Mini Review - Interventional Cardiology (2022)
Two-Dimensional Perfusion Angiography (2DPA) to predict outcome of peripheral angioplasty: A mini-review
- Corresponding Author:
- Vincent van Weel
Department of Vascular Surgery,
Meander Medical Centre,
Amersfoort,
The Netherlands,
E-mail: v.van.weel@meandermc.nl
Received date: 09-Jun-2022, Manuscript No. FMIC-22-66241; Editor assigned: 13-Jun-2022, PreQC No. FMIC-22-66241 (PQ); Reviewed date: 04-Jul-2022, QC No. FMIC-22-66241;Revised date: 11-Jul-2022, Manuscript No. FMIC-22-66241 (R);Published date: 18-Jul-2022, DOI: 10.37532/1755-5310.2022.14(S11).259
Abstract
Predicting clinical outcome of endovascular procedures is subjective and difficult to quantify. Two-Dimensional Perfusion Angiography (2DPA) is a promising tool to quantitatively assess reperfusion during surgery. However, important issues regarding imaging protocols and Time Density Curve (TDC) analysis should be further investigated in a large prospective study before clinical implementation is possible. The aim of this mini-review was to highlight essential topics that must be addressed before clinical implementation of 2DPA is valid. The added value of 2DPA to predict clinical outcome should be assessed by investigating the relation between perioperative TDC parameter ratios (after/before angioplasty) and validated clinical scores and vascular tests during follow up. Standardization of imaging and study protocols is essential to provide reliable and reproducible measures and allow for correct assessment of the added value of 2DPA for predicting clinical outcome after peripheral arterial intervention.
Keywords
2D perfusion angiography • 2DPA • Peripheral arterial disease • Lower extremity • Surgical decision making
Introduction
Peripheral arterial disease of the lower extremities is caused by atherosclerosis, which leads to narrowing or blockage (stenosis or occlusion) of the arteries and reduced blood flow to the distal tissues. The reduced supply of oxygenated blood to the extremities may cause intermittent claudication in the legs, slow or non-healing ulcers at the level of the lower limb or feet, or gangrene [1]. In 2015, the overall prevalence of peripheral arterial disease was estimated at 4.64%, the prevalence in the age group ≥ 70 years was as high as 16.62% and numbers are expected to increase even further in the near future [2,3].
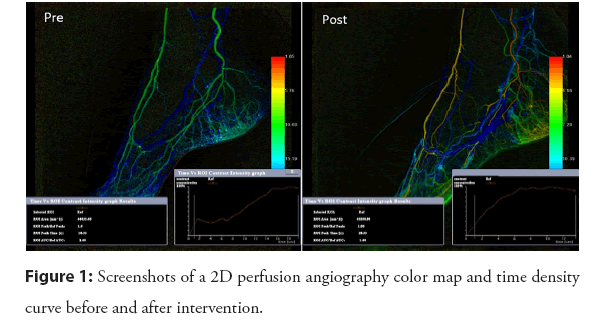
Peripheral arterial disease can be treated by managing symptoms, drug therapy and exercise (supervised by a physiotherapist), or in more severe cases with endovascular percutaneous transluminal angioplasty with or without stent placement or bypass surgery [1]. Endovascular techniques are rapidly developing and procedures are performed in more and more challenging cases of peripheral arterial disease, however, predicting treatment outcomes of endovascular procedures remain challenging [4]. In clinical practice, the degree of revascularization as seen on Digital Subtraction Angiography (DSA) images is used to determine whether sufficient increase in blood flow has been achieved [4,5]. However, this visual assessment of change in flow of contrast through the treated arteries is subjective and difficult to quantify. Two-Dimensional Perfusion Angiography (2DPA) is a relatively new technique that maps the flow of contrast imaged with DSA in a 2D colormap and subsequently generates a time density curve (TDC), enabling quantitative assessment of flow parameters. Therefore, 2DPA allows for quantification of reperfusion after or during surgery by assessment of the 2D color map and TDC parameters (Figure 1) [6]. This software package is increasingly being implemented in hybrid operating rooms, but is only scarcely used by clinicians. 2DPA has great potential, but our previous work also showed important practical limitations with respect to interpretation and clinical application [7]. This mini-review highlights essential topics that must be addressed before clinical implementation is valid.
Figure 1: Screenshots of a 2D perfusion angiography color map and time density curve before and after intervention.
Literature Review
The literature search included a search in PubMed in June 2022 using the search criteria: “perfusion angiography”. No search filter for publication date, study or article type was used. In total, 47 studies were reviewed based on title and abstract, of which 12 studies utilized 2DPA in patients with peripheral arterial disease of the legs. These 12 studies were included in this mini-review.
