Case Report - Interventional Cardiology (2026) Volume 18, Issue 1
The Silent Giant : Incidental Discovery of a Large Left Atrial Myxoma
- Corresponding Author:
- Fadoua Lachhab
Cardiovascular Surgery Department, Faculty of Medicine and Pharmacy of Rabat, Mohammed V University, Rabat, Morocco.
E-mail: fadoulachhab@gmail.com
Received date: 21-Jan-2026, Manuscript No. FMIC-26-183137; Editor assigned: 23-Jan-2026, Pre QC No. FMIC-26-183137 (PQ); Reviewed date: 06-Feb-2026, QC No. FMIC-26-183137; Revised date: 13-Feb-2026, Manuscript No. FMIC-26-183137 (R); Published date: 23-Feb-2026, DOI: 10.37532/1755-5310.2026.18(1).1034
Abstract
Primary cardiac tumors are uncommon, with atrial myxomas constituting the most prevalent benign subtype and predominantly arising in the left atrium. Although traditionally associated with obstructive, embolic, and constitutional manifestations, their clinical presentation is highly heterogeneous. Tumor size is often presumed to correlate with symptom severity; however, this association remains inconsistent and appears to be influenced by additional factors such as tumor mobility, site of attachment, and degree of valvular interference. We report the incidental diagnosis of a giant left atrial myxoma with a surface area of 37 cm² in a 54-year-old man presenting with non-specific symptoms and no evidence of significant hemodynamic compromise. Despite its remarkable size and diastolic prolapse through the mitral valve, the tumor was not associated with severe obstruction or embolic events. T his case underscores the potentially silent yet life-threatening nature of large atrial myxomas and highlights the importance of early imaging-based diagnosis and prompt surgical resection, even in patients with minimal or absent symptoms.Keywords
Left atrial myxoma • Primary cardiac tumor • Incidental diagnosis • Echocardiography • Surgical excision
Introduction
Primary cardiac tumors are rare, with reported incidences ranging from 0.17% to 0.19% in autopsy-based studies. Approximately three-quarters of these tumors are benign, among which atrial myxomas account for nearly half. Myxomas most frequently originate in the left atrium, typically from the interatrial septum at the level of the fossa ovalis, although they may arise from other intracardiac structures.
The clinical presentation of atrial myxomas is characterized by marked variability, ranging from asymptomatic cases to life-threatening complications. The classic symptom triad includes intracardiac obstruction, systemic embolization, and constitutional manifestations. However, many patients present with vague or nonspecific symptoms, and the diagnosis is often established incidentally during cardiac imaging performed for unrelated indications. The relationship between tumor size and clinical severity remains poorly defined, as small lesions may result in catastrophic embolic events, whereas large tumors may remain clinically silent.
The majority are sporadic, without genetic predisposition, and occur as solitary tumors with a single anatomical location [1].
The present report describes an incidentally discovered giant left atrial myxoma in a minimally symptomatic patient, emphasizing the unpredictable natural history of this tumor and the critical role of systematic cardiac imaging and timely surgical management.
Case Presentation
A 54-year-old male with no prior history of cardiovascular disease was referred for transthoracic echocardiography due to persistent fatigue of unclear etiology. The patient denied dyspnea, chest pain, syncope, palpitations, or prior thromboembolic events. There was no history of fever, weight loss, or other constitutional symptoms. Physical examination revealed stable hemodynamic parameters. Cardiac auscultation demonstrated a faint systolic murmur at the apex without signs of congestive heart failure or peripheral embolization. Electrocardiography showed normal sinus rhythm, and routine laboratory tests, including inflammatory markers, were within normal limits.
