Case Report - Diabetes Management (2018) Volume 8, Issue 4
A physician-patient’s perspective on lowering glycemic variability–Part III: the role of lifestyle
- *Corresponding Author:
- Elsamma Chacko
Connecticut Valley Hospital
1000 Silver Street, Middletown, CT 06457, USA
E-mail: elsammac@msn.com
Abstract
A physician with type 2 diabetes for 19 years and impaired awareness of hypoglycemia sought to lower the hypoglycemia risk with the help of continuous glucose monitoring. The idea was to optimize the medications-meals-exercise triad. When the patient adopted a personalized low carb, balanced meal plan, glargine insulin dose came down from 36 units to 18. The meal plan called for eating every 2-4 hours. Also, more carbohydrate ingestion during early part of the day lowered glycemic variability. Insulin dose came down further to 7 units when a third medication, dulaglutide, was added to metformin and insulin. In the course of this lifestyle modification the patient made a remarkable observation: although four glycogen depleting exercises, followed by a brief walk offered comparable immediate glycemia benefits, the effect extending significantly to the next day was seen only with the split exercise (pre-breakfast+ post-breakfast walk). Several exercise combinations were identified for lowering glycemic variability. Weight, HbA1c and lipids have been moving in the right direction with the new lifestyle.
Keywords
hypoglycemia, pre-breakfast walk, mid-postprandial walk, fasting glucose
Abbreviations
IAH–impaired awareness of hypoglycemia, CGM–continuous glucose monitor, AE–aerobic exercise, RE–resistance exercise, HIIE–high intensity interval exercise, St Ex–stair exercise, FBG–fasting blood glucose, PPG–postprandial glucose, Pre-post Ex (Split Ex)–pre-meal walk+post-meal walk.
Introduction
Lowering glycemic variability is thought to be more beneficial than lowering HbA1c or fasting blood glucose for moderating diabetes complications [1]. Lowering glycemic variability also meant less hyperglycemia and hypoglycemia. The net, real-time response of blood glucose levels to medications, meals and exercise activities can be complex. The glucose response is determined by the interplay among a large number of variables, including the state of diabetes, type and dosage of medications, meal timing and meal composition, and timing, intensity, duration and sequence of exercise. It is challenging to sort out the effects of meal and exercise on glucose response under free-living conditions. Although an impressive body of results has accumulated on glycemia benefits of meal composition and meal timing, the latter has not made it into the guidelines yet [2]. Similarly, the effect of exercise timing is not mentioned in the guidelines [2]. Translational efforts are exceedingly slow in this area. On the other hand, most of the factors affecting glycemia can be kept constant when a single subject explores the glucose response to a variety of exercise options with the help of continuous glucose monitoring (CGM). Part I of this series reported the role of meal timing in lowering glycemic variability [3]. Part II reported immediate glycemia benefits of four exercise options [4]. This paper, Part III, sheds some light on the short term and long term effects of the new lifestyle on metabolic markers. This patient, who has been living with T2D for 19 years now, also developed impaired awareness of hypoglycemia (IAH) [5-7]. After a second seizure episode while driving to work, some 2½ h after a resistance exercise, her endocrinologist recommended CGM (Dexcom G5) to adjust the medications-meals-exercise triad with a view to reducing the hypoglycemia risk. She was on metformin 1 gm twice a day and glargine insulin 36 units once a day when she had the first seizure incident which came after not eating for eight h on a busy day. Insulin dose came down to 18 units when she started a breakfast-centered low-carb (75-90 gm carbohydrates) balanced meal plan which called for eating every 3-4 h [8]. After the second seizure episode the patient would eat within two h after RE. The insulin dose came down further to 7 units when dulaglutide, 0.75 mg/week, was added to the med regimen. Keeping the same med regimen and meal plan she explored different exercise options.