2DPA was first introduced by Jens, et al., sharing their first experience with this technique [6]. Since then, studies have mainly focused on analyzing differences in TDC parameters before and after revascularization [8-13]. Noteworthy are the large differences in study protocols, making comparison of results between studies difficult. Firstly, major variations in inclusion criteria for patient selection were found; classification of peripheral arterial disease differed between patients, ranging from intermittent claudication to severe tissue loss, and treatment consisted of both below and above the knee procedures. Secondly, imaging protocols varied widely between studies for contrast bolus (4 to 15 ml), contrast injection speed (2 to 5 ml/s or by hand), number of DSA frames (10 to 90 frames) and frame rate (1 to 3 /s). Some essential parameters are not even mentioned in most studies, such as location of catheter tip during contrast injection and use of a footrest to minimize movement artefacts. Thirdly, Region Of Interest (ROI) definitions and calculated TDC parameters varied 260between studies; Area Under the Curve (AUC) and Time To Peak (TTP) are mostly used (n=5), followed by Time Of Arrival (TOA), Mean Transit Time (MTT), Peak Density (PD), Wash-in Rate (WiR) (n=3) and curve width (n=1) [8-13] . Nevertheless, all studies found improved perfusion after revascularization, based on one or more TDC parameter [8-13].
Only 4 out of 12 included studies mentioned clinical follow-up [7-9,11], of which 2 studies investigated the association between TDC parameters after revascularization and clinical outcome [7,8]. Troisi, et al. found a marginal association between increase in peak density (>50%) and higher 6-month wound healing rate [8]. Our research team found only a limited association between TDC parameters (specifically, AUC and TOA) and peripheral arterial disease classification scores (Fontaine classification, American Medical Association whole person impairment classification (AMA) and average Wound, Ischemia, foot Infection (WIfI) score) [7,14,15]. In addition, it should be noted that we found a large variance in TDCs between patients, reflecting the diversity of this population [7].
Common limitations of studies investigating 2DPA are the limited population size (5 to 39 patients), the lack of a control group and lack of extensive follow-up. After reviewing these studies, we believe that standardization of patient population, imaging protocol and ROI/TDC definition is crucial. As a next step, a prospective cohort study is mandatory to demonstrate a reliable and relevant relation between TDC parameters and clinical outcome, and thus its value for clinical decision making.
Discussion
This mini-review assessed the shortcomings, missing links and essential topics that must be addressed before clinical implementation of 2DPA to support decision-making during endovascular treatments is valid. Although multiple studies have shown that an increase in blood flow can be quantitatively demonstrated using TDC parameters, it is yet unknown how these parameters should be interpreted in clinical practice [16]. Ideally, a threshold or range of TDC parameters should be defined to support surgeons and/or interventional radiologists with perioperative decision making and to estimate whether sufficient revascularization has been achieved. Some technical recommendations on 2DPA discussed below may offer researchers in this field a stepping stone towards improving interpretability and, ultimately, achieve clinical application of 2DPA for endovascular treatment. A carefully designed study with new improved protocols for 2DPA will hopefully demonstrate the value of 2DPA for clinical practice.
Methodological choices during intervention that should be considered are technical choices regarding contrast admission and imaging protocol as these largely affect the shape of TDCs. Contrast injection should be done with the tip of the catheter at a fixed location, because this largely affects TOA, for example, at the mid-popliteal region for below-the-knee interventions. Also, a standardized bolus of contrast should be injected by pump to ensure a constant flow of contrast. Images should be acquired with a fixed frame rate (e.g. 3/s), starting simultaneously with contrast injection and stopping after venous outflow of contrast. Venous outflow may vary per patient, thus imaging should not be performed with a fixed number of frames. Early termination of acquisition may affect AUC. Lateral positioning of the X-ray tube at fixed coordinates is preferred. In combination with a footrest, the fixed tube position will increase reproducibility and reduce variations in TDC parameters. Moreover, a footrest will reduce the chance for motion artifacts. However, care should be taken that immobilization does not result in compression of blood vessels.
Analytical choices include ROI definition and how TDC parameters are assessed. In most studies, ROI is defined as the area between the tibiotalar joint and toes. Toes are susceptible to motion artifacts and should not be included in the ROI. Additionally, 2DPA series before and after revascularization should be performed to be able to calculate flow ratios (TDC values after /before revascularization). This probably has the best predictive value since TDC’s vary greatly between patients and may be patient and/or disease dependent [6,7]. However, this has not yet been confirmed in solid studies and therefore requires further investigation.
Clinical success after revascularization should be assessed in future studisaes using quantitative perfusion measurements, such as toe pressure, transcutaneous oxygen pressure and ankle brachial index/duplex, and/or with validated clinical scoring systems. These scores should not only encompass wound healing, as most patients do not necessarily have a foot ulcer; claudication is the most common symptom. TDC ratios should be compared to the scoring systems and/or quantitative perfusion measures to be able to define a threshold that indicates good clinical outcome.
Finally, a major limitation of 2DPA is the 2D projection of 3D structures, causing loss of potentially critical information [16]. In future studies we also see opportunities for 3DPA to further improve decision making [17].
