Commentary - Interventional Cardiology (2014) Volume 6, Issue 1
Avoiding cardiovascular cul-de-sacs in congenital intervention: keeping our options open
- Corresponding Author:
- Gareth J Morgan
Departments of Paediatric & Adult Congenital Heart Disease,
Evelina London Children’s Hospital, Westminster Bridge Road, London, SE1 7EH, UK
Tel: +44 207 1884562
Fax: +44 207 1884556
E-mail: gareth.morgan@gstt.nhs.uk
Abstract
The evolution of the treatment and palliation of congenital heart disease has been celebrated over the last 20 years as, the literature describes improving longevity in all but the most extreme conditions.
Keywords
adolescence, adult, atrial septal defect, catheter interventions, coarctation, congenital heart disease, stent
The evolution of the treatment and palliation of congenital heart disease has been celebrated over the last 20 years as, the literature describes improving longevity in all but the most extreme conditions [1,2]. Patients with disease states once considered to have uniform neonatal or infantile mortality, such as hypoplastic left heart syndrome and pulmonary atresia with intact ventricular septum, now have a reasonable expectation of survival into teenage years and adulthood [3].
Advances in the field have created new challenges for our patients, including psychological, socioeconomic, chronic comorbidity and inheritance issues [4]. It has also brought new challenges for cardiologists, in all aspects of care, not least with respect to interventional management.
The range of applications for interventional techniques in the adult congenital population is expanding; from procedures as common place as device closure of atrial septal defects (ASDs), to growing experience in percutaneous valve replacement in at least three and, potentially, all four intracardiac valves [5,6]. In addition to the procedures themselves, we must consider that the types of patient on whom we are intervening in adult congenital practice are growing more complex, with increasing numbers of patients returning for interventions following previous ‘corrective’ or palliative surgery and intervention.
Of particular relevance to those with a mixed pediatric and adult congenital practice is the necessity to consider longevity and adulthood, even when we are intervening in neonatal hearts. The implications of lifesaving and disease-altering pediatric interventions must be considered in the context of the patient (and their heart) increasing their weight (and their stroke volume) tenfold or more as they progress toward adult life.
Preservation of vascular access
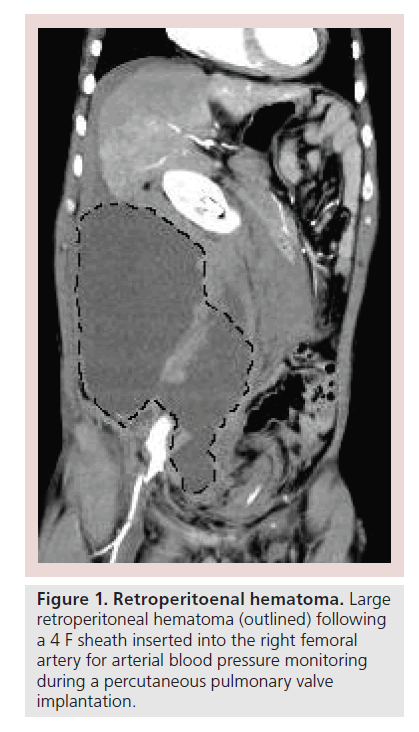
In a wide range of conditions there is a significant likelihood of requiring repeat interventions in adult life. In the majority of cases, these interventions will require vascular access via groin or neck vessels with large calibre sheaths. Preservation of vascular access is essential. For an adult congenital patient to be denied an intervention owing to poorly planned and managed vascular access in childhood would be regrettable [7]. Planning procedures, such as central venous cannulation and interventional sheath insertion, should be carried out with the assumption that repeat access to those sites in the future will be required. Likewise, access should be managed carefully and not left to the most junior member of the team. Use of ultrasound-guided access as well as a careful vessel puncture technique have been shown to decrease vascular complications in a number of settings [8,9]. Hemostasis should be performed using the most clinically appropriate technique, again striving to avoid complications associated with inappropriate use of vessel closure devices and hematoma formation [10]. Considering the necessity of arterial monitoring, access is important in cases where the intervention will be performed via the venous route. Where clinically necessary, arterial monitoring should be performed through a small cannula, and not a sheath, to minimize the risk and impact of arterial access complications (Figure 1).
All pediatric interventionists are conscious of the long-term consequences of stent implantation. Implanting a structure with a finite lifespan and size in a growing child’s heart will always impose limitations on the investigation and treatment of that patient as they develop. These can range from minor problems, such as decreasing the accuracy of quantification in MRI, through to potentially life-limiting exclusion of further interventional or surgical options. By way of an example that spans across this range, consider an infant who, for comorbid reasons, has been treated for coarctation with implantation of a coronary stent; assessment and imaging of the flow through the stent and the tissues surrounding the implant will be affected and may limit the available useful information. Realistically the largest effective diameter achievable by serial dilation of most coronary stents is 6 mm (without considering intimal in-growth); therefore, the aorta will not reach an effective adult size without difficult surgical revision of the stented area or an attempt to fracture the original stent and implant a larger one. The indication for the index procedure as described is often in the context of a life-limiting syndrome or other contraindications to a primary surgical procedure; however, iatrogenic limitation of further treatment must always be considered [11].