▪ Short term glycemia benefits of four exercise options
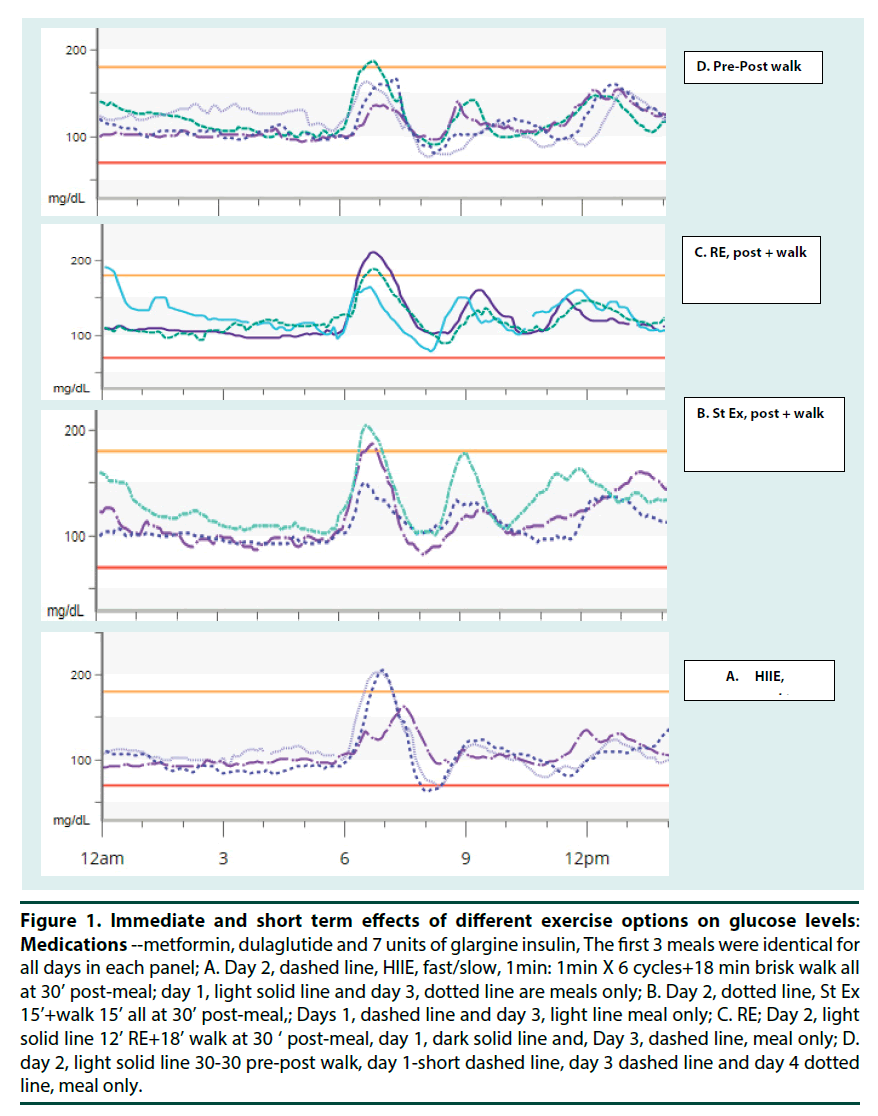
Immediate glycemia benefit of four glycogen-depleting exercises followed by a short walk has been reported as more effective in lowering the post-meal glucose surge than a post-meal walk alone [4]. Short term effects of these four exercise modalities on glucose levels are shown in (Figures 1A-1D). Here, the exercise was done on the second day: the first, third and fourth day were meal-only days. The first 3 meals were identical in each panel. Breakfast and second meal included ½ cup toasted oats, ½ cup sliced strawberries and ½ cup 1% milk, totaling 1½ carbs (22.5 gm carbohydrates) per meal. Lunch was a cup of split pea vegetable soup containing ~1 carb (15 gm carbohydrates). Evening snack and supper contained ¾ carb each. The main finding was that although the immediate glycemia benefit was comparable in these three post-meal exercise options, Figures 1A-1C, the glycemia benefit extending to the next day was most impressive for the split exercise (Split Ex or 30-30 pre-post walk) where 30 min pre-meal walk was followed by a 30 min post-meal walk (Figure 1D).