Conclusion
With this mini-review, we found major variations in 2DPA study protocols making comparison between studies difficult. In addition, we could not find substantial evidence for the relationship between 2DPA TDC parameters and clinical outcome. To warrant clinical application of 2DPA, a well-designed prospective study should be performed. Such a study should standardize protocols and investigate the relation between TDC parameter ratios (after/before intervention) and clinical outcome as assessed with validated scores, questionnaires and quantitative perfusion measures.
References
- Aboyans V, Ricco J, Bartelink MEL, et al. ESC Guidelines on the diagnosis and treatment of peripheral arterial diseases, in collaboration with the European Society for Vascular Surgery (ESVS). Eur Heart J. 39(9): 763-821 (2018).
[CrossRef] [Google Scholar] [PubMed]
- Eraso LH, Fukaya E, Mohler ER, et al. Peripheral arterial disease, prevalence and cumulative risk factor profile analysis. Eur J Prev Cardiol. 21(6): 704-711 (2014).
[CrossRef] [Google Scholar] [PubMed]
- Conte MS, Pomposelli FB, Clair DG, et al. Society for Vascular Surgery practice guidelines for atherosclerotic occlusive disease of the lower extremities: Management of asymptomatic disease and claudication. J Vasc Surg. 61(S3): 2S-41S (2015).
[CrossRef] [Google Scholar] [PubMed]
- Cooper KJ, Peña C, Benenati J. Determining end points for critical limb ischemia interventions. Tech Vasc Interv Radiol. 19(2): 104-112 (2016).
[CrossRef] [Google Scholar] [PubMed]
- Utsunomiya M, Takahara M, Iida O, et al. Wound blush obtainment is the most important angiographic endpoint for wound healing. JACC Cardiovasc Interv. 10(2): 188-194 (2017).
[CrossRef] [Google Scholar] [PubMed]
- Jens S, Marquering HA, Koelemay MJW, et al. Perfusion angiography of the foot in patients with critical limb ischemia: Description of the technique. Cardiovasc Intervent Radiol. 38(1): 201-205 (2015).
[CrossRef] [Google Scholar] [PubMed]
- Verschuur AS, Groot Jebbink E, Lo-A-Njoe PE, et al. Clinical validation of 2D perfusion angiography using Syngo iFlow software during peripheral arterial interventions. Vascular. 29(3): 380-386 (2021).
[CrossRef] [Google Scholar] [PubMed]
- Troisi N, Michelagnoli S, Panci S, et al. Association of 2D perfusion angiography and wound healing rate in combined femoro-popliteal and below-the-knee lesions in ischemic patients undergoing isolated femoro-popliteal endovascular revascularization. Int J Low Extrem Wounds. (2021).
[CrossRef] [Google Scholar] [PubMed]
- Pärsson HN, Lundin N, Lindgren H. 2D perfusion-angiography during endovascular intervention for critical limb threatening ischemia: A feasibility study. JRSM Cardiovascular Disease. 9: 1-7 (2020).
[CrossRef] [Google Scholar] [PubMed]
- Ikeoka K, Watanabe T, Shinoda Y, et al. Below-the-ankle arrival time as a novel limb tissue perfusion index: Two-dimensional perfusion angiography evaluation. J Endovasc Ther. 27(2): 198-204 (2020).
[CrossRef] [Google Scholar] [PubMed]
- Yoneyama F, Osaka M, Sato F, et al. Efficacy of two-dimensional perfusion angiography for evaluations after infrapopliteal bypass surgery for critical limb ischemia. Ann Vasc Dis. 11(2): 248-251 (2018).
[CrossRef] [Google Scholar] [PubMed]
- Hinrichs JB, Murray T, Akin M, et al. Evaluation of a novel 2D perfusion angiography technique independent of pump injections for assessment of interventional treatment of peripheral vascular disease. Int J Cardiovasc Imaging. 33(3): 295-301 (2016).
[CrossRef] [Google Scholar] [PubMed]
- Murray T, Rodt T, Lee MJ. Two-dimensional perfusion angiography of the foot: Technical considerations and initial analysis. J Endovasc Ther. 23(1): 58-64 (2016).
[CrossRef] [Google Scholar] [PubMed]
- Hardman RL, Jazaeri O, Yi J, et al. Overview of classification systems in peripheral artery disease. Semin Intervent Radiol. 31(04): 378-387 (2014).
[CrossRef] [Google Scholar] [PubMed]
- Darling JD, Mccallum JC, Soden PA, et al. Predictive ability of the SVS WIfI classification system following first-time lower extremity revascularizations. J Vasc Surg. 65(3): 695-704 (2017).
[CrossRef] [Google Scholar] [PubMed]
- Ma J, Biu B. Selecting proper region of interest and parameters affects feasibility of two dimensional perfusion angiography. Eur J Vasc Endovasc Surg. 62(4): 660 (2021).
[CrossRef] [Google Scholar] [PubMed]
- Gurgitano M, Signorelli G, Rodà GM, et al. Use of perfusional CBCT imaging for intraprocedural evaluation of endovascular treatment in patients with diabetic foot: A concept paper. Acta Biomed. 91(10-S): 1-8 (2020).
[CrossRef] [Google Scholar] [PubMed]