Research Article - Interventional Cardiology (2020) Volume 12, Issue 7
Angiographical coronary artery calcification predicts antegrade guidewire crossing during percutaneous coronary intervention of chronic total occlusion
- Corresponding Author:
- Tomotaka Dohi
Department of Cardiovascular Biology and Medicine,
Juntendo University Graduate School of Medicine,
Tokyo, Japan, 2-1-1 Hongo, Bunkyo-ku,
Tokyo 113-0033, Japan,
E-mail: tdohi@juntendo.ac.jp
Received date: November 17, 2020; Accepted date: December 01, 2020;Published date: December 08, 2020
Abstract
Background: Coronary artery calcification (CAC) is a predictor of failed revascularization in Chronic Total Occlusion (CTO) Percutaneous Coronary Intervention (PCI). However, CTO PCI operators believe that angiographical CAC shows promise as a landmark in the manipulation of antegrade guidewire crossing. We aimed to identify the relationship between angiographical CAC and successful antegrade guidewire crossing in CTO PCI.
Methods and Findings: This single-center, retrospective, observational study enrolled 48 consecutive patients who underwent Coronary Computed Tomography Angiography (CCTA) before PCI for CTO. They were divided, according to the level of antegrade guidewire crossing success, into the success (≤ 30 min; n=25, 50%) and unsuccess (>30 min; n=25, 50%) groups. All the CCTA and coronary angiographic findings obtained before PCI were analyzed. We assessed the relationship between angiographical CAC and successful antegrade guidewire crossing and other factors associated with it. The baseline characteristics were similar between the groups. Angiographical CAC was more frequently observed in the success group than in the unsuccess group (68% vs. 24%, p=0.080). There were no statistically significant differences in the CTO entry morphology, bridging collateral, vessel bending, distal reference diameter, occlusion length, and calcification severity between the groups. In the multivariable analysis, angiographical CAC in the CTO lesion predicted successful antegrade guidewire crossing within 30 min (odds ratio=0.043, 95% confidence interval=0.003–0.614, p=0.006).
Conclusion: Angiographical CAC may be a promising landmark for antegrade wire manipulation in patients with CTO.
Abbreviations: CAC: Coronary Artery Calcification; CAG: Coronary Angiography; CART: Controlled Antegrade and Retrograde Subintimal Tracking; CCTA: Coronary Computed Tomography Angiography; CTO: Chronic Total Occlusion; IVUS: Intravascular Ultrasound; J-CTO: Multicenter CTO Registry of Japan; LAD: Left Descending Artery; LCX: Left Circumflex Artery; PCI: Percutaneous Coronary Intervention; RCA: Right Coronary Artery; TIMI: Thrombolysis in Myocardial Infarction
Keywords
Percutaneous Coronary Intervention • Chronic Total Occlusion • CTO
Introduction
Chronic Total Occlusion (CTO) occurs in approximately 15%-20% of patients with coronary artery disease undergoing Coronary Angiography (CAG) [1-3]. Percutaneous Coronary Intervention (PCI) for CTO remains a challenging treatment owing to the associated operational difficulties and lesion severity. With the development of novel devices and strategies in recent times, the total revascularization success rate in CTO PCI is 85%-90% [4-6]. The retrograde approach, which is performed through the collateral channels using Controlled Antegrade And Retrograde subintimal Tracking (CART) and reverse CART [7,8], yields success rates of 83%-90% [9-11]. However, the antegrade approach shows a success rate of only 60%-70% [11-12]; this rate needs improvement for the achievement of total success rate enhancements. Among the important factors associated with antegrade, wiring is the obtainment of promising landmarks of vessel course. While expert operators believe that the presence of angiographical coronary artery Calcification (CAC) in CTO lesions is among the promising landmarks for wire manipulation, previous studies showed that CAC is an independent predictor of failed CTO PCI [13-15]; there is a lack of clarity on the relationship between angiographical CAC and antegrade guidewire crossing success. Additionally, there are some discrepancies in the existing findings pertaining to the effect of CAC in CTO PCI. Accordingly, this study aimed to identify the relationship between angiographical CAC and successful antegrade guidewire crossing for CTO PCI. This aim was achieved that angiographical CAC in the CTO lesion predicts successful antegrade guidewire crossing.