Figure 1. Immediate and short term effects of different exercise options on glucose levels: Medications --metformin, dulaglutide and 7 units of glargine insulin, The first 3 meals were identical for all days in each panel; A. Day 2, dashed line, HIIE, fast/slow, 1min: 1min X 6 cycles+18 min brisk walk all at 30’ post-meal; day 1, light solid line and day 3, dotted line are meals only; B. Day 2, dotted line, St Ex 15’+walk 15’ all at 30’ post-meal,; Days 1, dashed line and day 3, light line meal only; C. RE; Day 2, light solid line 12’ RE+18’ walk at 30 ‘ post-meal, day 1, dark solid line and, Day 3, dashed line, meal only; D. day 2, light solid line 30-30 pre-post walk, day 1-short dashed line, day 3 dashed line and day 4 dotted line, meal only.
▪ Mid-postprandial exercise
Although high intensity exercises during mid-postprandial period offer some glycogen depletion, there is considerable glycogen sparing [9]. This is because exogenous glucose is readily available for most of the energy needs and insulin levels are high. The glycogen sparing during mid-postprandial exercise might explain why some glycogen repletion is there but the insulin sensitivity improvement lasted only for a few hours. Although glucose levels come down with post-meal physical activities the best timing for lowering glucose surge is to start the activity 30 min before the anticipated glucose peak [10- 19]. A 45-60 min brisk walk or yard work or a short bout of HIIE [20], RE [21] or St Ex [22] followed by a brief walk, at 25-30 min post-breakfast, do lower glucose surge after meal. Lunch and supper may be more balanced with protein, fiber, non-starchy vegetables and healthy fat offering a delayed glucose peak compared to breakfast. In this case, the starting time of the exercise may have to be delayed accordingly. Since insulin sensitivity improvement is short-lived, the main goal of post-meal exercise is to lower glucose surge in real time.
▪ Pre-meal exercise
Glycogen depletion (liver or muscle) is considerably more efficient when the exercise is done pre-breakfast. Here, the main fuels are endogenous glucose from liver glycogen, muscle glycogen and free fatty acids. The glycogen depletion is followed by glycogen repletion, which leads to insulin sensitivity improvement lasting 24 h or beyond [23-26]. Other benefits of exercise arising from molecular adaptations also seem more pronounced after pre-meal exercise. There is evidence that glycogen content and GLUT-4 protein levels are high when training is done under pre-meal conditions [27]. If muscle conditioning, body composition or performance improvement is the goal, training under pre-meal conditions is the way to go. Although a short bout of pre-meal exercise results in post-exertion glucose elevation [23,28-30] long duration pre-meal exercises do offer glycemia benefits [31-33] likely because of significant glycogen depletion. The patient noticed that short duration pre-meal exercises may be most beneficial provided a post-meal walk is also done to use up the extra glucose coming to the blood stream forming the post-exertion glucose elevation (Figure 1D and Figure 2D) light solid line. Hexhi and colleagues showed that lunch time split exercise was better than post-meal exercise in lowering the glucose surge of the meal [34].
