Research Article - Clinical Practice (2021) Volume 18, Issue 6
Characteristics of patients with coronavirus disease SARS-Cov-2 in Jordan: Retrospective cross-sectional study
- Corresponding Author:
- Amjad Shdaifat
Department of Medicine and Family Medicine
The Hashemite University, Jordan
E-mail: drshdifat@hu.edu.jo
Abstract
Background: COVID-19 exacerbations and severity are more prevalent in older patients with pre-existing comorbidities. This study aimed to explore the characteristics, outcomes and analyze the predictors of severe COVID-19 disease in patients admitted to public hospitals in Amman Jordan.
Methods: Clinical and laboratory data were collected from the medical records of 112 confirmed COVID-19 patients admitted to public hospitals in Jordan. Analysis was performed using appropriate statistical tests with SPSS version 24.
Results: Age ≥ 65 years (vs. <65 years), diabetes mellitus, hypertension, lymphocyte count <0.8 × 109/L (vs. ≥ 0.8 × 109/L), and C-reactive protein ≥ 50 mg/L (vs. <50 mg/L), were significantly associated with serious disease. After adjusting for age, lymphocyte count <0.8 × 109/L (Adjusted Odds Ratio [AOR] 62.4; Confidence Interval [CI] 1.3-3079.5; p=0.038) and C-reactive protein ≥ 50 mg/L (AOR 19.0; CI 2.3-156.5; p=0.006) remained significant predictors of serious disease.
Conclusion: The findings suggest that the health system in Jordan could identify older age and inflammatory blood markers as predictors of disease severity and death associated with COVID-19. This can enhance targeted preventive inventions and control measures.
Keywords
COVID-19, morbidity factors
Introduction
The novel Coronavirus Disease 2019 (SARSCoV- 2) was first reported on December 8, 2019, in an individual who presented with symptoms of pneumonia [1]. Since then it has spread worldwide and as of July 2, 2021, there have been 182 319 261 confirmed cases across 216 countries and 3 954 324 deaths [2]. Currently, the death rate worldwide is 2%. In Jordan 752,617 confirmed cases have been reported and 9,769 deaths as of July 2, 2021, [2].
Risk factors associated with severe disease and deaths are emerging. A study looking at the genome of SARS-CoV-2 and SARS-CoV reported a 79.6% sequence similarity and 96% identity to a Bat coronavirus (CoV RaTG13) [3]. Patients with SARS-CoV-2 often clinically present with pneumonia which can result in high mortality [4]. An early study in Wuhan, China showed that of 41 confirmed SARS-CoV-2, six resulted in death (15%) [4]. However, a larger study looking at 138 patients reported a mortality rate of 4.3% [5]. Currently, there is no cure for SARS-CoV-2 and vaccines seem to be the only hope to control this global pandemic. In the meantime, the health system must identify the most vulnerable, create awareness and provide care to moderate and severe cases.
The emerging literature regarding the risk factors of COVID-19 indicates that certain demographics of a population are more prone to developing exacerbations in SARS-CoV-2. Most initial studies were carried out in China particularly in the district of Wuhan. The patients identified most severe for COVID-19 were older patients with pre-existing comorbidities, and were more likely to go on towards developing multi-organ failure and death compared to younger patients [6]. Other prognostic factors that compared between severe patients and milder patients showed certain risk factors exacerbated the outcome of the disease. One particular study found that male gender, increased lactate dehydrogenase level, cardiac injury, high blood glucose, leucocytosis and high intake of corticosteroids decreased survival [7].
Currently, there is limited research presented from the Arab countries where risk factors such as obesity, diabetes mellitus, hypertension, and smoking are more prevalent than in other regions [8]. This study aimed to look into the characteristics, outcomes and analyze the predictors of severe COVID-19 disease in patients admitted to public hospitals in Amman Jordan.
Method
■ Study population
A total of 112 confirmed COVID-19 cases admitted to public hospitals in Jordan during the period 5th March and 22 April 2020 were included in this study. The medical records of these patients were reviewed, and the inclusion criteria were having at least one positive COVID-19 RT-PCR test performed by an accredited referral lab. The demographic characteristics, documented clinical presentation, medical history, and laboratory data were extracted from the medical records of these patients. The ethical approval of this study was issued from the Hashemite University IRB, The confidentiality of data was secured by the research team all through the data collection and analysis stages.
