Editorial - Research on Chronic Diseases (2018) Volume 2, Issue 1
Heat shock protein aggregation and chronic kidney disease
- *Corresponding Author:
- Ian James Martins
Centre of Excellence in Alzheimer’s Disease Research and Care
Sarich Neuroscience Research Institute
Edith Cowan University
Verdun Street, Nedlands, Australia
E-mail: i.martins@ecu.edu.au
Abstract
Keywords
global, chronic kidney disease, autoimmune disease, Sirtuin 1, heat shock protein, amyloid beta, aggregation, mitophagy, core body temperature, treatment
Editorial
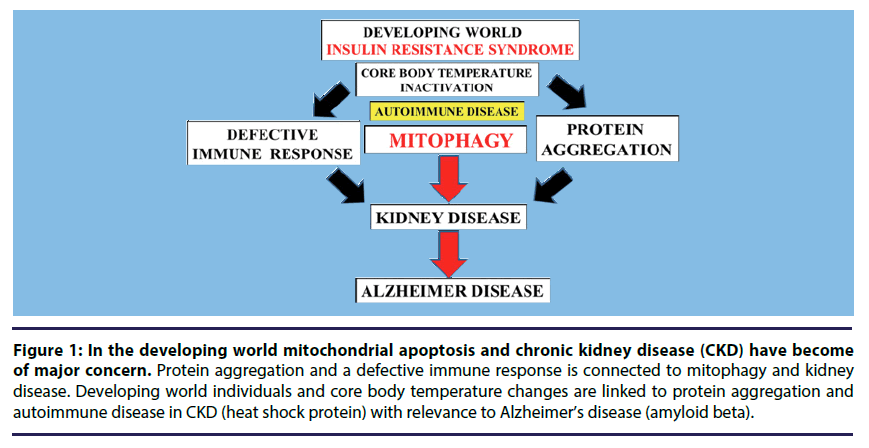
Chronic Kidney Disease (CKD) has now become of major concern [1-5] to various global health diseases such as cardiovascular disease, diabetes [6,7] and neurodegenerative diseases [8]. In the developed and developing world the high cost of kidney treatment may reach 40-70 billion dollars. In the developing world mitochondrial apoptosis in CKD has become important with relevance to acute kidney injury that may lead to end stage renal disease. Interests in environmental factors that may induce mitochondrial apoptosis and kidney disease [9,10] has accelerated with the discovery of the heat shock gene Sirtuin 1 (Sirt 1) that is connected to heat shock proteins (HSP), mitophagy and programmed cell death (Figure 1) [11]. The nuclear receptor Sirt 1 and HSP play a major role in the maintenance of kidney health and disease with Sirt 1 repression and HSP critical to maintain kidney cell survival [12-17].
Figure 1: In the developing world mitochondrial apoptosis and chronic kidney disease (CKD) have become of major concern. Protein aggregation and a defective immune response is connected to mitophagy and kidney disease. Developing world individuals and core body temperature changes are linked to protein aggregation and autoimmune disease in CKD (heat shock protein) with relevance to Alzheimer’s disease (amyloid beta).
Sirt 1 is connected to core body temperature, autoimmune and mitochondrial disease [11,18,19] with Sirt 1 repression connected to the control of the adaptive immunity and linked to acute kidney injury. Core body temperature disturbances inactivate the heat shock gene Sirt 1 with acceleration of protein aggregation that involve the HSP and amyloid beta [11] and linked to acute kidney injury. The kidney is important in the clearance of amyloid beta [20,21] and HSP is important to kidney amyloid beta clearance and formation of amyloid beta oligomers that are associated with kidney and Alzheimer’ disease [11,22-24]. Sirt 1 and autoimmune disease of the kidney [12,13,22-24] are now intertwined and involve dysregulation of immunogenic HSP and amyloid beta proteins with primary defect in mitochondrial apoptosis [11].
