Case Report - International Journal of Clinical Rheumatology (2017) Volume 12, Issue 4
Association of sarcoidosis and psoriasis: a difficult diagnosis
- *Corresponding Author:
- Houda Sahraoui
Rheumatology Service
Hospital Arrazi
CHU Mohammed VI, Marrakech, Morocco
E-mail: fmpm2006.hs@gmail.com
Abstract
Sarcoidosis is a chronic multisystem disorder characterized by the formation of non caseating epithelioid cell granulomas affecting multiple organ systems. The role of the type 1 helper T (TH1) cells and type 17 helper T (TH17) cells in sarcoidal granuloma formation has been well documented. Moreover, TH17 and TH1 cells are also known to be involved in the pathogenesis of psoriasis. Thus, the concomitance of sarcoidosis and psoriasis is plausible based on these shared underlying immunological pathways. We report a case of a 46-years-old female with concomitant sarcoidosis and psoriasis, who has been misdiagnosed with psoriatic arthritis for a long time.
Keywords
sarcoidosis, psoriasis, psoriatic arthritis, TH1 lymphocytes, TH17 lymphocytes
Introduction
Sarcoidosis is a multi-system granulomatosis of unknown cause. It affects most commonly the lungs, the joints in less than 5% of the cases [1] and the skin, usually with erythema nodosum and small, medium and large sarcoids nodules. In this report, the authors described the association between skin psoriasis and sarcoidosis which joint manifestations has been mistakenly attributed to a psoriatic arthritis.
Observation
Mrs. A.E., aged 46, with a history of skin and nail psoriasis since 1992. In 2009, she complained of chronic inflammatory bilateral asymmetrical polyarthralgia involving the metacarpophalangeal, the proximal interphalangeal, the distal interphalangeal, the wrists and knees joints. She was also bothered by inflammatory low back pain without buttock pain or heel pain. All these symptoms were accompanied with a dry mouth, a grade 2 dyspnea and a history of a recurrent painful red eye for the last year (3 episodes per year).
Paraclinical investigations have showed a discreet biological inflammatory syndrome with a lymphopenia at 600/mm3. Tests for HLA B27, rheumatoid factors, Human Immunodeficiency Virus (HIV) and hepatitis serologies were negative.
She had no radiological signs of sacroiliitis, nor syndesmophytes or enthesitis, and the sacroiliac and lumbar spine MRI came back normal. The thoracic CT scan identified mediastinal hilar lymphadenopathy, without pulmonary infiltrate. Bronchoscopy and biopsy were in favor of a nonspecific lymphadenitis. The Pathergy test, the intradermo-reaction to tuberculin and the culture sputum for tuberculosis were negative. The serum levels of Lactates Dehydrogenase (LDH), β2 microglobulin were normal. Calcium and Phosphor levels in blood and in a 24 h urine sample were normal too. However, the Angiotensin Converting Enzyme level was at the upper limits of normal.
The accessory salivary gland biopsy found a nonspecific chronic sialadenitis grade 2. The ophthalmological examination showed a signs of an anterior non granulomatous uveitis with inflammatory cells.
Due to the involvement of the axial and peripheral joints on one hand, and the dermatological lesions on the other, we diagnosed her with psoriatic arthritis. The patient received 15 mg per week of Methotrexate and used a topical treatment for the psoriatic lesions. This resulted in a complete clearance of these lesions, and a partial improvement of low back pain and arthralgia. However, the uveitis recurrence got up to 8 episodes per year.
In 2016, the patient was hospitalized again for a new polyarthitis episode. She had inflammatory polyarthralgia involving the metacarpophalangeal, the proximal interphalangeal, the distal interphalangeal, the wrists, the knees and the ankles. They were accompanied with an inflammatory back pain, heel pain, and psoriatic lesions. Her Bath Ankylosing Spondylitis Disease Activity Index (BASDAI) was at 74/100 and the Bath Ankylosing Spondylitis Functional Index (BASFI) was at 64/100. Moreover, she still had recurrent uveitis.