Methods
Study population
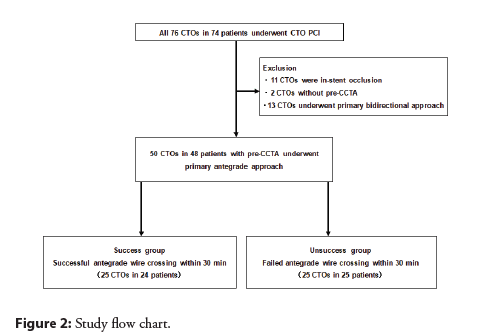
This single-center, retrospective, observational study enrolled 48 consecutive patients with 50 CTO lesions who underwent Coronary Computed Tomography Angiography (CCTA) before PCI for CTO at Saiseikai Utsunomiya Hospital, Tochigi, Japan, between May 2016 and December 2017. The exclusion criteria were as follows: a history of acute myocardial infarction within 1 month, absence of CCTA before CTO PCI, planned primary retrograde approach for CTO PCI, and in-stent occlusion presence.
CTO was defined as a Thrombolysis in Myocardial Infarction (TIMI) flow grade of 0 in the coronary lesion for at least 3 months based on clinical information and previous angiography findings [16]. In the Multicenter CTO Registry of Japan (J-CTO) score [17], successful guidewire crossing within 30 min is a primary endpoint in the determination of difficulties in CTO lesion treatment; this is one of the most objective parameters reflective of guidewire crossing-related difficulties. Additionally, in the Computed Tomography Registry of Chronic Total Occlusion Revascularization (CT-RECTOR) [14], the primary endpoint was the achievement of guidewire crossing success within 30 min. We, therefore, defined successful antegrade guidewire crossing as that which was achieved within 30 min (success group). Patients in whom antegrade guidewire crossing success was achieved at over 30 min and those who switched to the retrograde approach after antegrade approach failure were assigned to the unsuccess group. This study was performed in compliance with the Declaration of Helsinki and was approved by the institutional review board of Saiseikai Utsunomiya Hospital.
CCTA protocol and analysis
A wide detector 64-slice dual source CT scanner (SOMATOM Definition, SIEMENS, Munich, Germany) was used for CCTA performance within 3 months before CTO PCI. Drugs for rate control were not administered to any of the patients. Sublingual nitroglycerin was administered before CCTA. During CCTA, 0.7 mL/kg contrast (Iopamiron 370, Bayer, and Berlin Germany) was injected, followed by saline. Retrospective electrocardiographically gated CCTA was performed with the following parameters: tube voltage 120 kV; tube current 606 mA (dual source); collimation 64 x 0.6 mm; rotation time 330 ms; pitch 0.2-0.5; and slice thickness 0.75 mm. The contrast transit time was estimated by the test bolus tracking technique.
The CCTA findings were analyzed on a workstation (ZIO STATION, ZIO soft, Tokyo, Japan) by one cardiologist and one radiologist, and evaluated using the axial, multi-planar reconstruction, volume rendering, and maximum intensity projection techniques. The following points were evaluated on CCTA: entry morphology (tapered or blunt), lesion length (mm), and calcification degree in the cross-sectional image (<50% or ≥ 50%), ostial or bifurcation lesion, lesion tortuosity (<45% or ≥ 45%), and distal end morphology (presence or absence of bifurcation). If calcification was present in the CTO lesion, the distribution of calcification and calcification degree were evaluated in detail.
Coronary angiography and intervention
All the enrolled patients underwent CAG before CTO PCI, and we developed strategies for CTO PCI based on the CAG and CCTA findings. The morphology of the CTO distal end (presence or absence of bifurcation), quality of CTO distal end visuality, and renal function were important factors associated with the choice of the first approach for CTO (primary antegrade approach or primary bidirectional approach) in our hospital. If the CTO distal end included bifurcation or showed poor visualization on contrast, and the patient had renal insufficiency, with an estimated glomerular filtration rate <30 ml/min/1.73 m2, we considered the primary bidirectional approach. The primary approach was finally selected according to the operator’s preference.
Angiographical CAC was defined as the presence of radiopacities within the vascular wall during some cardiac cycles before contrast injection [18]; additionally, we defined it as the detection of radiopacities from at least two different angles on CAG. Six experienced cardiologists who were blinded to the clinical data and CTO PCI results performed the angiographical CAC evaluations.