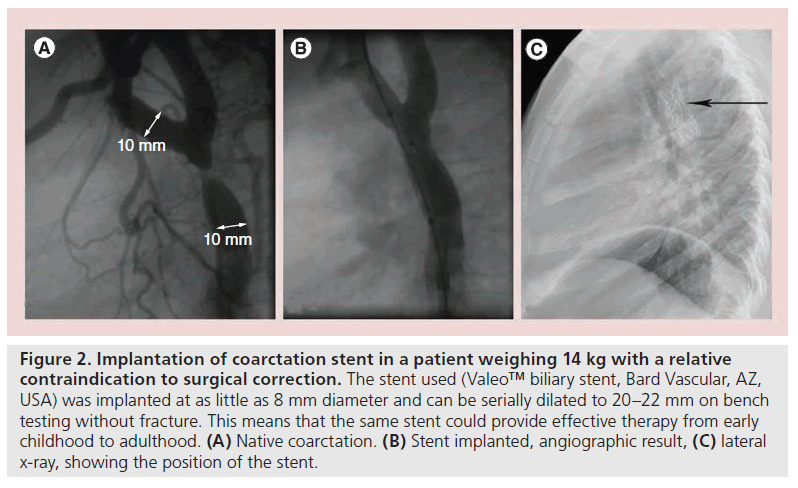
As stent technology develops, there are now more options in such cases. Technology such as biodegradable stents, under development by several different groups, may allow for confident stenting of neonatal coarctation with the hope for either growth of the native tissue after stent degradation or the ability to implant an adultsized stent after a period of time, without the restriction of the original stent size [12]. This technology may be applicable in clinical practice hopefully in the near future. We already have stents available that can be implanted in children and have the potential ability to reach adult size without fracture. This could make procedures such as coarctation stenting in infancy (where there is a relative surgical contraindication) a less palliative procedure (Figure 2).
Figure 2. Implantation of coarctation stent in a patient weighing 14 kg with a relative contraindication to surgical correction. The stent used (Valeo™ biliary stent, Bard Vascular, AZ, USA) was implanted at as little as 8 mm diameter and can be serially dilated to 20–22 mm on bench testing without fracture. This means that the same stent could provide effective therapy from early childhood to adulthood. (A) Native coarctation. (B) Stent implanted, angiographic result, (C) lateral x-ray, showing the position of the stent.
The same applies to stent implantation in the pulmonary arteries; we are now in a position where age and weight are no longer major barriers to stent implantation. Combining flexible sheaths and low profile stent-delivery systems with the improved stents mentioned above could allow percutaneous, hybrid or intraoperative placement with good longevity. Redilating and preserving the integrity of the stents may now be possible for those of us who treat these patients into adulthood.
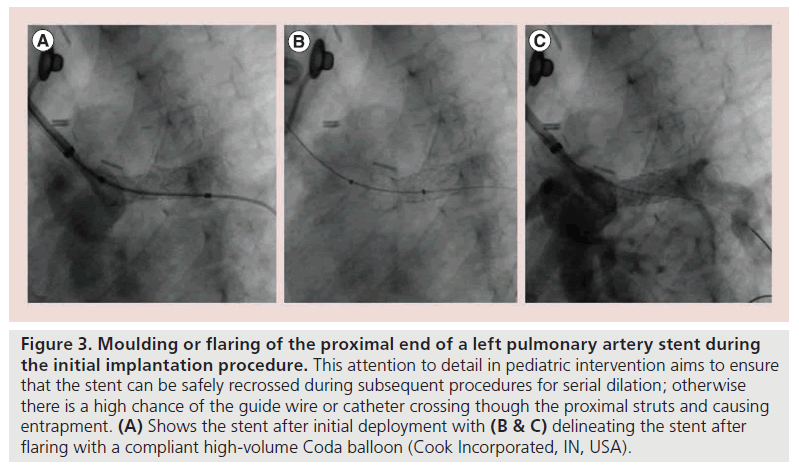
The conduct of the procedure may also change when taking reintervention and adult growth into consideration. In pulmonary artery stenting, rather than leaving the difficult or impossible task of recrossing a previously implanted stent, which by necessity hangs into the main pulmonary artery, this can be eased by pre-emptively flaring the proximal end of the stent using a compliant high volume balloon during the initial procedure (Figure 3). This improves the safety and efficacy of subsequent interventions by inviting wires and catheters to cross the true lumen of the stent and not weave through the stent struts.
Figure 3. Moulding or flaring of the proximal end of a left pulmonary artery stent during the initial implantation procedure. This attention to detail in pediatric intervention aims to ensure that the stent can be safely recrossed during subsequent procedures for serial dilation; otherwise there is a high chance of the guide wire or catheter crossing though the proximal struts and causing entrapment. (A) Shows the stent after initial deployment with (B & C) delineating the stent after flaring with a compliant high-volume Coda balloon (Cook Incorporated, IN, USA).