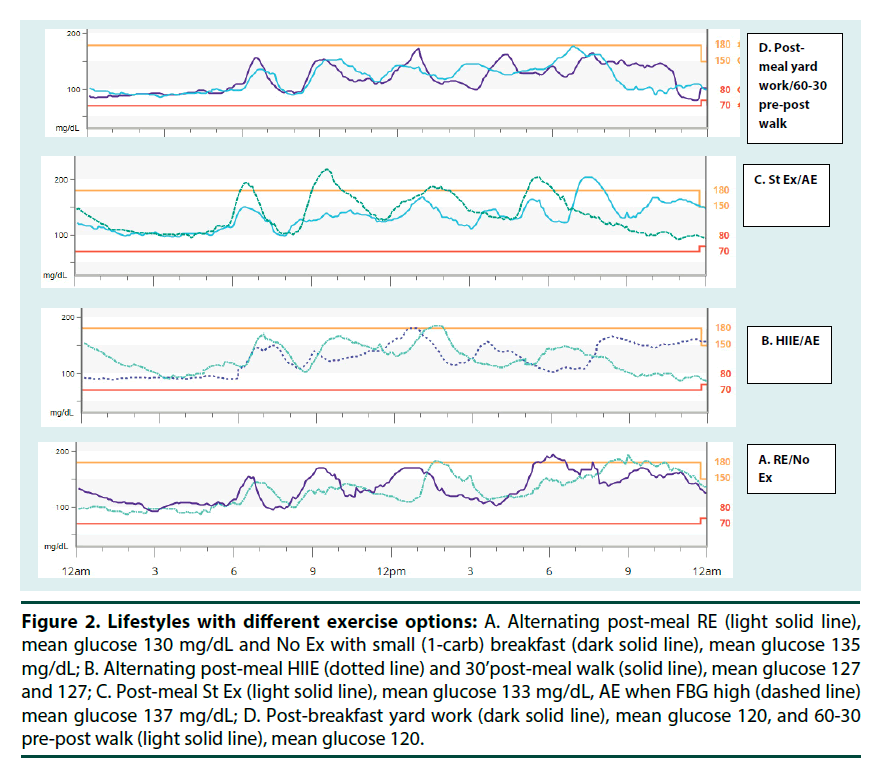
Figure 2. Lifestyles with different exercise options: A. Alternating post-meal RE (light solid line), mean glucose 130 mg/dL and No Ex with small (1-carb) breakfast (dark solid line), mean glucose 135 mg/dL; B. Alternating post-meal HIIE (dotted line) and 30’post-meal walk (solid line), mean glucose 127 and 127; C. Post-meal St Ex (light solid line), mean glucose 133 mg/dL, AE when FBG high (dashed line) mean glucose 137 mg/dL; D. Post-breakfast yard work (dark solid line), mean glucose 120, and 60-30 pre-post walk (light solid line), mean glucose 120.
▪ The new lifestyle options
The patient identified six exercise modalities, five post-meal options and the pre-post option as effective in lowering glycemic variability (Figures 2A, 1D). These were picked on the basis of various studies reported in the literature and the results seen in (Figures 1A-1D). Identical breakfast containing 1½ carbs (with an expected glucose peak of over 200 without exercise as in Figure 1 was used on days when exercise was done. In case no elective exercise could be done, a smaller breakfast (1-carb) was used. Carb count was the same for all other meals: 2-carbs for the second meal, 1 carb for the lunch and ¾ carb each for the pm snack and supper.
▪ Post-meal exercises
High-intensity exercise (RE/HIIE/St Ex) post-meal+ short walk: These brief post-breakfast exercises at 30 min post-meal may work for people who like to do some high intensity exercises. (Figure 2A), light solid line, shows the glucose profile when a 12 min RE was used. (Figure 2B) dotted line, shows glucose profile using 10 min HIIE and (Figure 2C) light solid line, is that for 15 min of St Ex. These exercise options may be most suited for working people on working days. The patient has been doing one of these, 3 times a week, Mondays, Wednesdays and Fridays. Tuesdays and Thursdays she may go without exercise or with a 30 min post-meal walk. Post-breakfast yard work or walk. One hour of light yardwork at 30 min post-breakfast lowered the glucose surge (Figure 2D, dark solid line). A 30 min post-breakfast walk was also effective, Figure 2B solid line. Yard work or post-meal walk may be done after every big meal. These options might work for retired people every day and also for working people on weekends.
▪ Pre-meal exercises
Pre-post walk, (30-30 or 60-30): This is probably the best exercise option for glycemia benefit and other physical health benefits for people who do not want to or cannot do high intensity exercises. Post-exertion glucose elevation might be moderated by doing a post-meal walk also after the pre-meal walk. Even when there is some post-exertion glucose elevation it is known to become progressively smaller with weekly training [35]. (Figure 1D and Figure 2D) light solid line show the glucose response to 30-30 and 60-30 pre-post walks respectively. The immediate and short term glycemia benefit makes it superior to the post-meal options, for overall glycemia benefit. Long duration pre-meal walk has been known to improve fasting glucose [31]. These pre-meal options may work for retired individuals every day and also for working people on weekends.