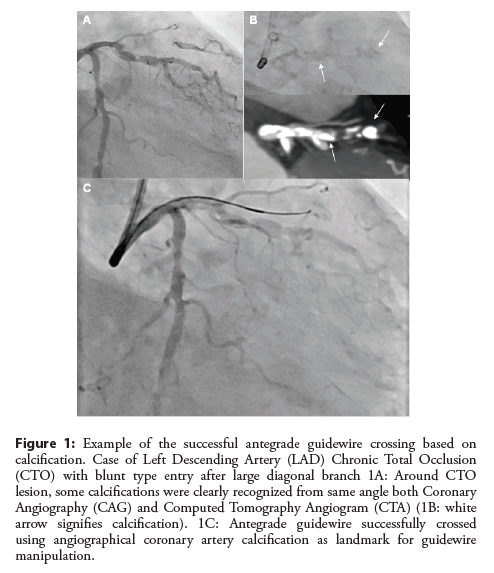
CTO PCI was performed by six operators. One of the operators was a CTO PCI expert; he provided assistance with the CTO PCI procedures performed by the other operators too. The distribution of calcification on CCTA and angiography and the vessel course on CCTA were used as references in guidewire crossing (Figure 1). Guidewire crossing was deemed successful when it crossed the lesion into the distal true lumen. Procedural success was defined as a post-PCI TIMI flow grade of 3 and residual stenosis degree <30%.
Figure 1: Example of the successful antegrade guidewire crossing based on calcification. Case of Left Descending Artery (LAD) Chronic Total Occlusion (CTO) with blunt type entry after large diagonal branch 1A: Around CTO lesion, some calcifications were clearly recognized from same angle both Coronary Angiography (CAG) and Computed Tomography Angiogram (CTA) (1B: white arrow signifies calcification). 1C: Antegrade guidewire successfully crossed using angiographical coronary artery calcification as landmark for guidewire manipulation.
Statistical analysis
All statistical analyses were performed with JMP version 15.1.0 (SAS Institute, Cary, North Carolina, US). Quantitative variables were expressed as mean ± standard deviation and qualitative variables as percentages. Chi-square tests were used for categorical variables, and Student’s T tests and Wilcoxon tests for continuous variables. For the identification of factors predictive of successful antegrade guidewire crossing, the patients’ demographic and cardiovascular risk factors, clinical status, and CCTA and angiographic findings were examined in the univariate analysis. Multivariate logistic regression analyses were used for the selection of factors correlated with successful antegrade guidewire crossing. Variables entered into the multivariable model were those that reached significance (p<0.10) following univariate logistic regression analysis and were deemed clinically significant. A p value <0.05 was considered statistically significant.
Results
Clinical characteristics and procedural outcomes
Between May 2016 and December 2017, 74 patients with 76 CTO lesions were screened. We excluded 11 CTO cases owing to in-stent occlusion, two due to the unavailability of pre-procedural CCTA data, and 13 in whom the primary retrograde approach was planned. Finally, 48 patients with 50 CTO lesions in whom the primary antegrade approach was planned were enrolled in this study (Figure 2).
The patients’ baseline clinical characteristics and procedural outcomes are shown in Table 1. The mean patient age was 67.9 ± 12.2 years, and 82% of the patients were men. There was no significant difference between the success and unsuccess groups in terms of hypertension, diabetes mellitus (glycated hemoglobin >8.0%), low-density lipoprotein cholesterol, current smoking, and hemodialysis.
