Case Report - International Journal of Clinical Rheumatology (2021) Volume 16, Issue 1
Kimura disease and IgG4-related disease: Same disease spectrum or separate co-existing conditions?
- Corresponding Author:
- Tamir Malley
Department of Rheumatology, Wexham Park Hospital, United Kingdom
E-mail: tam.malley@doctors.org.uk
Abstract
Here we report the case of a previously healthy 42-year old gentleman presenting with lacrimal, parotid and submandibular gland enlargement with associated pulmonary nodules, a pancreatic cyst and radiographic features of pancreatitis. Lacrimal gland biopsy indicated features in keeping with Kimura disease; however the clinical picture was not entirely typical. Prior to commencing treatment a further biopsy was sought from an alternative site, left submandibular gland, which confirmed features of IgG4-related disease. Although Kimura disease and IgG4-related disease share various clinical, serological and histopathological features; they are recognised as two distinct clinical entities. We discuss the possibility that the two conditions are on the same disease spectrum.
Introduction
Kimura Disease is a rare chronic inflammatory soft tissue disorder of yet unknown aetiology often presenting with subcutaneous masses and co-existing lymphadenopathy in the head and neck region without systemic symptoms [1]. It predominantly affects Asian males in the 2nd to 4th decade of life [1]. IgG4-related disease is a relatively recently established disease concept characterised by fibrosis of affected tissue and organs, with lymphoplasmacytic infiltrate enriched in IgG4-positive plasma cells. The condition links a variety of disorders previously thought to be unrelated [2].
Case Report
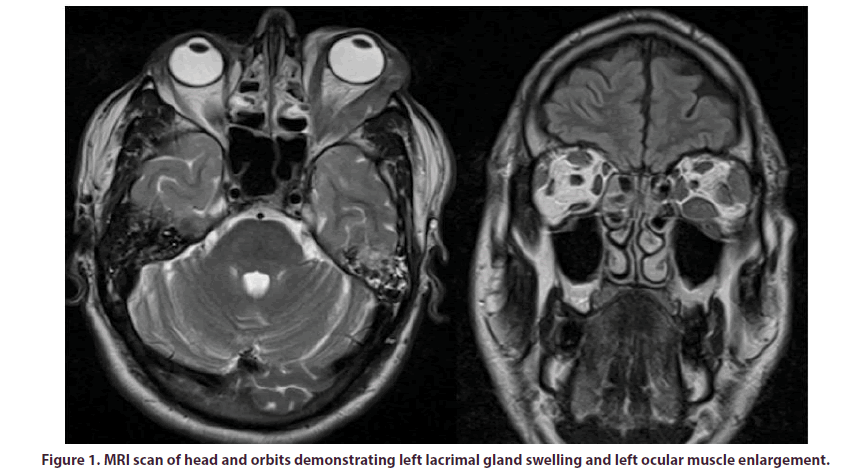
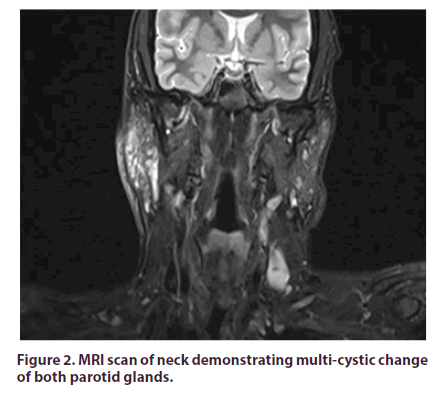
A 42-year old gentleman, of Indian origin, presented to us with a 3-year history of left upper eyelid swelling. He has a past medical history of asthma (Beclometasone inhaler), hypertension (Lacidipine 4 mg daily) and a congenital left-sided ptosis treated surgically in India at the age of 14. His eyelid swelling was initially treated as eczema by the General Practitioner without improvement. Prior to seeing us, the patient had been reviewed by the Ophthalmology team who organised an MRI scan of his head and orbits which demonstrated left lacrimal gland swelling and left ocular muscle enlargement (Figure 1). Subsequent biopsy of the left lacrimal gland revealed chronic inflammatory cell infiltrate involving fibrous tissue and fat, with lymphoid follicle formation, vascular proliferation and areas of prominent eosinophilic infiltration. Features were in favour of a possible diagnosis of Kimura disease; initially ruling out Ig4-related disease, malignancy, vasculitis and sarcoidosis. He was then reviewed by the Ear, Nose and Throat team following the development of contralateral lacrimal gland, left parotid gland and right submandibular gland swelling. An MRI scan of his neck demonstrated multi-cystic change of both parotid glands (Figure 2). Fine needle aspiration of the right submandibular gland yielded a non-diagnostic low volume and cellularity specimen.