■ Statistical analysis
Descriptive statistical analysis was used for the determination of demographic and clinical, laboratory results. Chi-square and Fisher’s exact tests were used to assess the association between the study participants’ characteristics and the disease severity, The level of statistical significance was set at ≤ 0.05. Data were analyzed using Microsoft Excel-2010 and SPSS version 24.0.
Results
■ Patient characteristics
Overall, 112 patients with confirmed infection with SARS-CoV-2 were admitted between 5th March and 22 April 2020. The median age was 35 years (Interquartile Range [IQR] 22- 53) and 64 (57.1%) were men. Overall, 26 individuals (25.2%) reported close contact with a confirmed case of COVID-19, whereas 34 (33.0%) reported recent international travel. The most commonly reported comorbidities were hypertension (N=20; 18.0%), diabetes mellitus (N=18; 16.2%), cardiovascular disease (N=9; 8.1%), and chronic lung disease (N=6; 5.4%). Nineteen patients (19.4%) were current smokers.
The most common symptoms were one or more of the following: cough (N=48; 47.1%), fever (N=47; 44.8%), abdominal pain (N=38; 37.3%), loss of smell and/or taste (N=34; 33.3%), sore throat (N=26; 25.5%), headache (N=26; 25.5%), and shortness of breath (N=23; 22.5%).
■ Clinical progression
The vast majority (N=102; 91.1%) had mild disease, i.e. they could be managed with supportive care at an ordinary isolation unit. Thirty patients (28.3%) had Computer Tomography (CT) findings consistent with COVID-19 including ground-glass opacities (FIGURE 1), vascular enlargement, bilateral abnormalities, lower lobe involvement, and posterior predilection, and 67 (63.2%) had a normal CT scan at admission. Ten patients (9.0%) were transferred to the Intensive Care Unit (ICU) for mechanical ventilation and/or other advanced support, and four (3.6%) of these patients died. A minority of patients (N=6; 5.4%) received antiviral treatment. TABLE 1 summarizes the clinical characteristics of the patients in this study.
| Characteristic | All patients |
|---|---|
| N=112 | |
| Sex | |
| Men | 64/112 (57.1) |
| Women | 48/112 (42.9) |
| Age (years) | |
| Median (IQR) Distribution | 35 (22-53) |
| 0-19 | 22/111 (19.8) |
| 20-49 | 57/111 (51.4) |
| 50-64 | 24/111 (21.6) |
| ≥ 65 | 8/111 (7.2) |
| Contact with known case | |
| Yes | 26/103 (25.2) |
| No | 77/103 (74.8) |
| Recent international travel | |
| Yes | 34/103 (33.0) |
| No | 69/103 (67.0) |
| Co-morbidity | |
| Cardiovascular disease | 9/111 (8.1) |
| Diabetes mellitus | 18/111 (16.2) |
| Hypertension | 20/111 (18.0) |
| Chronic lung disease | 6/111 (5.4) |
| Cancer | 1/111 (0.9) |
| Chronic kidney disease | 2/105 (1.9) |
| Smoking | |
| Yes | 19/98 (19.4) |
| Stopped | 11/98 (11.2) |
| Never | 68/98 (69.4) |
| Reason to test | |
| Symptoms | 34/102 (33.3) |
| Travel | 21/102 (20.6) |
| Known exposition | 47/102 (46.1) |
| Symptoms | |
| Fever | 47/105 (44.8) |
| Sore throat | 26/102 (25.5) |
| Cough | 48/102 (47.1) |
| Shortness of breath | 23/102 (22.5) |
| Headache | 26/102 (25.5) |
| Abdominal pain | 38/102 (37.3) |
| Vomiting | 6/102 (5.9) |
| Diarrhoea | 13/102 (12.7) |
| Loss of smell or taste | 34/102 (33.3) |
| Other | 59/105 (56.2) |
| CT findings | |
| Normal | 67/106 (63.2) |
| Consistent with COVID | 30/106 (28.3) |
| Other findings | 9/106 (8.5) |
| Antiviral therapy | |
| Yes | 6/112 (5.4) |
| No | 106/112 (94.6) |
| Antibacterial therapy | |
| Yes | 12/112 (10.7) |
| No | 100/112 (89.3) |
| Severity | |
| Mild | 102/112 (91.1) |
| ICU | 6/112 (5.4) |
| Dead | 4/112 (3.6) |
| Abbreviations: IQR: Interquartile Range; CT: Computer Tomography; ICU: Intensive Care Unit. | |
Table 1. Clinical characteristics of SARS-CoV-2 positive patients in Jordan, March-April 2020.
