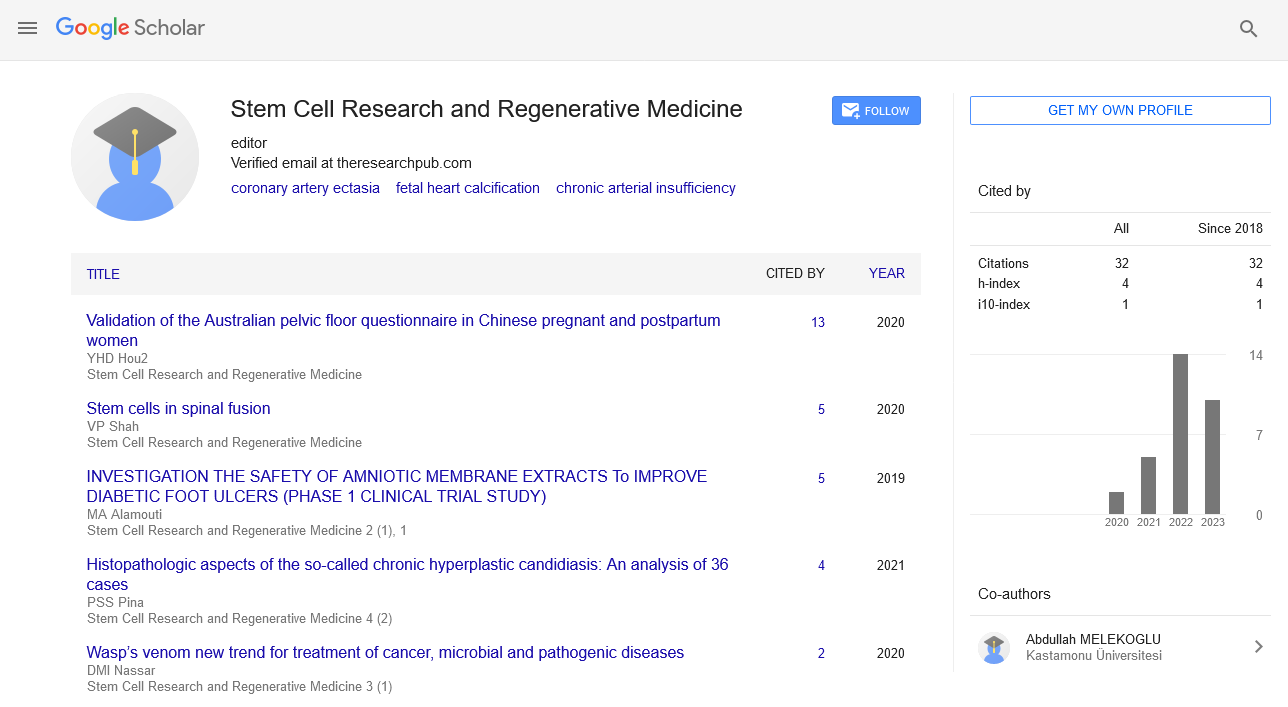
Review Article - Stem Cell Research and Regenerative Medicine (2023) Volume 6, Issue 3
Spinal Teratoma to Investigate Cognitive Nerve Dysfunction and Growth
Afzal Khan*
Department of Stem Cell and Research, Macao
Department of Stem Cell and Research, Macao
E-mail: afzaalnahk@edu.co
Received: 01-June-2023, Manuscript No. srrm-23-101878; Editor assigned: 05-June-2023, Pre-QC No. srrm-23- 101878 (PQ); Reviewed: 19-June- 2023, QC No. srrm-23-101878; Revised: 24-June-2023, Manuscript No. srrm-23-101878 (R); Published: 30-June-2023, DOI: 10.37532/ srrm.2023.6(3).57-60
Abstract
Motor neuron disorders (MNDs) are a heterogeneous group of disorders that affect the brain and/or spinal cord motor neurons (spMN), spinal cord sensory neurons, and the muscular system. Although they have been studied for decades, a comprehensive understanding of the underlying molecular mechanisms is still lacking. Therefore, effective treatments are lacking. Model organisms and relatively simple two-dimensional cell culture systems have contributed significantly to our current knowledge of neuromuscular disease pathogenesis. However, in recent years, 3D human in vitro models have changed the disease modeling landscape. Although cerebral organoids have received the most attention, interest in spinal cord organoids (SCOs) has also increased. Pluripotent stem cell (PSC)-based protocols to generate SpC-like structures, in some cases including adjacent mesoderm and derived skeletal muscle, are constantly being refined and used to study early neuromuscular development and disease in humans. applied. In this review, we outline the development of a human PSC-derived model to generate spMNs and recapitulate SpC development. We also describe how these models have been applied to investigate the basis of human neurological and neurodegenerative diseases. Finally, we outline the main challenges to overcome in order to generate more physiologically relevant human SpC models and propose some exciting new perspectives.