| Overall | Success group (n=25) | Unsuccess group (n=25) | p value | |
|---|---|---|---|---|
| Baseline characteristics | ||||
| Age, years | 67.9 ± 12.2 | 66.0 ± 13.9 | 69.9 ± 10.1 | 0.299 |
| Body mass index, kg/m2 | 24.9 ± 3.5 | 24.8 ± 4.1 | 25.0 ± 3.0 | 0.265 |
| Male, n(%) | 41(82) | 22(88) | 19(76) | 0.265 |
| Hypertension, n(%) | 28(56) | 11(44) | 17(68) | 0.11 |
| Diabetes mellitus (HbA1c >8.0%), n(%) | 9(18) | 3(12) | 6(24) | 0.185 |
| Insulin required, n (%) | 5(10) | 2(8) | 3(12) | 0.664 |
| LDL-C, mg/dL | 88.2 ± 23.6 | 89.1 ±2 4.2 | 87.4 ± 23.4 | 0.784 |
| Statin, n(%) | 50(100) | 25(100) | 25(100) | 1 |
| Current smoker, n(%) | 13(26) | 8(32) | 5(20) | 0.377 |
| Hemodialysis, n(%) | 2(4) | 1(4) | 1(4) | 1 |
| LVEF, % | 59.9 ± 12.6 | 68.6 ± 8.4 | 56.1 ± 15.0 | 0.018 |
| eGFR, mL/min/1.73 m2 | 61.6 ± 18.2 | 58.5 ± 19.0 | 54.8 ± 14.7 | 0.018 |
| Previous Myocardial Infarction, n(%) | 26(52) | 10(40) | 16(64) | 0.137 |
| Previous PCI, n(%) | 36(72) | 15(60) | 21(84) | 0.083 |
| Previous CABG, n(%) | 8(16) | 2(8) | 6(24) | 0.162 |
| Reattempt, n(%) | 5(10) | 2(8) | 3(12) | 0.574 |
| Procedural characteristics | ||||
| Overall procedure success, n(%) | 44(88) | 25(100) | 19(76) | 0.01 |
| Antegrade procedure success, n(%) | 38(76) | 25(100) | 13(52) | <0.001 |
| Antegrade wire cross, n(%) | 39(78) | 25(100) | 14(56) | <0.001 |
| Procedure time, min | 210 ± 97 | 158 ± 78 | 263 ± 87 | <0.001 |
| Guidewire crossing time, min | 66 ± 65 | 21 ± 8 | 118 ± 63 | <0.001 |
| Air kerma, Gy | 6.5 ± 3.9 | 4.6 ± 3.4 | 8.3 ± 3.4 | <0.001 |
| Fluoroscopic time, min | 88 ± 47 | 63 ± 39 | 113 ± 42 | <0.001 |
Values are numbers (%) or mean ± SD. Abbreviations: LDL-C: Low-Density Lipoprotein-Cholesterol; LVEF: Left Ventricular Ejection Fraction; eGFR: estimated Glomerular Filtration Rate; PCI: Percutaneous Coronary Intervention; CABG: Coronary Artery Bypass Grafting.
Table 1: Baseline clinical and procedural characteristics of overall and between success to cross guidewire ≤ 30 min and failure groups.
The overall procedure success rate was 88% (100% vs. 76%, p=0.010). In the unsuccess group, 14 of 25 patients (56%) showed successful antegrade guidewire crossing after over 30 min. Of the 11 cases (44%) with antegrade guidewire crossing failure, six switched to the retrograde approach and recanalization success was finally achieved; the remaining five (20%) patients exhibited complete antegrade and retrograde approach as well as procedure failure. The overall procedure success rate (100% vs. 76%, p=0.010), antegrade procedure success rate (100% vs. 52%, P<0.001), and antegrade guidewire crossing rate (100% vs. 56%, P<0.001) were higher in the success group than in the unsuccess group.
CCTA and angiographic CTO lesion characteristics
The CCTA and angiographic CTO lesion characteristics are shown in Table 2. The CTO was located in the Right Coronary Artery (RCA) in 42% of the CTO lesions, Left Descending Artery (LAD) in 30%, and Left Circumflex Artery (LCX) in 28%. The number of RCA CTO lesions was higher in the unsuccess group and that of LAD CTO lesions was higher in the success group. Angiographical CAC was more frequently observed in the success group than in the unsuccess group (68% vs. 24%, p=0.080).