■ Laboratory result
Baseline laboratory results of patients included in this study are given in TABLE 2. The median results of all laboratory tests were within the normal range. However, there were significant differences between patients with serious disease compared to those with mild disease for the following variables: neutrophil count (median 5.1 vs. 3.3 × 109/L; p=0.018), lymphocyte count (median 1.0 vs. 2.1 × 109/L; p=0.020), C-reactive protein (median 155 vs. 4 mg/L; p=0.005), and D-dimer (median 1.01 vs. 0.27 mg/L; p=0.044). For total white blood cell count, liver transaminases, creatinine, urea, and electrolytes there were no significant differences between the two groups.
| Variable | Total | Mild | Severe | p |
|---|---|---|---|---|
| N=112 | N=102 | N=10 | ||
| White blood cell count (× 109/L)1 | ||||
| Median (IQR) Distribution | 6.2 (4.9-8.0) | 6.2 (4.8-8.0) | 7.2 (6.1-9.5) | 0.693 |
| <4 | 12/106 (11.3) | 11/96 (11.5) | 1/10 (10.0) | |
| 4-9.9 | 83/106 (78.3) | 76/96 (79.2) | 7/10 (70.0) | |
| ≥ 10 | 11/106 (10.4) | 9/96 (9.4) | 2/10 (20.0) | |
| Neutrophils (× 109/L)2 | ||||
| Median (IQR) | 3.5 (2.4-4.8) | 3.3 (2.3-4.6) | 5.1 (4.0-7.8) | 0.018 |
| Lymphocytes (× 109/L)1 | ||||
| Median (IQR) Distribution | 2.0 (1.4-2.8) | 2.1 (1.5-2.9) | 1.0 (0.6-1.6) | 0.02 |
| <0.8 | 9/106 (8.5) | 6/96 (6.3) | 3/10 (30.0) | |
| ≥0.8 | 97/106 (91.5) | 90/96 (93.7) | 7/10 (70.0) | |
| ALT (U/L)3 | ||||
| Median (IQR) | 21 (13-31) | 19 (13-30) | 23 (17-42) | 0.475 |
| AST (U/L)4 | ||||
| Median (IQR) | 22 (18-30) | 21 (17-26) | 37 (23-72) | 0.078 |
| C-reactive protein (mg/L)5 | ||||
| Median (IQR) Distribution | 5 (2-30) | 4 (1-18) | 155 (39-208) | 0.005 |
| <10 | 54/86(62.8) | 54/77 (70.1) | 0/9 (0.0) | |
| 10-49 | 22/86 (25.6) | 19/77 (24.8) | 2/9 (22.2) | |
| ≥ 50 | 10/86 (11.6) | 4/77 (5.2) | 7/9 (77.8) | |
| D-dimer (mg/L)6 | ||||
| Median (IQR) Distribution | 0.28 (0.27-0.61) | 0.27 (0.27-0.54) | 1.01 (0.73-1.11) | 0.044 |
| 0-0.9 | 28/33 (84.8) | 26/28 (92.9) | 2/5 (40.0) | |
| ≥1.0 | 5/33 (15.2) | 2/28 (7.1) | 3/5 (60.0) | |
| Creatinine (umol/L)7 | ||||
| Median (IQR) | 70 (61-87) | 69 (61-85) | 81 (67-108) | 0.459 |
| Urea (mmol/L)8 | ||||
| Median (IQR) | 4.4 (3.6-5.6) | 4.4 (3.5-5.5) | 6.5 (4.3-12.8) | 0.418 |
| Na (mmol/L)9 | ||||
| Median (IQR) | 139 (137-140) | 139 (138-140) | 135 (134-140) | 0.43 |
| K (mmol/L)10 | ||||
| Median (IQR) | 4.1 (3.8-4.3) | 4.1 (3.9-4.1) | 3.8 (3.5-4.1) | 0.145 |
| Abbreviations: IQR: Interquartile Range; ALT: Alanine Aminotransferase; AST: Aspartate Aminotransferase. 1 N=106, 2 N=103,3 N=70, 4 N=69, 5 N=86, 6 N=33, 7 N=87, 8 N=89, 9 N=90, 10 N=82. |
||||
TABLE 2. Baseline laboratory results among SARS-CoV-2 positive patients, according to disease severity.