Keywords
Motor neuron diseases (MNDs) • Teratoma • Spinal teratoma (SCOs)
Introduction
The spinal cord (SpC) plays an important role in the transmission of signals between the brain and the peripheral body, including the transmission of sensory information and motor instructions, reflex movements, and the generation of motor activity [1]. These motor functions involve different cell types, interneurons and motor neurons (MNs), cholinergic neurons found in the anterior horn and brainstem of SpC. The anatomical placement of spinal cord MNs (spMNs) within SpCs correlates with the muscles they innervate. According to them, spMN will be organized into his spMN pool [2]. This is a contiguous group of spMNs with similar intrinsic properties that are anatomically arranged to connect to a single target muscle in the periphery [3]. spMNs are innervated either directly from upper MNs located in the motor cortex of the brain or after one or more conductions on spinal cord interneurons. spMN can also be classified according to various characteristics, such as: B. Based on axonal morphology and size, firing rate, or the rostralcaudal segment of SpCs where they are located. Based on this, spMN are classified as: It is branched and located in the brainstem and is responsible for controlling the muscles of the face and neck [4]. It innervates the internal organs, part of the autonomic nervous system, and regulates smooth muscles. Somatic cells then innervate skeletal muscles [5].
Most of our knowledge about spMN diversity and biology, as well as SpC evolution, has come from in vivo studies in model organisms. However, advances in in vitro models for studying human development and disease over the past decade have enabled scientists to address questions that were difficult to answer in vivo. Three-dimensional tissue-like models generated in vitro are complex systems derived from pluripotent stem cells (PSCs), resulting from the self-assembly of multiple cell types, exhibiting unique cellular architecture and representing the functional characteristics of organs. partially reproduced. These tissue-like structures are grouped together under the generic term teratomas (ORGs). Spheroids represent a simpler tissue-like model, usually composed of homogeneous cell types and obtained by combining one or more individually structured cell types with limited self-organization properties in 3D culture. You can His ORG of the nervous system is named after the specific anatomical region it mimics. B. Cortical teratoma, retinal teratoma, hypothalamic teratoma, or SpC teratoma (SCO). In 2013, Sasai’s group published breakthroughs in the selforganizing ability of human embryonic stem cells (hESCs) when cultured under specific suspension conditions, including axial polarity, inside-out stratification, patterning and curved morphology. reported insightful insights. human neocortex. Around the same time, Knoblich’s team developed the first of his 3D culture models to mimic human brain development, coining the term “cerebral organoids.” Shortly thereafter, Camp et al [6]. applied scRNAseq technology to pinpoint the gene expression program that drives such human induced pluripotent stem cell (hiPSC)-derived cerebral teratomas to promote neocortical development in human fetuses. reproduced and eventually shown to lead to the first cell-driven generation. Atlas of human corticogenesis.
Despite our comprehensive understanding of SpC anatomy, its cellular organization, neural circuitry, and developmental development, many fundamental questions remain unanswered. How do extracellular signals regulate the expression of lineagematched transcription factors (TFs) during spMN development and determine the signaling state of the regulatory machinery that maintains this developmental fate? How can these signals be manipulated to distinguish between specific her MN subtypes, and how is stratified organization of SpCs achieved at the molecular level? Could abnormalities in SpC development in humans underlie or contribute to the development of neurodegenerative diseases (NDs)? To answer these and other questions, the scientific community has spent the last decade Over the years, we have used stem cell (SC)-based in vitro approaches as a valuable complement to in vivo studies [7].
Although various reports continue to streamline protocols for the generation and study of cortical teratomas, SCO have been tracked significantly less. However, in recent years, SCO has emerged as a useful resource for studying her SpC development in humans. In this review, we provide an overview of the evolution of SpCs and spMNs, the methods used to generate such cultures, their evolution, and the current best practices that have contributed significantly to the study and common modeling of SpC evolution. Summarize the leading edge approach. Neuromuscular diseases such as spinal muscular atrophy (SMA) and amyotrophic lateral sclerosis (ALS). Furthermore, to what extent these approaches reflect human physiology and pathology, and what are the recent advances and challenges in this area for enhancing the formation of human SpClike structures in vitro [8].