▪ Long duration pre-meal high-intensity exercises
This is based on the HIIE study [10] where the high-intensity component was for 16 minutes and the total exercise lasted for about an h. Although overall glycemia benefit was very impressive, this may not be practical for most diabetes patients. Those who like intense pre-meal exercise this option may be done 3 times a week alternating with a 30 min post-meal walks or no exercise. Other high-intensity exercises such as a 5-mile run in the morning before breakfast may work for some people. (This patient did not try this option).
▪ Fasting glucose
It should be noted that on many occasions, fasting glucose was high. Two of these are shown in Figure 2A, dark solid line (141 mg/dL) and Figure 2C, dashed line (116 mg/dL). In the former case, a small 1-carb breakfast was eaten (non-exercise day) hoping to correct the fasting glucose quickly. In the latter case, there was no glycemia benefit with a 30 min post-breakfast walk as reported earlier [4]. Here, the overall glycemia was worse for the 2nd meal also. There was strict carbohydrate restriction for the rest of the day and fasting glucose improved the day after (105 mg/dL).
▪ Blood tests
As different medications, meal plans and exercise options were explored with the help of CGM, A1c improved from 7.5 to 6.8 to 6.2 to 6.0%. Lipids also improved progressively: HDL went up from 48 to 50 to 52 mg/dL. TG went down from 76 to 66 to 57 mg/dL. LDL changed from 89 to 95 to 76 mg/dL. Eventually the patient got settled in to a steady lifestyle. Medications have been metformin 1000 mg PO two times a day, Dulaglutide 0.75 mg IM once a week and glargine insulin, 7 units sc, once a day. Meal plan has been 5 carbs a day for non-exercise days and 5½ carbs on exercise days: breakfast contains 1 carb and 1½ carbs respectively. The rest of the meals have been a 2 carb second meal, 1 carb lunch and ¾ carb pm snack and supper. Exercise options are high intensity options (HIIE/RE/ St Ex) Mondays, Wednesdays and Fridays, 30 min post-meal walk on Tuesdays and Thursdays and yard work or pre-post walks on weekends (Figure 2D). Lab tests are expected to improve as the state of diabetes gets better. Meal timing and timing, intensity, duration and sequence of exercise are important for lowering glycemic variability and these are factors the patient can control. There is a new thinking that diabetes management does not have to be glucocentric. The patient felt why not have low HbA1c and other markers using personalized lifestyle provided there is minimum hypoglycemia risk.
▪ Limitations
The results presented here are observed by a single patient, under free-living conditions. Although individual findings have been reproducible, these need to be confirmed in large scale conventional studies for accelerated translation.
Summary
▪ Minimizing hypoglycemia risk
Lowering insulin dose via adjusting medications-meals- exercise triad reduces hypoglycemia risk. Eating every 2-4 h prevents hypoglycemia. As far as hypoglycemia is concerned, physical activities during pre-breakfast and mid-postprandial periods may be undertaken safely.
▪ Fasting glucose
Fasting glucose has to be normal to achieve glycemia benefit from exercises. When fasting glucose is high postprandial glucose goes up and no glycemia benefit is seen with exercise. Strict carb control and a long pre-meal walk can correct fasting glucose in as little as 1-2 days.
▪ Post-meal exercises
Insulin sensitivity improvements are short-lived. The main purpose of post-meal exercises is to lower the post-meal glucose surge. Five options are identified here: A short bout of high-intensity exercise (RE/ HIIE/ St Ex) followed by a short walk, yard work and a 30-45 min walk. Glucose levels come down with post-meal physical activities but the best timing to lower glucose surge is to start the walk 30 min before the anticipated peak. Other activities such as dancing, swimming, hiking, step aerobics or zumba should work as long as the energy expenditure is appropriate: if high-intensity exercise is used, brief duration is safe.
▪ Pre-meal walks
Although post exertion glucose elevation is seen with pre-meal exercises glycogen depletion is very efficient and insulin sensitivity improvement is long lasting. The patient noticed that a 30 min post-meal walk following the pre-meal walk can moderate the post-exercise glucose elevation. This answers one drawback pre-meal exercise has namely the post-exertion glucose elevation. This option, however, may not be practical for most working people. The patient is convinced that the 30-30 pre-post option is worth the extra effort involved in incorporating it into her lifestyle. She is considering switching to it three times a week on weekdays for her next lifestyle. A 30-30 pre-post walk or a long duration pre-meal walk may offer glycemia benefit and other health advantages.