■ Predictors of disease severity
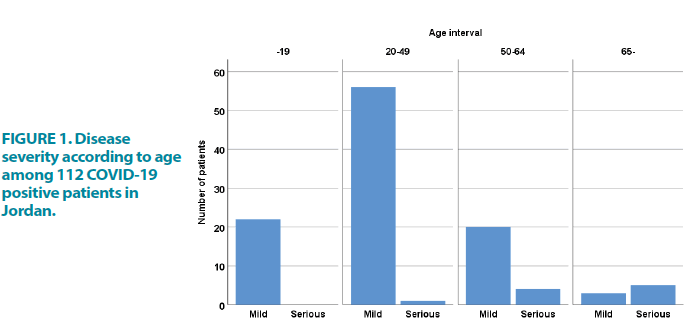
Ten patients had a serious disease, i.e. they required ICU treatment and/or died. These patients were on average older than those who had the mild disease (median age 67 vs. 35 years; p=0.002). FIGURE 1 illustrates the proportion of serious diseases according to age group. Indeed, there were no serious cases among patients below 20 years, compared to 1.8, 16.7, and 62.5% in the age groups 20-49, 50-64, and ≥ 65 years, respectively.
In univariable analysis, age ≥ 65 years (vs. <65 years), diabetes mellitus, hypertension, lymphocyte count <0.8 × 109/L (vs. ≥ 0.8 × 109/L), and C-reactive protein ≥ 50 mg/L (vs. <50 mg/L), were significantly associated with serious disease. After adjusting for age, lymphocyte count <0.8 × 109/L (Adjusted Odds Ratio [AOR] 62.4; Confidence Interval [CI] 1.3-3079.5; p=0.038) and C-reactive protein ≥ 50 mg/L (AOR 19.0; CI 2.3-156.5; p=0.006) remained significant predictors of serious disease (TABLE 3).
| Variable | Unadjusted | Adjusted for age | ||
|---|---|---|---|---|
| Odds ratio (95% CI) | p | Odds ratio (95% CI) | p | |
| Sex | ||||
| Men (vs. women) | 1.1 (0.3-4.3) | 0.848 | 1.9 (0.3-11.9) | 0.503 |
| Age (years)1 | ||||
| ≥ 65 (vs. <65) | 32.7 (6.0-177.0) | <0.001 | N/A | |
| Co-morbidity1 | ||||
| Cardiovascular disease | 3.9 (0.7-22.3) | 0.129 | 0.2 (0.0-3.1) | 0.227 |
| Diabetes mellitus | 8.6 (2.0-36.0) | 0.003 | 2.4 (0.4-14.0) | 0.338 |
| Hypertension | 7.3 (1.7-30.1) | 0.006 | 1.1 (0.2-6.5) | 0.913 |
| Smoking2 | ||||
| Yes (vs. never or former smoker) | 2.1 (0.2-24.9) | 0.544 | N/S | |
| Lymphocytes (× 109/L)3 | ||||
| <0.8 (vs. ≥ 0.8) | 12.7 (2.2-75.1) | 0.005 | 62.4 (1.3-3079.5) | 0.038 |
| C-reactive protein (mg/L)4 | ||||
| ≥50 (vs. <50) | 36.5 (6.6-202.4) | <0.001 | 19.0 (2.3-156.5) | 0.006 |
| Abbreviations: CI: Confidence Interval; N/A: Not Appropriate; N/S: Not Significant. 1 N=111, 2 N=98, 3 N=106, 4 N=86. |
||||
TABLE 3. Predictors of serious disease (death or transfer to intensive care unit) among SARSCoV- 2 positive patients in Jordan.
