Case Report - Imaging in Medicine (2014) Volume 6, Issue 1
Contrast echocardiography test for intrapleural extravasation by central venous catheter
Amalia Schiavetti*,1 & Flavia Ventriglia21Department of Pediatrics, Oncology Unit, “Sapienza” University, Rome, Italy
2Department of Pediatrics, Cardiology Unit, “Sapienza” University, Rome, Italy
- Corresponding Author:
- Amalia Schiavetti
Department of Pediatrics
Oncology Unit, “Sapienza” University, Rome, Italy
Tel: +39 06 4997 9357
Fax: +39 06 4997 0356
E-mail: amalia.schiavetti@uniroma1.it
Abstract
Pleural effusion resulting in respiratory distress is a rare complication associated with central venous catheters (CVCs) in children with cancer. Early diagnosis is required to avoid life-threatening complications. Herein, we report the diagnosis by safe echocardiography test in a child with nephroblastoma who experienced acute respiratory distress after drug infusion by CVC. The suspicion of catheter displacement was not confirmed by usual first step examination: chest x-ray showed the tip of the catheter projected in site; moreover, the catheter was perfectly functional to aspiration and injection. A contrast echocardiography test with agitated saline demonstrated microbubbles in the pleural effusion and allowed the diagnosis of intrapleural extravasation, probably caused by primary perforation and malposition of the catheter tip.
Keywords
catheter • contrast echocardiography test • respiratory distress
Central venous catheters (CVCs) are of vital importance in children with cancer. They have been used for more than 20 years and have greatly improved the quality of cure. Indeed, the use of CVC has became a standard of care in childhood cancer. Most common complications associated with these devices are related to catheter malfunction, catheter thrombosis and infection, whereas chemotherapy drug extravasation is a rare, but is a significant complication [1–3]. Mechanical complications of central venous access are no exception and are related to the venous access procedure or to the catheter itself. A risk of a life-endangering event from the vascular access procedure alone is approximately one event per 1000 access procedures [1,4]. Pleural effusion resulting in respiratory distress is usually due to the migration of the catheter into the pleural space. In these cases, early diagnosis is necessary to avoid life-threatening complications [5–8]. Herein, we describe the diagnosis and management of pleural drug extravasation without apparent CVC displacement in a child.
Case report
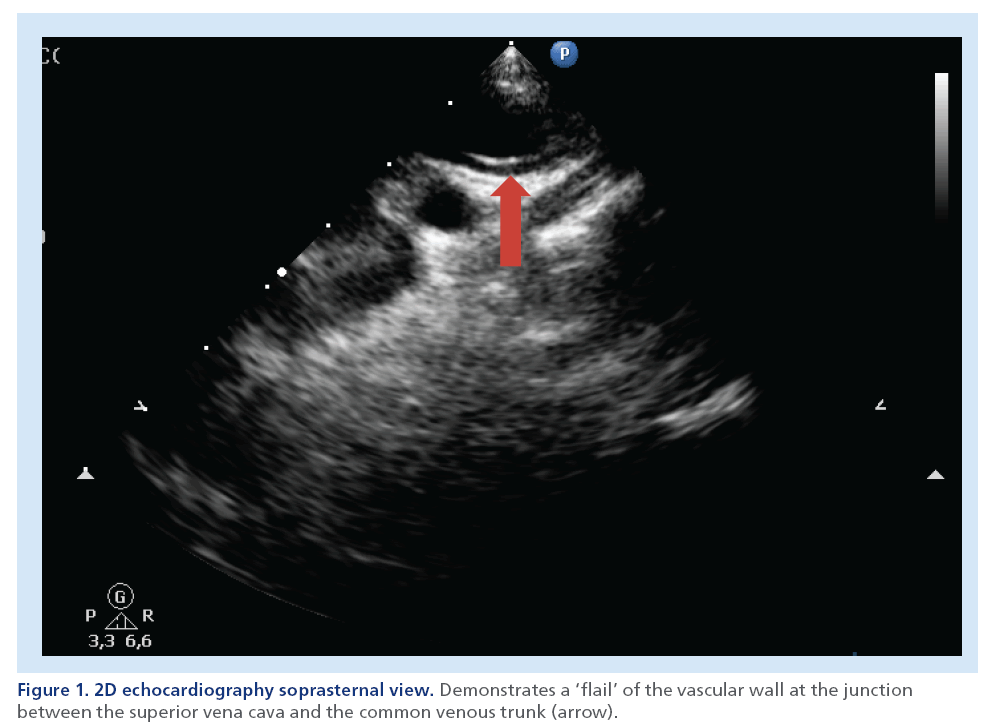
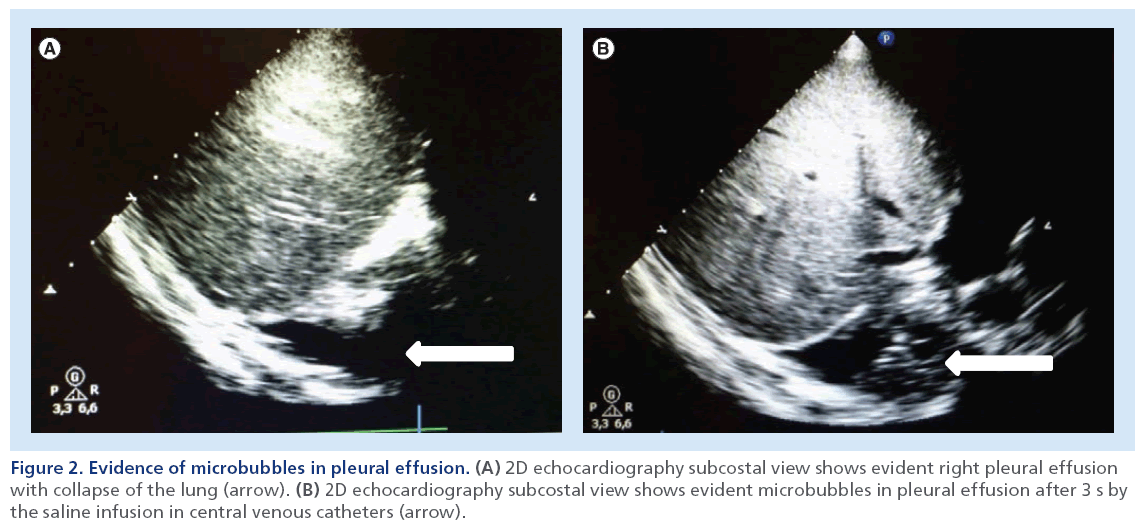