| Overall | Success group (n=25) | Unsuccess group (n= 25) | p value | |
|---|---|---|---|---|
| Target vessel | 0.175 | |||
| RCA, n(%) | 21(42) | 5(20) | 16(64) | |
| LAD, n(%) | 15(30) | 14(56) | 1(4) | |
| LCX, n(%) | 14(28) | 6(24) | 8(32) | |
| Angiographical characteristics | ||||
| Tapered type entry, n(%) | 25(50) | 13(52) | 12(48) | 0.746 |
| Ostial lesion, n(%) | 16(32) | 10(40) | 6(24) | 0.212 |
| Micro channel, n(%) | 17(34) | 8(20) | 9(36) | 0.802 |
| Bridging collateral, n(%) | 9(18) | 4(16) | 5(20) | 0.664 |
| Calcification, n(%) | 23(46) | 17(68) | 6(24) | 0.08 |
| Proximal tortuosity (≤ 45°), n(%) | 44(88) | 23(92) | 21(84) | 0.162 |
| CCTA lesion characteristics | ||||
| Distal reference diameter, mm | 2.87 ± 0.64 | 2.87 ± 0.62 | 2.88 ± 0.67 | 0.845 |
| Bending in CTO (≤ 45°), n(%) | 46(92) | 23(92) | 23(92) | 1 |
| Occlusion length (≤ 20 mm), n(%) | 29(58) | 17(68) | 12(48) | 0.134 |
| Calcification degree | 0.509 | |||
| None, n(%) | 8(16) | 5(20) | 3(12) | |
| Cross-section ≤ 50%, n(%) | 25(50) | 12(48) | 13(52) | |
| Cross-section >50%, n(%) | 17(34) | 8(32) | 9(36) | |
Values are numbers (%) or mean ± SD. Abbreviations: LDL-C: Low-Density Lipoprotein-Cholesterol; LVEF: Left Ventricular Ejection Fraction; eGFR, estimated Glomerular Filtration Rate; PCI: Percutaneous Coronary Intervention; CABG: Coronary Artery Bypass Grafting.
Table 2: Angiographical and CCTA lesion characteristics of overall and between success to cross guidewire ≤ 30 min and failure groups.
In the angiographical analysis, there were no statistically significant differences in terms of tapered type entry, microchannel, and bridging collateral. Ostial lesion and proximal tortuosity were more frequently observed in the success group than the unsuccess group, but the difference was not statistically significant. In the CCTA lesion analysis, there were no statistically significant differences in the distal reference diameter of the CTO lesion, vessel bending in the CTO lesion, occlusion length (≤ 20 mm), and calcification degree, as analyzed by CCTA cross-sectional images, between the groups.
Multivariate analysis of successful antegrade guidewire crossing
The results of the multivariate analysis of successful antegrade guidewire crossing are shown in Table 3. In the multivariate model, angiographical CAC (Odds Ratio [OR] =0.043, 95% Confidence Interval [Cl] =0.003–0.614, p=0.006) and left ventricular ejection fraction (OR=0.388, 95% Cl=0.151–0.997, p=0.013) were independent predictors of successful antegrade guidewire crossing within 30 min.
| Odds Ratio | 95% CI | p Value | |
|---|---|---|---|
| Angiographical CAC | 0.043 | 0.003-0.614 | 0.006 |
| LVEF | 0.388 | 0.151-0.997 | 0.013 |
| eGFR | 0.685 | 0.421-1.114 | 0.098 |
CAC: Coronary Artery Calcification; LVEF: Left Ventricular Ejection Fraction; eGFR, estimated Glomerular Filtration rate; 95% CI, 95% confidence interval.
Table 3: Multivariate analysis for successful antegrade guidewire crossing.
Discussion
The major findings of the present report are as follows: 1) Angiographical CAC was found more frequently in the success group than in the unsuccess group; 2) in the CCTA analysis, CAC severity was not related to the rate of antegrade guidewire crossing success; and 3) lesion morphology, except that pertaining to angiographical CAC, was not significantly different between the success and unsuccess groups. To the best of our knowledge, this study is the first to examine the relationship between angiographical CAC and the rate of antegrade guidewire crossing success.
In a previous study, CAC was identified as a predictor of failed CTO PCI and several scoring systems focusing on the success rate of CTO PCI reported that CAC was predictive of CTO PCI failure [14,15,17]. The J-CTO score is among the objective indices associated with CTO PCI procedure-related difficulties [17]. In this score, angiographical CAC is a negative predictor of successful guidewire crossing within 30 min. However, expert operators state that angiographical CAC is among the promising landmarks for antegrade guidewire manipulation. There are some discrepancies pertaining to the effect of CAC in CTO PCI between that observed in previous studies and that noted by operators. At the time of antegrade guidewire manipulation for guidewire crossing, it is important to set promising landmarks indicative of vessel course and the CTO distal true lumen. Previous coronary artery stent, contrasted distal true lumen, and guidewire via collateral channel are promising vessel landmarks. While angiographical CAC is a promising landmark, no previous studies have provided clarity on the same. We used angiographical CAC as the landmark for antegrade guidewire manipulation in CTO PCI; therefore, our finding that the success rate of antegrade guidewire crossing within 30 min is higher in cases with angiographical CAC than those with non-angiographical CAC demonstrates that angiographical CAC is a promising landmark for antegrade guidewire manipulation. In this study, we analyzed all the patients’ CCTA findings before the CTO PCI procedure. It is not possible to obtain accurate information on CAC location, distribution, and characteristics only through angiographical analysis as the CTO lesion cannot be viewed on angiography. On the other hand, CCTA is a suitable imaging modality for the analysis of the CTO lesion and significantly increase the success rate of CTO PCI [19]. CCTA is a tool necessary for CAG antegrade guidewire manipulation.