Upon our assessment, history revealed no prodromal or current features of systemic illness, connective disease or vasculitis. There was no relevant family history and he did not drink alcohol, smoke cigarettes or use recreational drugs. His vision was previously affected by his congenital left-sided ptosis but did not report any new visual changes. On examination, there was bilateral upper eye lid swelling with scleral inflammation and longstanding left-sided ptosis and inability to perform downward-lateral gaze of left eye, without diplopia. He had tender and swollen parotid glands bilaterally. The remainder of his physical examinationincluding cardiovascular, respiratory, gastrointestinal, neurological and dermatological systems-was unremarkable.
Initial laboratory results were as follows: Haemoglobin 159g/L [reference range: 125-180 g/L], WCC 9.2x109/L (4-11), eosinophils 1.81x109/L (0.0-0.3), platelets 352x109/L (150-450), C-reactive protein 3 mg/L (0-4), erythrocyte sedimentation rate 29 mm/h (1-7), Albumin 44 g/L (38-51), estimated glomerular filtration rate 70 mL/min/1.73 m2 (>60). Liver function tests, bone profile, thyroid stimulating hormone and creatine kinase were all normal. Immunology revealed a weakly positive Anti-Nuclear Antibody (ANA) titre 1:160 homogeneous pattern, with a negative extractable nuclear antibody and a negative double-stranded DNA. c-ANCA was negative, p-ANCA was obscured by positive ANA, with negative MPO and PR3. Complement levels were normal. IgG was raised 23 g/L (6-16) (IgG lambda paraprotein on serum electrophoresis), reduced IgM and normal IgA. Serum IgG4 was raised at 20.8 (0.0- 1.3), as was IgE 1190 kU/L (0-81) and serum ACE 80U/L (8-52). Salivary gland antibodies were negative. His virology, including HIV, hepatitis B and C, were negative. Urinalysis was normal.
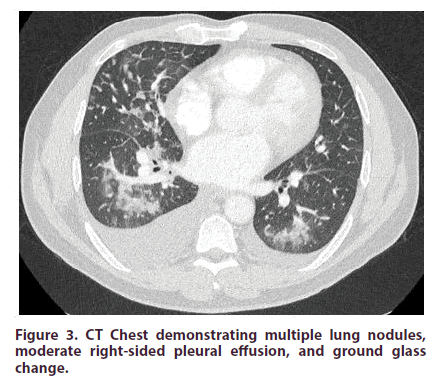
The initial CT chest-abdomen-pelvis with contrast revealed multiple lung nodules, moderate right-sided pleural effusionand ground glass changes (Figure 3). The scan also showed a 2.4 cm cyst on the pancreas with co-existent radiographic features of autoimmune pancreatitis.
Figure 3: CT Chest demonstrating multiple lung nodules, moderate right-sided pleural effusion, and ground glass change.
Further biopsy, on this occasion the left submandibular gland, was performed which demonstrated a moderately dense lymphoplasmacytic infiltrate with some eosinophils and significant fibrosis with several thick-walled blood vessels. Additionally, IgG4 immunoperoxidase was diffusely positive in the plasma cells (IgG4+ plasma cells >100 per high-power field with IgG4-IgG ratio >60%), highly suggestive of IgG4-related disease. As such, with robust evidence of a steroid responsive condition, prednisolone 40mg was commenced with positive effect with a plan to progressively wean the dose over the following months. Unfortunately, his disease course was complicated by an ST-elevation myocardial infarction, requiring percutaneous coronary intervention to the left anterior descending artery, followed by an episode of acute pulmonary oedema requiring an admission to the coronary care unit. An echocardiogram revealed a left ventricular ejection fraction of 30%, reduced from 60% on a previous echocardiogram prior to this cardiac event.