In the developing world the global chronic disease epidemic has major implications with relevance to Sirt 1 gene inactivation and CKD. Repressors of Sirt 1 [25] such as gram negative bacterial lipopolysaccharides (LPS) should be measured in the plasma to avoid CKD in the developing world [26-28]. Xenobiotics and the developing world are closely associated [29] and CKD will present elevated xenobiotics that enter the brain and associated with Sirt 1 inactivation and neuron apoptosis with relevance to Alzheimer’s disease and neurodegenerative diseases. Developing world individuals that contain Sirt 1 inhibitors such as LPS/xenobiotics may be referred to as environment sensitive individuals to core body temperature changes with accelerated HSP-amyloid beta aggregation that induces acute kidney disease with chronic renal failure. Gram negative bacterial amyloid peptide [30] may further interfere with HSP-amyloid beta aggregation [11] with acceleration of kidney associated amyloidosis and mitochondrial apoptosis.
Genomic medicine [31,32] is now critical to the treatment of mitophagy in CKD without genomic medicine treatment in nephrology the kidney is the major concern in the global chronic disease epidemic. In the developing world various nutrients [11] need to be consumed to prevent uncontrolled protein aggregation that lead to kidney damage. In developing world individuals the pathway for immunogenic HSP-amyloid beta pathway is defective [11] and linked to autoimmune disease, mitophagy and CKD. Diabetics and CKD is now of major concern for Alzheimer’s disease with Sirt 1 now closely linked to neurodegeneration and longevity [33] in developing world individuals. Appetite control may be critical to prevent autoimmune disease [18] with the kidney the most sensitive organ to undergo accelerated autoimmune disease in the global chronic disease epidemic.
Conclusion
Environmental factors have become of major concern to mitophagy and kidney disease in individuals from the developing world. Heat shock gene Sirt 1 inactivation leads to protein aggregation and chronic kidney disease. Food consumption in the developing world is of major interest to reverse and stabilize uncontrolled protein aggregation and maintain Sirt 1 activation to prevent kidney cell apoptosis in these populations.
Acknowledgement
This work was supported by grants from Edith Cowan University, the McCusker Alzheimer’s Research Foundation and the National Health and Medical Research Council.
References
- Barsoum RS. Chronic kidney disease in the developing world. N. Engl. J. Med. 354, 997–999 (2006).
- Nugent RA, Fathima SF, Feigl AB, et al. The burden of chronic kidney disease on developing nations: a 21stcentury challenge in global health. Nephron. Clin. Pract. 118, c269–277 (2011).
- Hamer RA, El Nahas MA. The burden of chronic kidney disease. Is rising rapidly worldwide. BMJ. 332, 563–564 (2006).
- Hill NR, Fatoba ST, Oke JL, et al. Global Prevalence of Chronic Kidney Disease – A Systematic Review and Meta-Analysis. PLoS One. 11, e0158765 (2016).
- Glassock RJ, Warnock DG, Pierre Delanaye. The global burden of chronic kidney disease: estimates, variability and pitfalls. Nat. Rev. Nephrol. 13, 104–114 (2017).
- Arnold R, Issar T, Krishnan AV, et al. Neurological complications in chronic kidney disease. JRSM Cardiovasc. Dis. 5, 2048004016677687 (2016).
- Tabish SA. Is Diabetes Becoming the Biggest Epidemic of the Twenty-first Century? Int. J. Health. Sci. (Qassim). 1, V–VIII (2007).
- Venkat Narayan KM. The mysteries of type 2 diabetes in developing countries. Bull. World. Health. Organ. 94, 241–242 (2016).
- Bhargava P, Schnellmann RG. Mitochondrial energetics in the kidney. Nat. Rev. Nephrol.10, 629–646 (2017).
- Duann P, Lin PH. Mitochondria Damage and Kidney Disease. Adv. Exp. Med. Biol. 982, 529–551 (2017).
- Martins IJ. Heat Shock Gene Inactivation and Protein Aggregation with Links to Chronic Diseases. Diseases. 6(39), 1–5 (2018).
- Hao C-M, Haase VH. Sirtuins and Their Relevance to the Kidney. J. Am. Soc. Nephrol. 21, 1620–1627 (2010).
