizable anatomic sections. This paper’s novel methodology thus permits a stratified regional evaluation of aortic arch calcium. These AAC screening metrics lead the path for atherosclerotic disease risk stratification among vulnerable patients receiving lung CT assessment, without requiring additional scanning.
Conclusion
Segmented aortic arch calcium measurements can be obtained reproducibly from routine ungated lung CT scans, and may help improve cardiovascular risk assessment in a vulnerable population with smoking history and COPD. They suggest that this AAC segmenting method should allow for atherosclerotic disease risk stratification among patients undergoing lung CT evaluation, without requiring additional scanning.
It is imperative that we provide stronger evidence regarding the prognostic importance of AAC, CAC, and calcium density measured from nongated CT scans. If these imaging techniques prove to be independent predictors of CVD events, this method will provide outcome data to allow widespread dissemination and utilization. Doing so will help promote clinicians describe the presence of both AAC and CAC as a standard integrant of thoracic CT reads. Including coronary and aortic in all non-contrast chest CT reports may greatly improve early diagnosis, treatment, and patient education for coronary artery disease and atherosclerosis. This practice works to advance both preventative care and treatment adherence in CVD.
Acknowledgement
The project described was supported by the National Center for Advancing Translational Sciences through UCLA CTSI Grant UL1TR001881. The content is solely the responsibility of the authors and does not necessarily represent the official views of the NIH. Research reported in this publication was supported by the National Heart, Lung, and Blood Institute of the National Institutes of Health under Award Number K01HL125858 and K08 HL097029 (SML). The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Institutes of Health. The coronary calcification data used in this manuscript is supported by the Tobacco-Related Disease Research Program Award No. 20XT-0014 (PI: Dr. Matthew Budoff). This work was also supported by National Heart, Lung and Blood Institute NHLBI P01 HL105339
Funding
Not applicable.
Conflicts of Interest/Competing Interests
This paper has not been published and is not consideration for publication elsewhere. We have no conflicts of interest to disclose. All authors have read and approved this manuscript.
Author Contribution Statement
All authors confirm responsibility and contribution to the paper for the following: study conception and design, data collection, analysis and interpretation of results, and manuscript preparation. All authors reviewed the results and approved the final version of the manuscript.
Availability of Data and Material
Not applicable.
Code Availability
Not applicable.
Declaration
Additional declarations for articles in life science journals that report the results of studies involving humans and/or animals
Ethics Approval
Not applicable.
Consent to Participate
Not applicable.
Consent for Publication
Not applicable.
References
- Bhatt SP, Kazerooni E, Newell Jr JD et al. Visual estimate of coronary artery calcium predicts cardiovascular disease in COPD. Chest. 154, 579-587 (2018)
- Chen W, Thomas J, Sadatsafavi M et al. Risk of cardiovascular comorbidity inpatients with chronic obstructive pulmonary disease: a systematic review and metaanalysis. Lancet Respir Med. 3, 631-639 (2015).
- Portegies ML, Lahousse L, Joos GF et al. Chronic Obstructive Pulmonary Disease andthe Risk of Stroke. The Rotterdam Study. Am J Respir Crit Care Med. 193, 251-258 (2016).
- Greenland P, Blaha MJ, Budoff MJ et al. Coronary Calcium Score and Cardiovascular Risk. J Am Coll Cardiol. 72, 434-447 (2018).
- Budoff MJ, Takasu J, Katz R et al. Reproducibility of CT measurements of aortic valve calcification, mitral annulus calcification, and aortic wall calcification in the multi-ethnic study of atherosclerosis. Acad Radiol. 13, 166-72 (2006).
- Budoff MJ, Lutz SM, Kinney GL et al. Coronary Artery Calcium on Noncontrast Thoracic Computerized Tomography Scans and All-Cause Mortality. Circulation. 138, 2437-2438 (2018).
- Nakanishi R, Berman D, Budoff MJ et al. Current but not pastsmoking increases the risk of cardiac events: insights from coronary computedtomographic angiography. Eur Heart J. 36, 1031-1040 (2015).
- Budoff MJ, Young R, Burke G et al. Ten-year association of coronary artery calciumwith atherosclerotic cardiovascular disease (ASCVD) events: the multiethnicstudy of atherosclerosis (MESA). Eur Heart J. 39, 2401-2408 (2018).
- Shavelle DM, Takasu J, Budoff MJ et al. HMG CoA reductase inhibitor (statin) and aortic valve calcium. Lancet. 359, 1125-6 (2002).
- Otto CM, Lind BK, Kitzman DW et al. Association of aortic-valve sclerosis with cardiovascular mortality and morbidity in the elderly. N Engl J Med. 341, 142-7 (1999).
- Kronzon I, T.P.A.a.d.a.s.C.J. and -.d.C.P. 16818829.