Morphogen gradients direct SpC development
By late gastrulation, germ layer segregation in vertebrate embryos is nearly complete, and multipotent cells in the epiblast begin to differentiate into lineage-restricted progenitor cells. At the same time, neural induction occurs, and neuroepithelial cells form a sheet-like neural plate, undergoing a series of rapid morphological changes (convergence, expansion, elevation, and fusion) to become the primitive neural tube. This process occurs in her two modules, with the primary neuropil forming the brain and rostral SpCs and the secondary neuropil contributing to the caudal SpC [9]. In addition, SpCs undergo continuous growth during embryonic development, called axial elongation, resulting in four distinct anatomical regions along the rostral-caudal (RC) axis: cervical, thoracic, lumbar and sacral. His SpC segment is formed. As SpCs develop, new cells are continuously generated from a proliferating posterior growth zone called the tailbud. This is a transient structure at the caudal end of the embryo, composed of undifferentiated cells with SC-like properties. These cells, termed neuromesoderm progenitor cells (NMPs), are a bipotent cell population with self-renewal capacity, present only in the embryonic stage, and characterized by co-expression of TF TBXT and SOX2. A tightly regulated cascade of wingless integration signaling (WNT) and fibroblast growth factor (FGF) signaling maintains NMPs in an undifferentiated progenitor state.
The ability of NMP to contribute to multiple germ layers was demonstrated by performing transplantation experiments in chicken and GFP mouse embryos. The resulting chimeric organisms demonstrated that cells within the transplanted NMP contributed to both the neural and mesodermal sublineages of the host embryo, particularly the lumbar region of SpCs and the surrounding paraxial mesoderm. Lineage tracing experiments by Gouti et al. helped characterize the NMP transcriptome and provided conclusive evidence that these cells indeed gave rise to both SpC and caudal embryonic muscle. However, it remains controversial whether all NMPs first acquire a specific axial identity and then commit to a neural identity or vice versa [10].
Increasing complexity and heterogeneity
With delayed gastrulation, germ layer segregation in vertebrate embryos is nearly complete, and multipotent cells in the epiblast begin to differentiate into lineagerestricted progenitor cells. At the same time, neural induction occurs, and neuroepithelial cells form a sheet-like neural plate, which undergoes rapid morphological changes (convergence, expansion, swelling, and fusion) to become a primitive neural tube. This process He takes place in two modules, the primary neuropil forming the brain and rostral SpCs, and the secondary neuropil contributing to the caudal SpCs. In addition, SpCs undergo continuous growth during embryogenesis called axial elongation, resulting in the formation of four distinct anatomical regions along the rostral-caudal (RC) axis.
Cervical spine, thoracic spine, lumbar spine, sacrum. As SpCs develop, new cells are continuously generated from a proliferating posterior growth zone called the tail bud. This is a transient structure at the caudal end of the embryo, composed of undifferentiated cells with SC-like properties. These cells, termed neuromesoderm progenitor cells (NMPs), are a impotent cell population with self-renewal capacity, present only in the embryonic stage, and characterized by coexpression of TF TBXT and SOX2. A tightly regulated cascade of wingless integration signaling (WNT) and fibroblast growth factor (FGF) signaling maintains NMPs in an undifferentiated progenitor state. The ability of NMP to contribute to multiple germ layers was demonstrated by performing transplantation experiments in chick and her GFP mouse embryos. The resulting chimeric organisms demonstrated that cells within the transplanted NMP contributed to both the neural and mesodermal sub lineages of the host embryo, particularly the lumbar region of SpCs and the surrounding paraxial mesoderm. Lineage tracing experiments by Gouti et al. This helped characterize the NMP transcriptome and provided definitive evidence that these cells indeed gave rise to both her SpCs and the caudal embryonic muscle. However, it remains controversial whether all NMPs first acquire a specific axial identity and then commit to a neural identity or vice versa.
Discussion
Microfluidic devices are a more reliable alternative to generate multiple morphogen gradients simultaneously in an automized and highly controlled manner. Indeed, Demers and colleagues were able to form a neural tube slice by using a microfluidic device creating gradients of RA, SHH and BMP. Using a hexagonal microfluidic system to deliver RA and GDF11, Lim and colleagues produced branchial, thoracic and lumbar spMNs from a monolayer of hiPSCs. This study proved the applicability of microfluidic devices for increasing anterior–posterior heterogeneity of spMNs, albeit in a 2D culture. Microfluidic devices are also starting to be applied in MND research using 3D cultures as the main model system for drug screening purposes. Recent studies used these devices to generate gradients of drugs, such as demonstrated their suitability for optimizing drug concentration and efficacy. Teratoma cultured in combination with microfluidic devices are often termed organon- a-chip, and they have been recently been applied in the MND field.