A biotherapy was then indicated. Pretherapeutic laboratory tests showed a normal blood count, a sedimentation rate at 27 mm/h, a C-reactive protein at 15 mg/l. Serum Calcium level was at 1.8 mmol/l. The hepatic and renal enzyme and lipid profile came back normal. The intradermo-reaction to tuberculin, the culture sputum for tuberculosis and the QuantiFERON-TB Gold test were negative. Human immunodeficiency virus (HIV), hepatitis and syphilitic serologies were negative too. Electrophoresis of protein, Angiotensin converting enzyme, Lactate Dehydrogenase (LDH) and β2 microglobulin levels were normal. The chest X-ray and the thoracic CT scan revealed a highter diameter of the mediastinal-hilar lymphadenopathy than in 2009. The PET scan highlighted a moderate hyper-metabolism of the mediastinal lymph nodes, which is not an argument for follicular lymphoma. A second biopsy of mediastinal lymph nodes was performed this time by thoracotomy. Histopathologic examination described small sized nonnecrotizing epithelioid granulomas characterizing sarcoidosis (Figure 1A).
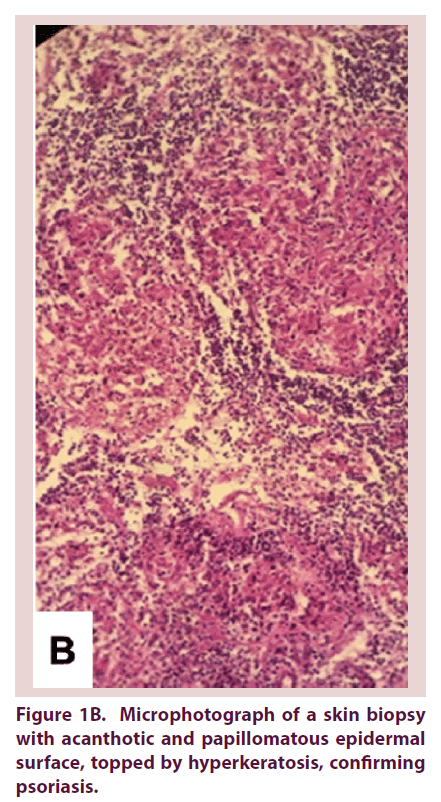
The skin biopsy confirmed psoriasis vulgaris: acanthotic and papillomatous epidermal surface with a thickened stratum corneum, a parakeratosis containing aggregated neutrophils (Munro-Sabouraud micro abscesses) and a perivascular lymphocytic inflammatory cell infiltrate in the dermis. There was no sarcoidal granuloma (Figure 1B).
Figure 1B. Microphotograph of a skin biopsy with acanthotic and papillomatous epidermal surface, topped by hyperkeratosis, confirming psoriasis.
As a result, we could confirm the diagnosis of sarcoidosis with joints, pulmonary and ocular involvement associated to psoriasis vulgaris, which has been mistaken for psoriatic arthritis.
Discussion
Sarcoidosis and psoriasis vulgaris association raises questions about the link between these two diseases and sometimes poses a diagnostic problem.
The diagnosis of psoriasis vulgaris is clinical whereas for sarcoidosis, it is based on clinical and radiological evidence. Supported by bronchoscopy coupled to a biopsy, a bronchoalveolar lavage and histopathological findings of nonnecrotizing epithelioid granulomas. In our report, the results of imaging and histopathology, as well as excluding other differential diagnosis were enough to make the diagnosis of sarcoidosis.
Sarcoidosis is a systemic granulomatosis of unknown cause, characterized by an overreaction of the immune system against an antigen yet to be identified [1]. TH1 and TH17 cells, two types of helper T-cells have an important role in the pathogenesis of sarcoidosis [2]. Recently, Facco et al. have shown that TH17 cells are involved in the formation of granulomas. They participate in the phase of alveolar granuloma and progression to the fibrous phase of the disease [3]. Existence of these cells has been widely exposed in various diseases characterized by an excessive inflammatory response, including psoriasis, rheumatoid arthritis, Crohn’s disease and tuberculosis.