A 3-year-old child was admitted to the oncology unit owing to abdominal pain. Ultrasound and computed tomography (CT) showed a tumor mass on the lower pole of the right kidney. Chest x-ray, ECG and echo cardiography were normal. The child received preoperative chemotherapy and surgery following the International Society of Pediatric Oncology protocol [9]. Histology showed a nephroblastoma with blastematous components. To administer postoperative chemotherapy for stage I high-risk nephroblastoma, a CVC (Groshong type) was inserted via the right subclavian vein approach by the percutaneous-type seldinger technique under general anesthesia. A chest fluoroscopy control during the procedure showed the tip of the catheter projecting into the superior vena cava. The catheter was functional to injection and aspiration and routine 1 g ceftriaxone diluted in 50 cc of saline was administered. Afterwards, short infusion of 2 mg/m2 vincristine followed by 250 cc of saline infusion were administered by CVC. A few hours later, the child presented with dyspnea, chest pain and fever (38.5°C). A chest x-ray showed a large right-sided pleural effusion in the absence of any relevant lung disease; the tip of the catheter was projected in site. The catheter was perfectly functional to both aspiration and injection. Blood was easily aspirated from CVC and chemistry was performed. The serum value of hemoglobin was abnormally low (3 g/dl), thus, the test was repeated from peripheral vein blood and the value of hemoglobin resulted higher (9 g/dl). Echocardiography was immediately performed. The images documented a ‘flail’ of the wall between the common trunk vein and superior vena cava (Figure 1). A contrast echocardiography test with agitated saline demonstrated microbubbles into the pleural effusion (Figure 2). This test confirmed the suspicion of the CVC displacement. The catheter was removed under general anesthesia in a setting where thoracic surgery is immediately available. The child was managed conservatively without specific therapy. Respiratory symptoms resolved spontaneously in 1 week. After 7 days, a chest x-ray showed the absence of pleural effusion and the presence of pleural thickening on the right side, which disappeared 1 month later. The patient concluded his chemotherapy protocol; at present, he is in good health, without disease at 3-year follow-up.
Figure 1: 2D echocardiography soprasternal view. Demonstrates a ‘flail’ of the vascular wall at the junction between the superior vena cava and the common venous trunk (arrow).
Figure 2: Evidence of microbubbles in pleural effusion. (A) 2D echocardiography subcostal view shows evident right pleural effusion with collapse of the lung (arrow). (B) 2D echocardiography subcostal view shows evident microbubbles in pleural effusion after 3 s by the saline infusion in central venous catheters (arrow).