▪ Long duration high-intensity pre-meal exercise
According to literature data a long duration high intensity exercise pre-meal, such as 16 min of interval exercise (total 60 min physical activity) could also offer overall glycemia benefit. This may be done 3 times a week. But this level of intense activity may be difficult for most diabetes patients. (Other long duration pre-meal physical activities such as running, swimming, weight training might work too). Co-ordinating meal and exercise as described here can be valuable for managing, reversing or preventing diabetes and other chronic diseases.
Acknowledgement
The authors thank Jorge Munoz, RN, APRN for assistance with preparing the figures used in this report.
Author’s contribution
Elsamma Chacko: Literature search, study design, data collection, data interpretation; put together the first draft. Christine Signore: Endocrinologist caring for the patient, devised the treatment plan, ordered medications, CGM and lab tests, data interpretation and extensive review of the various drafts of the paper.
References
- Ceriello A, Kilpatrick E. Glycemic variability: both sides of the story. Diabetes. Care .36(2), S272–S275 (2013).
- Lifestyle Management: Standards of Medical Care in Diabetes – 2018. Diabetes.Care . 41(S1), s38–s50 (2018).
- Chacko E, Signore, C. A physician-patient’s perspective on lowering glycemic variability: Part I the role of meals. Diabetes. Manag. 8(3), 053–56 (2018).
- Chacko E, Signore, C. A physician-patient’s perspective on lowering glycemic variability: Part II, the role of exercise. Diabetes. Manag. 8(3), 57–61 (2018).
- Zqbal A, Heller S. The role of structured education in the management of hypoglycaemia. Diabetologia. 61(4), 751–760 (2018).
- Maran A, Pavan P, Bonsembiante B. Continuous glucose monitoring reveals delayed nocturnal hypoglycaemia after intermittent high-intensity exercise in non-trained patients with type 1diabetes. Diabetes. Technol. Ther .12(10), 763–768 (2010).
- Zoungas S, Patel A, Chalmers J et al. Severe hypoglycaemia and risks of vascular events and death. New. Engl. J. Med. 363, 1410–1418 (2010).
- Chacko E, Awruch P, Swartz E. Breakfast-centered meal plan for people with diabetes: a modest cohort study under free-living conditions. Diabete. Manag . 8(1), 32–37(2018).
- De Bock K, Derawe W, Ramaekers E et al. Fiber type-specific muscle glycogen sparing due to carbohydrate intake before and during exercise. J. Appl. Physiol. 102(1), 183–188 (2007).
- Nelson J, Poussier P, Marliss E et al.Metabolic response of normal man and insulin-infused diabetics to postprandial exercise. Am. J. Physiol. 242,E309–E316 (1982).
- Caron D, Poussier P, Marliss E et al. The effect of poastprandial execise on meal-related glucose intolerance in insulin-dependent diabetic individuals. Diabetes. Care. 5, 364–369 (1982).
- Shin Y, Jung H, Ryu J et al. Effects of pre-exercise meal on plasma growth hormone response and fat oxidation during walking. Prev. Nutr. Food. Sci. 18(3),175–180 (2013).
- Chacko E. A time for exercise: the exercise window. J. Appl. Physiol .122, 206–209 (2007).
- Erickson M, Little J, Gay J et al. Postmeal exercise blunts postprandial glucose excursions in people on Metformin therapy. J. Appl .Physiol. 123, 444–450 (2017).
- Van Dijk J, Venema M, Van Mechelen W et al. Effect of moderate-intensity exercise versus activities of daily living on 24-hourblood glucose homeostasis in Male patients with type 2 diabetes. Diabetes .Care. 36(11), 3448–3453 (2013).
- Kirwan J, O'Gorman D, Cyr-Cambell D et al. Effects of a moderate glycemic meal on exercise duration and substrate utilization. Med. Sci. Sports. Exerc. 33(9),1517–1523 (2001).
- Borror A, Zieff G, Battaglini C et al. The effects of postprandial exercise on glucose control in individuals with type 2 diabetes: a systematic review. Sports. Med. 48(6),1479–1491(2018).