TH1 and TH17 cells are involved in the pathogenesis of psoriasis, by releasing inflammatory cytokines that promote the recruitment of immune cells, the proliferation of keratinocytes and the maintenance of the inflammatory response. Moreover, the IL23R (interleukin 23 receptor) gene has been described to be a genetic factor strongly associated with psoriasis, inflammatory bowel disease, the spondyloarthropathies and sarcoidosis [4]. It has also been reported that the expression of Antigen pso27 related to the pathogenesis of psoriasis is significantly increased in the lung of patients with sarcoidosis lung.
The association between psoriasis and sarcoidosis was rarely observed [5-8].
In fact, in sarcoidosis, the systemic immune response produces psoriasiform granulomatous skin lesions which pose the problem of differential diagnosis with psoriasis [9]. Thus, a cutaneous biopsy of an erythematous-squamous lesion was necessary to exclude skin sarcoidosis in its psoriasiform aspect. Karolyn et al. reported this association in 7 cases of which three also had skin lesions of sarcoidosis, and one patient had both diseases on the same sample [10].
In skin psoriasis, joint involvement is not always synonymous of psoriatic arthritis. It may be gout, sarcoidosis or simply osteoarthritis. The joint involvement was attributed to sarcoidosis since we had not enough evidence for psoriatic arthritis.
Concerning the eye involvement, sarcoid uveitis is a common manifestation. It is most often a chronic granulomatous anterior uveitis, uni or bilateral. But it can also be nongranulomatous half of the time, as it is the case for our patient [11].
Similarities in immunopathogenesis of psoriasis and sarcoidosis suggest a common pathway between these two diseases. This partly explains the association in our case report. It is conceivable that other common, yet unknown interactions between these two diseases could play an important role in their coexistence in a same individual.
Conclusion
We report a rare association between sarcoidosis and psoriasis who have a common immunopathogenesis and diagnosis is not always easy to establish, sometimes requiring further tests more advanced to keep the diagnosis through this observation.
References
- Iannuzzi MC, Rybicki BA, Teirstein AS. Sarcoidosis. N. Engl. J. Med. 357, 2153–2165 (2007).
- Wallis RS, Ehlers S. Tumor necrosis factor and granuloma biology: explaining the differential infection risk of etanercept and infliximab. Semin. Arthritis Rheum. 34, 34–38 (2005).
- Facco M, Cabrelle A, Teramo A et al. Sarcoidosis is a Th1/Th17 multisystem disorder. Thorax. 66, 144–150 (2011).
- Chung Y, Chang SH, Martinez GJ et al. Critical regulation of early Th17 cell differentiation by interleukin-1 signaling. Immunity. 30, 576–587 (2009).
- Usuki K, Hamada H, Terasaki Y et al. Sarcoidosis associated with psoriasis vulgaris. J. Dermatol. 28(2), 86–90 (2001).
- Nikolopoulou M, Katsenos S, Psathakis K et al. Pulmonary sarcoidosis associated with psoriasis vulgaris: coincidental occurrence or causal association? Case report. BMC Pulm. Med. 6, 26 (2006).
- Burgoyne JS, Wood MG. Psoriasiform sarcoidosis. Arch. Dermatol. 106(6), 896–898 (1972).
- Farmer JL Jr, Winkelmann RK. Psoriasis in association with sarcoidosis: Report of a case. Arch. Dermatol. 81, 983–986 (1960).
- Petroianni A, Halili I, Lagalla M, Mougkaraki E et al. Sarcoidosis at onset of Psoriasis: a common immunopathogenesis. Review and case report. Eur. Rev. Med. Pharmacol. Sci.19, 1773–1778 (2015).
- Wanat KA, Schaffer A, Richardson V et al. Sarcoidosis and psoriasis. a case series and review of the literature exploring co-incidence vs. coincidence. JAMA Dermatol. 149(7), 848–852 (2013).
- Khanna A, Sidhu U, Bajwa G et al. Pattern of ocular manifestations in patients with sarcoidosis in developing countries. Acta Ophtalmol. Scand. 85(6), 609–612 (2007).