Discussion
Rapid and accurate identification of the cause for acute dyspnea is a challenge to the clinician. The diagnosis of intrapleural extravasation should first be considered in any patient with a CVC who experiences acute respiratory distress. Confirmation of the correct position of the catheter is routinely performed by a chest x-ray and by withdrawal of blood from the catheter. In our patient, the suspicion of CVC displacement was not confirmed by the usual first-step examination. Chest x-ray showed the tip of the catheter projected in site; moreover, the catheter was perfectly functional to aspiration and injection. This particular situation could be explained by the perforation of the wall vein at the moment of the procedure with the catheter tip in a borderline position between the pleura and the vein. In these cases, chest CT or chest fluoroscopy with iodinated contrast injection have been used for diagnosis and considered the gold standard examination by some authors [8,9]. However, the dose of radiation and the need to move the patient into a radiology room are important issues to be aware of, particularly in childhood.
The noninvasive ultrasound-based diagnosis is an ideal, safe method for emergency conditions [10]. In our patient, although the echocardiography showed a ‘flail’ of the wall between the common trunk vein and the superior vena cava, only the safe contrast echocardiography test with saline allowed an immediate diagnosis of the pleural effusion through the CVC. Ultrasound can be used in order to check for immediate and life-threatening complications, as well as the catheter’s tip position as suggested by Lamperti et al. [11]. The use of a safe contrast echocardiography test via a catheter could be recommended at the time of insertion, both in adults and in children [11]. Concerning the veins involved in our case, it is difficult to know exactly if the catheter was in the internal/thoracic mammary vein or the pericardiophrenic vein or other vein. We did not perform angiography imaging studies, but we preferred to remove the catheter under general anesthesia in a setting where thoracic surgery is immediately available. In the literature, intrapleural vincristine was reported only in one other child because of catheter-related thrombosis diagnosed by venogram [5]. Although the vinca-alkaloids are vesicant drugs and extreme care must be taken to avoid extravasation, in this case, intrapleural vincristine was considered safe at long-term follow-up, probably because of diluted infusion [5]. In our case, the infusion of saline immediately after vincristine attenuated the vesicant effects of the chemotherapy–drug.
In the future, ultrasound catheter cannulation, ultrasound imaging and ultrasound needle visualization could both be used in adults and in childhood.
Financial & competing interests disclosure
The authors have no relevant affiliations or financial involvement with any organization or entity with a financial interest in or financial conflict with the subject matter or materials discussed in the manuscript. This includes employment, consultancies, honoraria, stock ownership or options, expert testimony, grants or patents received or pending, or royalties.
No writing assistance was utilized in the production of this manuscript.
Informed consent disclosure
The authors state that they have obtained verbal and written informed consent from the relevant parent(s)/guardian(s) for the inclusion of their child’s medical and treatment history within this case report.

References
- Weldon CB, Jaksic T, Shamberger RC. General principles of surgery. Pizzo PA, Poplack DG (Eds). In: Principles and Practice of Pediatric Oncology (60th Edition). Lippincott Williams & Wilkins, PA, USA, 386–387 (2011).
- Whickham R, Purl S, Welker D. Long-term central venous catheters: issues for care. Semin. Oncol. Nurs. 8, 133–147 (1992).
- Greene JN. Catheter-related complications of cancer therapy. Infect. Dis. Clin. North Am. 10, 255–295 (1996).
- Chung D, Ziegler M. Central venous catheter access. Nutrition 14, 119–123 (1998).
- Askegard-Giesmann JR, Caniano DA, Kenney BD. Rare but serious complications of central line insertion. Semin. Pediatr. Surg. 18, 73–83 (2009).
- Watterson J, Heisel M, Cich JA et al. Intrathoracic extravasation of sclerosing agents associated with central venous catheters. Am. J. Pediatr. Hematol. Oncol. 10, 249–251 (1988).
- Uges JW, Vollaard AM, Wilms EB et al. Intrapleural extravasation of epirubicin, 5-fluouracil and cyclophosphamide, treated with dexrazoxane. Int. J. Clin. Oncol. 11, 467–70 (2006).
- Tan PL, Chan C. Bilateral pleural effusions following central venous cannulation. J. Postgrad. Med. 53, 117–118 (2007).
- Graf N, van Tinteren H, Bergeron C et al. Characteristics and outcome of stage II and III non-anaplastic Wilms’ tumour treated according to the SIOP trial and study 93-01. Eur. J. Cancer 48, 3240–3248 (2012).
- Zanobetti M, Poggioni C, Pini R. Can chest ultrasonography replace standard chest radiography for evaluation of acute dyspnea in the ED? Chest 139, 1140–1147 (2011).
- Lamperti M, Bodenham AR, Pittiruti M et al. International evidence-based recommendations on ultrasound-guided vascular access. Intensive Care Med. 38, 1105–1117 (2012).