Discussion
This study focused on patients admitted with COVID-19 in Amman, Jordan. During the early phases of the COVID-19 outbreak in Jordan, the government enforced strict measures to control the spread of the pandemic [9]. This was crucial for disease prevention, surveillance, infection control, and public safety. One reason behind the rapid and stringent lockdown methods evolved from the not-so-distant fear from the Middle East respiratory syndrome coronavirus (MERS-CoV). The emergence of MERS-CoV in 2012 particularly in the Arabian Gulf countries indicated the lack of preparedness for such outbreaks. Major healthcare reforms have been implemented since to help strengthen and prepare countries for such outbreaks [10]. Nonetheless like most countries, the Middle East was not prepared to deal with such a pandemic as COVID-19 [11]. The impact brought from a pandemic is not simply an impact on the healthcare infrastructure, however, stress upon many sectors which are disrupted and recovery differs on a more complex level. However, that being said as of April 20, 2020, Jordan has recorded 112 cases and has been mentioned in the list of top countries to flatten their coronavirus curve and early response to the outbreak [12]. This was due to the restrictive movement of the population and the implementation of lockdown measures [2].
The data collated from this study was able to show that the more severe cases were identified in the age interval of 65+ whereas; milder cases were seen in 20-49 aged intervals. Few studies focus on COVID-19 in Amman, Jordan. A study looking at the “Response, scenarios, strategies and recommendation” within Jordan highlighted the need for the Government to push for more identification of high-risk areas and high-risk demographics [13]. This study is one of the first in Jordan to look into characteristics, outcomes and analyze predictors of severe disease of COVID-19 patients.
Here, in this study, we show that statistically, age is a potential determinant factor for severity in COVID-19. Other studies have also shown age to be a factor alongside an underlying disease [14,15]. We also show that COVID-19 patients presented with fever and cough as the main symptoms followed by shortness of breath which supports the findings from other studies [14,15]. One study looking at 99 COVID-19 patients showed 83% presented with fever, 82% with cough, and (31%) shortness of breath [14].
Studies have found age, non-white race, cardiac disease, hypertension, and chronic kidney disease to be linked to COVID-19 severity/death [16-18]. We found hypertension and diabetes mellitus correlated to disease severity but when adjusted for ages were no longer significant. We did not see a significant correlation between disease severity and cardiovascular disease, although there was a trend.
Interestingly, we see that there is a correlation between neutrophils, lymphocytes, and C-reactive proteins which could be, although not specific, markers for early diagnosis. This supports another study looking at baseline blood tests also identifying neutrophils levels which were higher (38%), lymphocyte to be decreased (35%) and C-reactive proteins to be increased in (86%) COVID-19 positive patients [14].
Even though this research has identified the major risk factor for developing severe COVID-19 and an increased probability of mortality is age. It has not ruled out other age groups’ vulnerability to develop the post-COVID-19 illness. A recent follow-up of patients in Lombardy northern Italy has shown that even non-vulnerable patients who developed mild or non-symptomatic COVID-19 are showing psychosis, insomnia, kidney disease, spinal infections, strokes, chronic tiredness, and mobility issues [19].
We also condone track and trace approaches of COVID-19 patients, as 26 of 102 patients tested due to contact with known COVID-19 patients.
For now, the prevention of acquiring COVID-19 must be paramount for all age groups especially the elderly, as the imminent danger is focused on this group of people.
This study is limited by the small sample size; however, it was conducted in the early phases of the COVID-19 pandemic in Jordan. The findings suggest that the health system in Jordan could identify older age and inflammatory blood markers as predictors of disease severity and death associated with COVID-19.