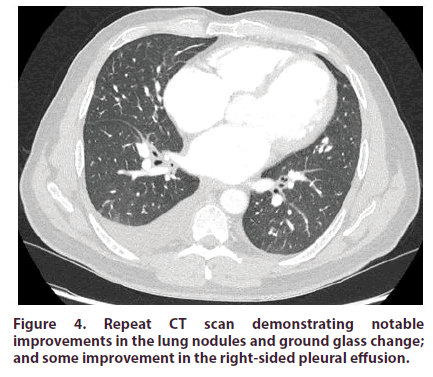
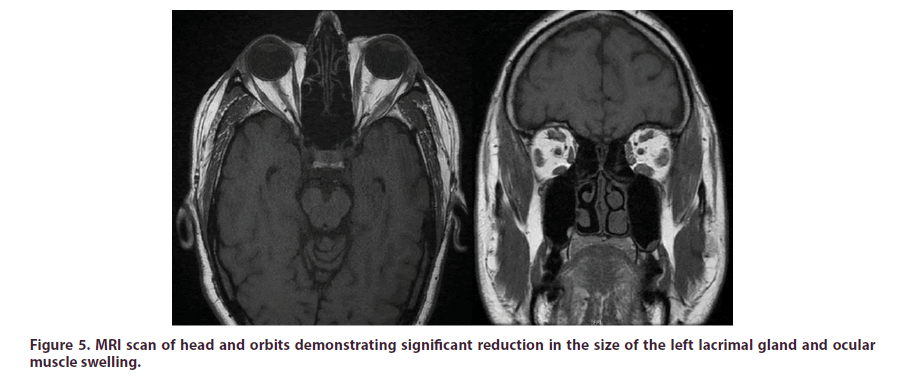
Repeat imaging was performed to assess his response to steroid therapy. CT chest-abdomen-pelvis with contrast revealed a significant improvement, with notable improvements in the lung nodules and ground glass changes; and some improvement in the rightsided pleural effusion (Figure 4). MRI orbits revealed significant reduction in the size of the left lacrimal gland and ocular muscles (Figure 5). Furthermore, there was a marked clinical improvement in his bilateral upper eyelid swelling. He was maintained on Prednisolone 15 mg daily whilst further multi-disciplinary team opinion was sought from our affiliated specialist centre who confirmed that the imaging and histological findings were typical of IgG4-related disease and suggested introducing Azathioprine (2 mg/kg) with continued steroid weaning.
Figure 4: Repeat CT scan demonstrating notable improvements in the lung nodules and ground glass change; and some improvement in the right-sided pleural effusion.
Discussion
Kimura disease is a rare chronic inflammatory condition characterised by painless solitary or multiple asymmetric subcutaneous nodules primarily affecting the head and neck region with co-existent lymphadenopathy [3]. Nodules typically occur in preauricular, submandibular and parotid glands, with few reports suggesting lacrimal gland involvement [4]. Biochemical findings include eosinophilia (>5% of white blood cell count, or >0.5 × 109/L count in blood) and elevated IgE levels [5]. Histologically, dense eosinophilic infiltration in the form of micro-abscesses and granuloma formation is usually observed. Additionally, tissue fibrosis, sclerosis and vascular proliferation can be present. Heavy IgE deposits with variable amounts of IgG, IgM, and fibrinogen are seen on immunofluorescence [1].
IgG4-related disease is a relatively recently established disease concept characterised by fibrosis of affected tissue and organs, with lymphoplasmacytic infiltrate enriched in IgG4-positive plasma cells [2]. Several different conditions which were once thought to be unrelated are now considered manifestations of the same histopathological process, thus the umbrella term IgG4-related disease [6]. It is a multisystemic condition with the potential to affect many organs including salivary glands, orbits, pituitary gland, pancreas, retroperitoneum and kidneys. Histology primarily features a lymphoplasmacytic infiltrate enriched in IgG4-positive plasma cells, fibrosis and eosinophilia. Elevated serum IgG4 is seen in 60-70% of patients with IgG4-related disease confirmed on histology. Elevated serum IgE and eosinophilia is also often found [2].