- Jones EF, Kalman JM, Calafiore P et al. Proximal aortic atheroma: an independent risk factor for cerebral ischemia. Stroke. 26, 218-224 (1995).
- Amarenco P, Cohen A, Tzourio C et al. Atherosclerotic disease of the aortic arch and the risk of ischemic stroke. N Engl J Med. 331, 1474-1479 (1994).
- Regan EA, Hokanson JE, Murphy JR et al. Genetic epidemiology of COPD (COPDGene) study design. Copd. 7, 32-43 (2010).
- Agatston AS, Janowitz WR, Hildner FJ et al. Quantification of coronary artery calcium using ultrafast computed tomography. J Am Coll Cardiol. 15, 827-32 (1990).
- Hecht HS, Cronin P, Blaha MJ et al. 2016 SCCT/STR guidelines for coronary artery calcium scoring of noncontrast noncardiac chest CT scans: A report of the Society of Cardiovascular Computed Tomography and Society of Thoracic Radiology. J Cardiovasc Comput Tomogr. 11, 74-84 (2017).
- Carr JJ, Nelson JC, Wong ND et al. Calcified coronary artery plaque measurement with cardiac CT in population-based studies: standardized protocol of Multi-Ethnic Study of Atherosclerosis (MESA) and Coronary Artery Risk Development in Young Adults (CARDIA) study. Radiology. 234, 35-43 (2005).
- Yamamoto H, Shavelle D, Takasu J et al. Valvular and thoracic aortic calcium as a marker of the extent and severity of angiographic coronary artery disease. Am Heart J. 146, 153-9 (2003).
- Allison MA, Criqui MH, Wright CM. Patterns and risk factors for systemic calcified atherosclerosis. Arterioscler Thromb Vasc Biol. 24, 331-6 (2004).
- Fleg JL, Stone GW, Fayad ZA et al. Detection of high-risk atherosclerotic plaque: report of the NHLBI Working Group on current status and future directions. JACC Cardiovasc Imaging. 5, 941-55 (2012).
- Ahmadi N, Nabavi V, Hajsadeghi F et al. Mortality incidence of patients with non-obstructive coronary artery disease diagnosed by computed tomography angiography. Am J Cardiol. 107, 10-6 (2011).
- Muller JE, Kaufmann PG, Luepker RV et al. Mechanisms precipitating acute cardiac events: review and recommendations of an NHLBI workshop. National Heart, Lung, and Blood Institute. Mechanisms Precipitating Acute Cardiac Events Participants. Circulation. 96, 3233-9 (1997).
- Finn AV, Nakano M, Narula, J et al. Concept of vulnerable/unstable plaque. Arterioscler Thromb Vasc Biol. 30, 1282-92 (2010).
- Budoff MJ, Nasir K, Kinney GL et al. Coronary artery and thoracic calcium on noncontrast thoracic CT scans: comparison of ungated and gated examinations in patients from the COPD Gene cohort. J Cardiovasc Comput Tomogr. 5, 113-8 (2011).
- Wu MT, Yang P, Huang YL et al. Coronary arterial calcification on low-dose ungated MDCT for lung cancer screening: concordance study with dedicated cardiac CT. AJR Am J Roentgenol. 190, 923-8 (2008).
- Honoris L, DeFranco A, Port S et al. Correlation of Coronary Artery Calcium Scoring on Ungated Computed Tomography Compared to Gated Cardiac Computed Tomography Scans from the Multi-Ethnic Study of Atherosclerosis. J Am Coll Cardiol. 65, A1063 (2015).
- Bos D, Leening MJ, Kavousi M et al. Comparison of Atherosclerotic Calcification in Major Vessel Beds on the Risk of All-Cause and Cause-Specific Mortality: The Rotterdam Study. Circ Cardiovasc Imaging. 8, e003843 (2015).
Indexed at, Google Scholar, Crossref
Indexed at, Google Scholar, Crossref
Indexed at, Google Scholar, Crossref
Indexed at, Google Scholar, Crossref
Indexed at, Google Scholar, Crossref
Indexed at, Google Scholar, Crossref
Indexed at, Google Scholar, Crossref
Indexed at, Google Scholar, Crossref
Indexed at, Google Scholar, Crossref
Indexed at, Google Scholar, Crossref
Indexed at, Google Scholar, Crossref
Indexed at, Google Scholar, Crossref
Indexed at, Google Scholar, Crossref
Indexed at, Google Scholar, Crossref
Indexed at, Google Scholar, Crossref
Indexed at, Google Scholar, Crossref
Indexed at, Google Scholar, Crossref
Indexed at, Google Scholar, Crossref
Indexed at, Google Scholar, Crossref
Indexed at, Google Scholar, Crossref
Indexed at, Google Scholar, Crossref
Indexed at, Google Scholar, Crossref
Indexed at, Google Scholar, Crossref