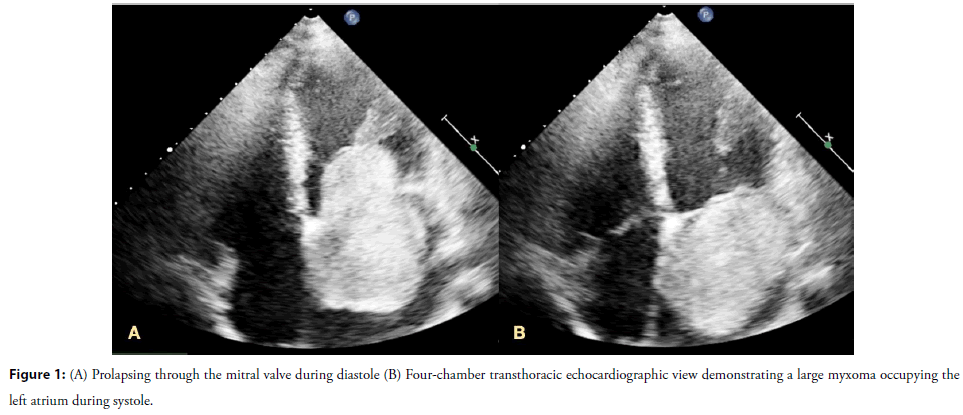
Transthoracic echocardiography identified a large, heterogeneous, and highly mobile intracavitary mass occupying a substantial portion of the left atrium during systole. The lesion measured 83 Ã 46 mm, corresponding to an estimated surface area of 37 cm2. It was attached to the interatrial septum by a pedunculated stalk and prolapsed through the mitral valve into the left ventricle during diastole. Notably, there was no significant transmitral gradient, and only mild mitral regurgitation was observed. Mild tricuspid regurgitation was present, with preserved right ventricular function and normal pulmonary arterial pressures (Figure 1, Video 1).
Video 1: Echocardiographic video in a four-chamber view illustrating the large left atrial myxoma prolapsing through the mitral valve during each diastolic phase.
Given the size, mobility, and embolic potential of the mass, urgent surgical intervention was indicated. The patient underwent tumor excision via median sternotomy under cardiopulmonary bypass with aortic cross-clamping. The myxoma was resected en bloc together with a portion of the interatrial septum, which was reconstructed using an autologous pericardial patch. Histopathological analysis confirmed the diagnosis of a benign atrial myxoma, characterized by scattered stellate and polygonal cells embedded within a myxoid stroma (Figure 2, Video 2).
Video 2: Intraoperative video documenting the surgical excision of the left atrial myxoma.
The postoperative course was uneventful. At six-month follow-up, the patient remained asymptomatic, and followup echocardiography demonstrated no evidence of tumor recurrence.
Results and Discussion
Atrial myxomas represent the most common primary cardiac neoplasms and are located in the left atrium in approximately 75% of cases. Their preferential origin from the interatrial septum supports the hypothesis of derivation from multipotent mesenchymal cells. While most myxomas occur sporadically as solitary lesions, a minority are associated with familial syndromes, most notably Carney Complex (CNC), which is characterized by multiple and recurrent myxomas and extracardiac manifestations.
Despite their benign histopathological features, atrial myxomas carry a substantial risk of morbidity and mortality due to their intracardiac location and propensity to cause obstruction, embolization, and arrhythmias [2]. Clinical manifestations are influenced by tumor size, consistency, mobility, and site of attachment. Obstructive symptoms may simulate mitral stenosis and are often position-dependent, whereas embolic complications, particularly cerebrovascular events, occur in up to 30% of patients and are more closely related to tumor friability than to absolute size. Constitutional symptoms, including fever, malaise, and weight loss, are believed to result from cytokine-mediated inflammatory responses, particularly involving interleukin-6 [3,4].
The present case exemplifies the lack of a linear relationship between tumor size and symptom burden. Despite its exceptional dimensions and mitral valve prolapse during diastole, the myxoma resulted in minimal clinical manifestations and no significant hemodynamic compromise. This phenomenon may be attributed to the tumorâs smooth surface, pedunculated attachment, and absence of sustained mitral inflow obstruction, allowing preserved diastolic filling.