References
- McGonagle D, O’Donnell JS, Sharif K, et al. Immune mechanisms of pulmonary intravascular coagulopathy in COVID-19 pneumonia. Lancet Rheumatol. 2, e437-445 (2020).
- Chammard TB, Schepers K, Breurec S, et al. Severe Thrombocytopenia after Zika Virus infection, guadeloupe, 2016. Emerg Infect Dis. 23, 696-698 (2017).
- Rashmi MV, Hamsaveena. Hematological and biochemical markers as predictors of dengue infection. Malays J Pathol. 37, 247-251 (2015).
- Yang M, Ng MH, Li CK. Thrombocytopenia in patients with severe acute respiratory syndrome (review). Hematology. 10, 101-105 (2005).
- Al-Tawfiq JA, Hinedi K, Abbasi S, et al. Hematologic, hepatic and renal function changes in hospitalized patients with Middle East respiratory syndrome coronavirus. Int J Lab Hematol. 39, 272-278 (2017).
- Guan WJ, Ni ZY, Hu Y, et al. Clinical characteristics of Coronavirus Disease 2019 in China. New England J Med. 382, 1708-1720 (2020).
- Chen N, Zhou M, Dong X, et al. Epidemiological and clinical characteristics of 99 cases of 2019 novel coronavirus pneumonia in Wuhan, China: a descriptive study. Lancet. 395, 507-513 (2020).
- Sun S, Cai X, Wang H, et al. Abnormalities of peripheral blood system in patients with COVID-19 in Wenzhou, China. Clin Chim Acta. 507, 174-180 (2020).
- Ruan Q, Yang K, Wang W, et al. Clinical predictors of mortality due to COVID-19 based on an analysis of data of 150 patients from Wuhan, China. Intensive Care Med. 46, 846-848 (2020).
- Xu P, Zhou Q, Xu J. Mechanism of thrombocytopenia in COVID-19 patients. Ann Hematol. 99, 1205-1208 (2020).
- Gupta GP, Massague J. Platelets and metastasis revisited a novel fatty link. J Clin Invest. 114, 1691-1693 (2004).
- Jagerschmidt A, Fleury V, Leory MA, et al. Human thrombopoietin structure-function relationships: identification of functionally important residues. Biochem J. 333, 729-734 (1998).
- Kaser A, Brandacher G, Steurer W, et al. Interleukin-6 stimulates thrombopoiesis through thrombopoietin: role in inflammatory thrombocytosis. Blood. 98, 2720-2725 (2001).
- Henry BM, de Oliveira S, Benoit S, et al. Lippi, Hematologic, biochemical and immune biomarker abnormalities associated with severe illness and mortality in coronavirus disease 2019 (COVID-19): a meta-analysis. Clin Chem Lab Med. 58, 1021-1028 (2020).
- Levesque V, Millaire E, Carrier FM, et al. Severe immune thrombocytopenic purpura in critical COVID-19. Intern J Hematol. 112, 746-750 (2020).
- Chen W, Yang B, Li Z, et al. Sudden severe thrombocytopenia in a patient in the recovery stage of COVID-19. Lancet Haematol. 7, e624 (2020).
- Guan WJ, Ni ZY, Hu Y, et al. Clinical characteristics of coronavirus disease 2019 in China. New Eng J Med. 381, 1708-1720 (2020).
- Chen W, Li Z, Yang B, et al. Delayed phase thrombocytopenia in patients with coronavirus disease 2019 (COVID-19). Br J Haematol. 190, 179-184 (2020).
- Sahu KK, Siddiqui AD, Rezaei N, et al. Challenges for management of immune thrombocytopenia during COVID-19 pandemic. J Med Virol. 92, 2277-2282 (2020).
- Pavord S, Thachil J, Hunt BJ, et al. Practical guidance for the management of adults with immune thrombocytopenia during the COVID-19 pandemic. Br J Haematol. 189, 1038-1043 (2020).
- Patel T, Stanton N, Gkikas I, et al. Severe thrombocytopenia secondary to COVID 19. Br Med J Case Rep. 13, e237645 (2020).