- Kong L, Wu H, Zhou W, et al. Sirtuin 1: A Target for Kidney Diseases. Mol. Med. 21, 87–97 (2015).
- Rao SR. The role of heat shock proteins in kidney disease. J. Transl. Int. Med. 4, 114–117 (2016).
- Sreedharan R, Van Why SK. Heat shock proteins in the kidney. Pediatr. Nephrol. 31, 1561–1570 (2016).
- Wang Z, Gall JM, Bonegio RG, et al. Induction of heat shock protein 70 inhibits ischemic renal injury. Kidney. Int. 79, 861–870 (2011).
- Chebotareva N, Bobkova I, Shilov E. Heat shock proteins and kidney disease: perspectives of HSP therapy. Cell. Stress. Chaperones. 22, 319–343 (2017).
- Martins IJ. Appetite Control and Biotherapy in the Management of Autoimmune Induced Global Chronic Diseases. J. Clin. Immunol. Res. 2, 1–4 (2018).
- Segelmark M, Hellmark T. Autoimmune kidney diseases. Autoimmun. Rev. 9, A366–A371 (2010).
- Xiang Y, Bu XL, Liu YH, et al. Physiological amyloid-beta clearance in the periphery and its therapeutic potential for Alzheimer’s disease. Acta.Neuropathol. 130, 487–499 (2015).
- Liu YH, Xiang Y, Wang YR, et al. Association between Serum Amyloid-Beta and Renal Functions: Implications for Roles of Kidney in Amyloid-Beta Clearance. Mol. Neurobiol.52, 115–119 (2015).
- Khalighi MA, Wallace DW, Palma-Diaz MF. Amyloid nephropathy. Clin. Kidney. J. 7, 97–106 (2014).
- Dember LM. Amyloidosis-associated kidney disease. J. Am. Soc. Nephrol.17, 3458–3471 (2006).
- Kumar S, Thangakani MA, Nagarajan R, et al. Autoimmune Responses to Soluble Aggregates of Amyloidogenic Proteins Involved in Neurodegenerative Diseases: Overlapping Aggregation Prone and Autoimmunogenic regions. Scientific. Reports. 6, 22258 (2016).
- Martins IJ. The Future of Genomic Medicine Involves the Maintenance of Sirtuin 1 in Global Populations. Int. J. Mol. Biol. 2, 00013 (2017).
- Chen H, Zhu J, Liu Y, et al. Lipopolysaccharide Induces Chronic Kidney Injury and Fibrosis through Activation of mTOR Signaling in Macrophages. Am. J. Nephrol. 42, 305–17 (2015).
- Takahashi K, Mizukami H, Kamata K, et al. Amelioration of acute kidney injury in lipopolysaccharide-induced systemic inflammatory response syndrome by an aldose reductase inhibitor. fidarestat. PLoS One. 7, e30134 (2012).
- Nozaki Y, Hino S, Ri J, et al. Lipopolysaccharide- Induced Acute Kidney Injury Is Dependent on an IL–18 Receptor Signaling Pathway. Int. J. Mol. Sci. 18, (2017).
- Martins IJ. Single Gene Inactivation with Implications to Diabetes and Multiple Organ Dysfunction Syndrome. J. Clin. Epigenet. 3, 1–8 (2017).
- Martins IJ. Antimicrobial Drugs and Bacterial Amyloid Peptide Induce Toxic Manifestations in Chronic Diseases. EC. Pharmacol. Toxicol. (6)1, 01–04 (2018).
- Martins IJ. Genomic Medicine and Acute Cardiovascular Disease Progression in Diabetes. Int. J. Med. Stud. 3, 124–130, 2018.
- Martins IJ. Genomic Medicine and Endocrine Autoimmunity as Key to Mitochondrial Disease. Glob J Endocrinological Metab. 2, 1–3 (2018).
- Martins IJ. Anti-Aging gene linked to Appetite Regulation determines longevity in Human and Animals. Int. J. Aging. Res. 1, 1–4, (2018).