In this study, CAC severity, as estimated by CCTA analysis, was not related to the success rate of antegrade guidewire crossing. A previous study showed that severe CAC, particularly “full-moon” shaped 360° calcification, is one of the obstacles associated with CTO PCI performance [4,20]. “Full-moon” calcification is an extremely significant obstacle in guidewire crossing, and CTO guidewires may easily enter the subintimal or extravascular space as a result of its avoidance. Our study did not enroll any patient with real “full-moon” calcification on CCTA; therefore, we could not evaluate the effect of “full-moon” calcification on the success rate of antegrade guidewire crossing. However, it is a fact that the presence of heavy calcification occupying the central lumen is a significant obstacle in antegrade guidewire crossing.
In the J-CTO score [17], blunt type entry, angiographical CAC, bending degree >45°, and occlusion length ≧20 mm are independent predictors of unsuccessful guidewire crossing within 30 min. In this study, there was no significant lesion morphologyrelated difference, except in terms of angiographical CAC, between the success and unsuccess groups. The presence of a non-tapered type entry, similar to blunt type entry, and ostial lesions leads to difficulties in the positioning of the guidewire at the correct CTO entry point and increases the chance of the guidewire entering the subintimal space. Additionally, the proximal cap ambiguity of the blunt type entry and ostial lesion is an obstacle to the achievement of antegrade guidewire crossing success. In the Asia Pacific (AP)- CTO algorithm [21], if ambiguity of the proximal cap is present, Intravascular Ultrasound (IVUS) guidance entry penetration is recommended. In our procedure for blunt type cases, we inserted the IVUS catheter in the branch around the CTO entry and manipulated the guidewire correctly while viewing the CTO entry using IVUS. This is among the reasons for the absence of a significant difference in the entry type between the success and unsuccess groups. In the CCTA analysis, we were able to accurately estimate the CTO lesion bending degree and occlusion length, and also confirm the relationship between CAC and CTO lesions. In the presence of angiographical CACs around the bending portion of the CTO, we controlled and manipulated the guidewire so as to follow the vessel bend using the angiographical CAC as a landmark. If angiographical CAC was present in the long CTO lesion, the performance of guidewire manipulation using the angiographical CAC as a landmark enabled intra-vessel tracking. Therefore, the presence of angiographical CAC may prove advantageous in antegrade guidewire crossing among patients with a long CTO and severely bent CTO.
This study had several limitations. First, it had a single-center, retrospective, observational design, and the sample size was very small; therefore, unknown factors may have affected our results. Second, a very small number of patients underwent hemodialysis, which is strongly related to severe calcification. Third, as this study did not include patients with severe CAC, particularly those with “full-moon” calcification, we could not assess all types of calcifications.
Conclusion
Angiographical CAC may be a promising landmark for antegrade wire manipulation in patients with CTO.
Funding
This research received no external funding.
Competing interests
The authors declare no competing interests.
References
- Fefer P, Knudtson ML, Cheema AN, et al. Current perspectives on coronary chronic total occlusions: the Canadian Multicenter Chronic Total Occlusions Registry. J Am Coll Cardiol. 59(11): 991-997 (2012).
- Råmunddal T, Hoebers LP, Hoebers L, et al. Chronic total occlusions in Sweden: A report from the Swedish Coronary Angiography and Angioplasty Registry (SCAAR). PLoS One. 9(8): e103850 ((2014)).
- Tomasello SD, Boukhris M, Giubilato S, et al. Management strategies in patients affected by chronic total occlusions: Results from the Italian Registry of Chronic Total Occlusions. Eur Heart J. 36(45): 3189-3198 (2015).
- Suzuki Y, Tsuchikane E, Katoh O, et al. Outcomes of percutaneous coronary interventions for chronic total occlusion performed by highly experienced Japanese specialists: The first report from the japanese CTO-PCI Expert Registry. JACC Cardiovasc Interv. 10(21): 2144-2154 (2017).