Conclusions
Despite decades of research and thousands of studies, it is clear that there is no effective treatment for MND. This is due in part to an incomplete understanding of the molecular and cellular basis of these human diseases, or of the development of the human neuromuscular system. Although human in vitro 2D cultures are highly informative in identifying cell-derived virulence factors and their responses to external regulation, a large number of non-cell-autonomous factors that contribute to MND pathogenesis are mediated by these simplifications. More complex and controllable 3D models have overcome this important limitation and explored the interactions between affected neurons and their muscle targets, the immunomodulatory role of glial cells, or the nutritional and Allows investigation of tissue homeostasis support. Cortical teratomas are the most developed and used to study human cortical development, but in recent years, a number of SpC-like tissue models have emerged to explore how SpCs develop and their pathology. There is an increasing understanding of how the neuromuscular system functions and deteriorates in stressful situations. To research the initial stages of neural tube creation, these models incorporate gastruloids; trunk-like structures to study neural tube elongation and somitogenesis; and more developed SCOs to study MNDs. In certain cases, these models also include the muscular and cortical components of the corticomotor system. We could most likely only fully explain how diseases manifest once we have a complete understanding of the human SpC’s embryonic development. The benefits they offer to investigate human-specific processes in an experimentally tractable way, as well as the ongoing refining of these 3D in vitro models, hold considerable promise for elucidating the spatiotemporal mechanisms of SpC morphogenesis and the molecular and cellular causes of MNDs. The addition of functional immune cells and vascular networks, the creation of morphogen gradients, and the inclusion of physiologically appropriate ECMs, among other developments, make SCOs a potent system to cover those knowledge gaps. These models offer a platform for discovering new therapy candidates and testing their efficacy, maybe in a patient-specific way, as well as for forecasting the emergence of disease phenotypes and their response to manipulation. Additionally, transplantation research in murine models is demonstrating that genome-engineered SCOs or their derivatives could be a useful source for stem cell therapy for MNDs, despite the fact that such applications are still distant from being considered clinically. The implementation of the swiftly developing discoveries and methods indicated here should help overcome the significant challenges outlined here, accelerating the search for those desperately needed medicines.
References
- Acharya UR, Faust O, Sree V et al. Linear and nonlinear analysis of normal and CAD-affected heart rate signals. Comput Methods Programs Bio. 113, 55–68 (2014).
- Kumar M, Pachori RB, Rajendra Acharya U et al. An efficient automated technique for CAD diagnosis using flexible analytic wavelet transform and entropy features extracted from HRV signals. Expert Syst Appl. 63, 165–172 (2016).
- Davari Dolatabadi A, Khadem SEZ, Asl BM et al. Automated diagnosis of coronary artery disease (CAD) patients using optimized SVM. Comput Methods Programs Bio. 138, 117–126 (2017).
- Patidar S, Pachori RB, Rajendra Acharya U et al. Automated diagnosis of coronary artery disease using tunable-Q wavelet transform applied on heart rate signals. Knowl Based Syst. 82, 1–10 (2015).
- Giri D, Acharya UR, Martis RJ et al. Automated diagnosis of coronary artery disease affected patients using LDA, PCA, ICA and discrete wavelet transform. Knowl Based Syst. 37, 274–282 (2013).
- Maglaveras N, Stamkopoulos T, Diamantaras K et al. ECG pattern recognition and classification using non-linear transformations and neural networks: a review. Int J Med Inform. 52,191–208 (1998).
- Rajkumar R, Anandakumar K, Bharathi A et al. Coronary artery disease (CAD) prediction and classification-a survey. Breast Cancer. 90, 945-955 (2006).
- Dwyer, Claire. ‘Highway to Heaven’: the creation of a multicultural, religious landscape in suburban Richmond, British Columbia. Soc Cult Geogr. 17, 667-693 (2016).
- Fonseca, Frederico Torres. Using ontologies for geographic information integration. Transactions in GIS.6,231-257 (2009).
- Harrison, Paul. How shall I say it…? Relating the nonrelational .Environ Plan A. 39, 590-608 (2007).
Indexed at, Google Scholar, Crossref
Indexed at, Google Scholar, Crossref
Indexed at, Google Scholar, Crossref
Indexed at, Google Scholar, Crossref
Indexed at, Google Scholar, Crossref
Indexed at, Google Scholar, Crossref