Echocardiography remains the diagnostic modality of choice for atrial myxomas. Transthoracic echocardiography offers high sensitivity and specificity, while transesophageal echocardiography provides superior visualization of tumor attachment and mobility. Advanced imaging modalities such as cardiac magnetic resonance imaging and computed tomography can further aid in tissue characterization and differentiation from other intracardiac masses, including thrombi and malignant neoplasms.
The differential diagnosis of a left atrial mass includes thrombus, infective vegetation, papillary fibroelastoma, lipoma, and primary or metastatic cardiac malignancies. In the absence of atrial fibrillation or severe left atrial dilation, the identification of a pedunculated mass arising from the interatrial septum strongly favors the diagnosis of myxoma.
Surgical excision remains the only definitive curative treatment and should be undertaken promptly after diagnosis, irrespective of symptom severity. Delayed intervention exposes patients to the risk of sudden cardiac death, acute valvular obstruction, or systemic embolization, with reported mortality rates of up to 8% of patients awaiting surgery. Intraoperative handling should be minimized prior to aortic cross-clamping to reduce the risk of tumor fragmentation and embolization.
In cases of known preoperative cerebral embolism without hemorrhage, tumor resection is ideally performed about seven days after the event to avoid further stroke while allowing time for cerebral stabilization prior to cardiopulmonary bypass [5,6].
Operative mortality is less than 5%, typically due to advanced age and comorbidities. Mortality is higher in ventricular myxoma surgery, approximately 10% [7].
The recurrence rate of sporadic, non-familial myxomas is 1â4%. In contrast, familial complex myxomas with genetic mutations have a recurrence rate as high as 22%. Recurrences are generally more frequent in younger patients and tend to occur in the heart, either at the original site or in a different chamber. Rarely, extra cardiac recurrence may occur, likely due to embolization followed by tumor growth and local invasion.
Long-term echocardiographic monitoring after myxoma resection is recommended but not yet standardized. Close follow-up is particularly advised for patients with multifocal tumors, tumors removed from unusual cardiac locations, suspected incomplete resections, or known genetic mutations especially in familial forms or Carney Complex (CNC).
Conclusion
This case highlights the clinically silent yet potentially fatal nature of giant left atrial myxomas and reinforces the concept that tumor size does not reliably predict symptom severity. Comprehensive cardiac imaging should be considered even in patients with nonspecific complaints. Prompt surgical resection remains essential to prevent severe complications and is associated with excellent longterm outcomes.
Statement
Ethical approval not required for this case report in accordance with the institutional policies. Written informed consent has been obtained from the patient for the publication of this case report and accompanying images.
Data availability statement
All data generated or analyzed During this case report are included in this published article.
References
- Reynen K. Cardiac myxomas. N Engl J Med. 333(24):1610-1617 (1995).
[CrossRef] [Google Scholar] [PubMed]
- Pinede L, Duhaut P, Loire R. Clinical presentation of left atrial cardiac myxoma: A series of 112 consecutive cases. Medicine. 80(3):159-172 (2001).
[CrossRef] [Google Scholar] [PubMed]
- Bjessmo S, Torbjorn Ivert MD. Cardiac myxoma: 40 yearsâ experience in 63 patients. Ann Thorac Surg. 63(3):697-700 (1997).
[CrossRef] [Google Scholar] [PubMed]
- Samanidis G, Khoury M, Balanika M, et al. Current challenges in the diagnosis and treatment of cardiac myxoma. Kardiol Pol. 78(4):269-277 (2020).
[CrossRef] [Google Scholar] [PubMed]
- Burke AP, Virmani R. Cardiac myxoma: A clinicopathologic study. Am J Clin Pathol. 100(6):671-80 (1993).
[CrossRef] [Google Scholar] [PubMed]
- Lee VH, Connolly HM, Brown RD. Central nervous system manifestations of cardiac myxoma. Arch Neurol. 64(8):1115-1120 (2007).
[CrossRef] [Google Scholar] [PubMed]
- El Sabbagh A, Al-Hijji M, Thaden JJ, et al. Cardiac tumors: Diagnosis, evaluation, and treatment. Curr Treat Options Cardiovasc Med. 21(4):16 (2019).