- Shambrook P, Kingsley M, Wundersitz D et al. Glucose response to exercise in the post‐prandial period is independent of exercise intensity. Scand. J. Med. Sci. Sports. 28(3), 939–946. (2018).
- Reynolds A, Williams S, Venn B. Advice to walk after meals is more effective for lowering postprandial glycaemia in type 2 diabetes mellitus than advice that does not specify timing: a randomised crossover study. Diabetologia. 59(12), 2572–2578 (2016).
- Gillen J, Little J, Punthakee Z et al. Acute high-intensity interval exercise reduces postprandial glucose response and prevalence of hyperglycemia in patients with type 2 diabetes. Diabetes. Obes. Metab. 14(6), 575–577 (2012).
- Heden T, Winn N, Mari A et al. Post-dinner resistance exercise improves postprandial risk factors more effectively than pre-dinner resistance exercise in patients with type 2 diabetes. J.Appl .Physiol. 118(5), 624–34 (2014).
- Takaishi T, Hayashi T. Stair Ascending-descending exercise accelerates the decrease in postprandial hyperglycemia more efficiently than bicycle exercise. BMJ. Open. Diab. Res. Care. 5, e000428 (2017).
- Kjaer M, Hollenbeck C, Frey-Hewitt B et al. Glucoregulation and hormonal responses to maximal exercise in non-insulin-dependent diabetes. J.Appl. Physiol. 68, 2067–2074 (1990).
- Oberlin D, Mikus C, Kearney M et al. One bout of exercise alters free-living postprandial glycemia in type 2 diabetes. Med .Sci. Sports. Exerc. 46(2), 232–238 (2014).
- Francois M, Baldi J, Manning P et al. Exercise snacks' before meals: a novel strategy to improve glycaemic control in individuals with insulin resistance. Diabetologia 57(7), 1437–1445 (2014).
- Perseghin G, Price T, Peterson K et al. Increased glucose Transport-phosphorilation and muscle glycogen synthesis after exercise training in insulin-resistant subjects. N.Engl .J. Med. 335(18), 1357-1362 (1996).
- Nybo L, Pedersen K, Christensen B et al. Impact of carbohydrate supplementation during endurance training on glycogen storage and performance. Acta. Physiol. 197(2), 117–127 (2009).
- Derave W, Mertens A, Muls E et al. Effects of post-absorptive and postprandial exercise on glucoregulation in metabolic syndrome. Obesity 15(3), 704–711 (2007).
- Colberg S, Zarrabi L, Bennington L et al. Post-prandial walking is better for lowering the 6-8,14,15]glycemic effect of dinner than pre-dinner exercise in type 2 diabetic individuals. J .Am. Med. Dir. Assoc. 10(6), 394–397 (2009).
- Di Pietro L, Gribok A, Stevens M et al.Three 15-min bouts of moderate post-meal walking significantly improves 24-h glycemic control in older people at risk for impaired glucose tolerance. Diabetes. Care. 36(10), 3262–3268 (2013).
- Borer K, Wuorinen E, Lukos J et al.Two bouts of exercise before meals, but not after meals, lower fasting blood glucose. Med. Sci. Sports. Exerc. 41,1606–1614 (2009).
- Terada T, Wilson B, Myette C et al. Targeting specific interstitial parameters with high-intensity interval exercise and fasted-state exercise in type 2 diabetes. Metabolism. 65(5), 599-60 (2016).
- Hatamoto Y, Yoshimura E, Yamada Y et al. Effect of exercise timing on elevated postprandial glucose levels. J. Appl. Physiol. 123(2), 278–284 (2017).
- Haxhi1 J, Leto G, di Palumbo AS et al. Exercise at lunchtime: effect on glycemic control and oxidative stress in middle aged men with type 2 diabetes. Eur. J. Appl. Physiol. 116(3), 573-82 (2016).
- Harmer A, Chisholm D, McKenna M et al. High-intensity training improves plasma glucose and acid-base regulation during intermittent maximal exercise in type 1diabetes. Diabetes. Care. 30(5),1269–1271 (2007).