- Tajti P, Karmpaliotis D, Alaswad K, et al. The hybrid approach to chronic total occlusion percutaneous coronary intervention: update from the PROGRESS CTO Registry. JACC Cardiovasc Interv. 11: 1325-1335 (2018).
- Morino Y, Kimura T, Hayashi Y, et al. In-hospital outcomes of contemporary percutaneous coronary intervention in patients with chronic total occlusion insights from the J-CTO Registry (Multicenter CTO Registry in Japan). JACC Cardiovasc Interv. 3(2): 143-151 (2010).
- Surmely JF, Tsuchikane E, Katoh O, et al. New concept for CTO recanalization using controlled antegrade and retrograde subintimal tracking: the CART technique. J Invasive Cardiol. 18(7): 334-338 (2006).
- Tsuchikane E, Katoh O, Kimura M, et al. The first clinical experience with a novel catheter for collateral channel tracking in retrograde approach for chronic coronary total occlusions. JACC Cardiovasc Interv. 3(2): 165-171 (2010).
- Rathore S, Katoh O, Matsuo H, et al. Retrograde percutaneous recanalization of chronic total occlusion of the coronary arteries: Procedural outcomes and predictors of success in contemporary practice. Circ Cardiovasc Interv. 2(2): 124-132 (2009).
- Kimura M, Katoh O, Tsuchikane E, et al. The efficacy of a bilateral approach for treating lesions with chronic total occlusions the CART (Controlled Antegrade and Retrograde Subintimal Tracking) registry. JACC Cardiovasc Interv. 2(11): 1135-1141 (2009).
- Yamane M, Muto M, Matsubara T, et al. Contemporary retrograde approach for the recanalisation of coronary chronic total occlusion: On behalf of the Japanese Retrograde Summit Group. EuroIntervention. 9(1): 102-109 (2013).
- Luo C, Huang M, Li J, et al. Predictors of Interventional Success of Antegrade PCI for CTO. JACC Cardiovasc Imaging. 8(7): 804-813 (2015).
- Martín-Yuste V, Barros A, Leta R, et al. Factors determining success in percutaneous revascularization of chronic total coronary occlusion: Multidetector computed tomography analysis. Rev Esp Cardiol (Engl Ed). 65(4): 334-340 (2012).
- Opolski MP, Achenbach S, Schuhbäck A, et al. Coronary computed tomographic prediction rule for time-efficient guidewire crossing through chronic total occlusion: Insights from the CT-RECTOR multicenter registry (Computed Tomography Registry of Chronic Total Occlusion Revascularization). JACC Cardiovasc Interv. 8(2): 257-267 (2015).
- Yu CW, Lee HJ, Suh J, et al. Coronary computed tomography angiography predicts guidewire crossing and success of percutaneous intervention for chronic total occlusion: Korean multicenter CTO CT Registry Score as a tool for assessing difficulty in chronic total occlusion percutaneous coronary intervention. Circ Cardiovasc Imaging. 10(4): e005800 (2017).
- Galassi AR, Werner GS, Boukhris M, et al. Percutaneous recanalisation of chronic total occlusions: 2019 consensus document from the EuroCTO Club. EuroIntervention. 15(2): 198-208 (2019).
- Morino Y, Abe M, Morimoto T, et al. Predicting successful guidewire crossing through chronic total occlusion of native coronary lesions within 30 minutes: The J-CTO (Multicenter CTO Registry in Japan) score as a difficulty grading and time assessment tool. JACC Cardiovasc Interv. 4(2): 213-221 (2011).
- Mintz GS, Popma JJ, Pichard AD, et al. Patterns of calcification in coronary artery disease. A statistical analysis of intravascular ultrasound and coronary angiography in 1155 lesions. Circulation. 91(7): 1959-1965 (1995).
- Rolf A, Werner GS, Schuhbäck A, et al. Preprocedural coronary CT angiography significantly improves success rates of PCI for chronic total occlusion. Int J Cardiovasc Imaging. 29(8): 1819-1827 (2013).
- Opolski MP, Achenbach S. CT angiography for revascularization of CTO: Crossing the borders of diagnosis and treatment. JACC Cardiovasc Imaging. 8(7): 846-858 (2015).
- Harding SA, Wu EB, Lo S, et al. A new algorithm for crossing chronic total occlusions from the Asia Pacific Chronic Total Occlusion Club. JACC Cardiovasc Interv. 10(21): 2135-2143 (2017).