While the debate continues about device selection and erosion following ASD closure; our choices also need to consider longevity and further intervention. We know that despite device or surgical ASD closure; arrhythmia still occurs in patients who have had significant secundum ASDs. Reaccessing the left atrium to interventionally deal with the arrhythmia can be made either difficult or impossible by the size, position and mechanical properties of ASD occluders. Development of devices with minimal septal area coverage and those with biodegradable components is ongoing; however, the loss of CE marked devices with 90% resorbable components from the market is disappointing [13]. As well as arrhythmia management, we note the development of left atrial appendage closure and transseptal mitral valve interventions; all of which will benefit from a device that either resorbs or is made mostly of material that can be perforated and crossed easily.
It also remains under debate when ASDs should be closed. If an ASD of considerable size is closed in adulthood, then the device will need to match the defect size. With decreasing atrial size after successful closure, the risk of erosion may increase. If such a defect is closed in childhood, then the growth of the heart may counteract these effects and the chances for erosion may be less.
Surgery
If interventional cardiologists need to consider the future, then so do our surgical colleagues. Choices related to conduits and prosthetic valves, techniques of reconstruction of branch pulmonary arteries and awareness of potential for present and future hybrid strategies, need to be taken into consideration. Many surgeons are unhappy with stent material in their operative field, so every interventionalist gripes about the orientation of the reconstructed branch pulmonary arteries or the position and nature of a baffle leak or Fontan fenestration when planning or carrying our subsequent intervention.
Conclusion
Medical and scientific history tell us that acknowledging our potential impact and anticipating what technological options will be available 10 years hence should influence and change the decisions we make today. As our patients live longer and experience the rigours of pregnancy and the chronic diseases of aging, they are exposed to a gambit of pathology previously kept from their reach. They require our attention to detail and our thoughtfulness to ensure that the consequences of our well-intentioned procedures do not push them down cardiovascular cul-de-sacs.
Financial & competing interests disclosure
The authors have no relevant affiliations or financial involvement with any organization or entity with a financial interest in or financial conflict with the subject matter or materials discussed in the manuscript. This includes employment, consultancies, honoraria, stock ownership or options, expert testimony, grants or patents received or pending, or royalties.
No writing assistance was utilized in the production of this manuscript.
References
- Stuart AG. Changing lesion demographics of the adult with congenital heart disease: an emerging population with complex needs. Future Cardiol. 8(2), 305–313 (2012).
- Verheugt CL, Uiterwaal CS, Grobbee DE, Mulder BJ. Long-term prognosis of congenital heart defects: a systematic review. Int. J. Cardiol. 131(1), 25–32 (2008).
- Said SM, Dearani JA, Silversides CK, Martinez RM, Drajpuch DB. Longer-term issues for young adults with hypoplastic left heart syndrome: contraception, pregnancy, transition, transfer, counselling, and re-operation. Cardiol. Young. 21(Suppl. 2), 93–100 (2011).
- Cornett L, Simms J. At the ‘heart’ of the matter: an exploration of the psychological impact of living with congenital heart disease in adulthood. J. Health Psychol. doi:10.1177/1359105312471569 (2013) (Epub ahead of print).
- McElhinney DB, Hennesen JT. The Melody® valve and Ensemble® delivery system for transcatheter pulmonary valve replacement. Ann. NY Acad. Sci. 1291, 77–85 (2013).
- Karimov JH, Massiello AL, Fukamachi K. Overview of current sutureless and transcatheter mitral valve replacement technology. Expert Rev. Med. Devices 10(1), 73–83 (2013).
- Roushdy AM, Abdelmonem N, El Fiky AA. Factors affecting vascular access complications in children undergoing congenital cardiac catheterization. Cardiol. Young 22(2), 136–144 (2012).
- Khoo SW, Han DC. The use of ultrasound in vascular procedures. Surg. Clin. North Am. 91(1), 173–184 (2011).
- Pirotte T. Ultrasound-guided vascular access in adults and children: beyond the internal jugular vein puncture. Acta Anaesthesiol. Belg. 59(3), 157–166 (2008).
- Das R, Ahmed K, Athanasiou T, Morgan RA, Belli AM. Arterial closure devices versus manual compression for femoral haemostasis in interventional radiological procedures: a systematic review and meta-analysis. Cardiovasc. Intervent. Radiol. 34(4), 723–738 (2011).
- Gorenflo M, Boshoff DE, Heying R et al. Bailout stenting for critical coarctation in premature/critical/complex/early recoarcted neonates.Catheter. Cardiovasc. Interv. 75(4), 553–561 (2010).
- Peters B, Ewert P, Berger F. The role of stents in the treatment of congenital heart disease: current status and future perspectives. Ann. Pediatr. Cardiol. 2(1), 3–23 (2009).
- Morgan G, Lee KJ, Chaturvedi R, Benson L. A biodegradable device (BioSTAR) for atrial septal defect closure in children. Catheter. Cardiovasc. Interv. 76(2), 241–245 (2010).