Kimura disease and IgG4-related disease share various clinical, serological and histopathological features. However, the relationship between these two conditions, if any, is not well recognised or understood. A case report from Japan by Tsubouchi and colleagues reports a 46-year old gentleman with Kimura disease in whom an abundance of IgG4-positive plasma cells were found on biopsy of a lung mass, which improved with steroid treatment [7]. They hypothesised that the two conditions were co-existent. Since then, however, case reports have emerged suggesting that the presence IgG4-positive plasma cells in the blood, lymph nodes and skin lesions are epiphenomena of Kimura’s disease. Jing Li and colleagues present the case of a 47-year old gentleman with left lacrimal gland swelling with histological features in keeping with Kimura disease [8]. Further immunohistochemical analysis of the sample demonstrated an IgG4 to IgG positive cell ratio of 35% which raised the possibility of a diagnosis of, or perhaps common features with, IgG4-related disease. They report a positive and sustained clinical and biochemical response (decreasing serum IgE and eosinophils) with one month of prednisone 25 mg, reducing by 5 mg every two weeks thereafter.8 Similar cases of confirmed Kimura disease with immunohistochemical evidence IgG4-positive plasma cells have also been documented by Lei Liu and colleagues [9]. To explain the presence of IgG4-positive plasma cells in Kimura disease they propose the “allergen-specific immunotherapy” model, whereby repeated exposure to allergens modify allergenspecific immune responses such that patients can tolerate higher doses of those allergens. Thus, persistent exposure to an allergen could trigger the occurrence of Kimura disease via cytokine release (IL-4, IL-5 and IL-13) leading to elevated IgE and eosinophils thus causing a type 1 hypersensitivity reaction. IL-4 and IL-13 promote class switching of B-cells to IgE and IgG4. Over time the type 1 hypersensitivity reaction disappears with prolonged allergen exposure, through induction of regulatory T-cells and suppression of helper T-cell mediated inflammation, therefore the presence of IgG4 would predominate leading to lesions rich in IgG4-positive plasma cells. Further work is required to establish the validity of this model but this may be challenging given the rarity of Kimura disease.
Conclusion
Kimura disease and IgG4-related disease sometimes share similar clinical and histopathological features which can make the diagnosis challenging. Despite a paucity of clinical data, it remains uncertain whether these two conditions are distinct clinical entities which occasionally occur together or, indeed, are on the same disease spectrum. Conveniently, both conditions are steroid responsive which minimises the impact of this clinical conundrum when it comes to initiating treatment.
Conflict of interest statement
The authors have no conflicts of interest to declare.
Funding
No funding source.
Ethical approval
Not required.
Consent
The patient provided written informed consent for their information and images to be published.
References
- Osuch-Wojcikiewicz E, Bruzgielewicz A, Lachowska M et al. Kimura Disease in a Caucasian Female: A Very Rare Cause of Lymphadenopathy. Case .Rep. Otolalyngol. 14, 1–4 (2014).
- Khosroshahi A, Stone JH. A clinical overview of IgG4-related systemic disease. Curr. Opin. Rheumatol. 23(1), 57–66 (2011).
- Li TJ, Chen XM, Wang SZ et al. Kimura's disease. A clinicopathologic study of 54 Chinese patients. Oral. Surg. Oral. Med. Oral. Pathol. 82(5),549–555 (1996).
- Chan KM, Mok JSW, Ng SK et al. Kimura's disease of the auricle. Otolaryngol. Head. Neck. Surg. 124(5),598–599 (2001).
- Wang DY, Mao JH, Zhang Yet al. Kimura disease: a case report and review of the Chinese literature. Nephron. Clin. Pract. 111, 55–61 (2009).
- Stone JH, Zen Y, Deshpande V. IgG4-Related Disease. N. Engl. J. Med. 366(6), 539–551 (2012).
- Tsubouchi K, Imanaga T, Yamamoto M et al. A case of IgG4-positive multi-organ lymphoproliferative syndrome associated with Kimura disease. Nihon. Kokyuki. Gakkai. Zasshi. 48(7), 524–528 (2010).
- Li J, Ge X, Ma J et al. Kimura’s disease of the lacrimal gland mimicking IgG4-related orbital disease. BMC.Ophthalmol. 14, 158 (2014).
- Liu L, Chen Y, Fang Z et al. Kimura's disease or IgG4-related disease? A case-based review. Clin. Rheumatol. 34(2), 385–389 (2013).