Research Article - Journal of Interventional Nephrology (2026) Volume 9, Issue 1
Extended Follow-Up of an Endovascular Venous Anastomotic Connector for Arteriovenous Graft Creation in Hemodialysis: A Multicenter Study
Ari Kramer1, KMLST Moorthi2, Lisa Grant*3 and Terry Litchfield4
1Spartanburg Regional Medical Center, Spartanburg, SC
2Northwestern University Feinberg School of Medicine, Chicago, IL
3LB Grant Clinical Research Consulting, St. Louis Park, MN
4Echelon Development Group, Blanchardville, WI
- *Corresponding Author:
- Lisa Grant
LB Grant Clinical Research Consulting, St. Louis Park, MN
E-mail: lgrant@phraxis.com
Received: 13-Feb-2026, Manuscript No: oain-26-184846, Editor Assigned: 16-Feb-2026, pre QC No: oain-26- 184846 (PQ), Reviewed: 20-Feb-2026, QC No oain-26-184846, Revised: 23-Feb-2026, Manuscript No: oain-26- 184846 (R), Published: 28-Feb-2026, DOI: 10.37532/oain.2026.9(1).418- 426
Abstract
Objective: Arteriovenous grafts (AVGs) are a key access option in hemodialysis but are prone to venous stenosis and thrombosis, often necessitating frequent interventions. The EndoForce endovascular connector offers a less-traumatic alternative to traditional sutured techniques. The purpose of this extended follow-up study is to evaluate the long-term cumulative patency of the EndoForce connector.
Methods: In this multicenter follow-up study, 37 participants from the original pivotal trial who underwent at least one intervention were observed for an additional six months from the last intervention. Outcomes included cumulative patency and intervention rates beyond the initial study period.
Results: Thirteen of 37 participants underwent 22 additional interventions (1.32 interventions per patient-year). Cumulative patency 180 days into the extended follow-up was 78.4%. All interventions were successful; no device-related complications such as migration, kinking, or fracture were reported.
Conclusion: These findings suggest that the EndoForce endovascular connector supports durable AVG patency and a low intervention burden, reinforcing its potential role in modern end stage renal disease (ESRD) vascular access planning.
Clinical Trial Registration: ClinicalTrials.gov Identifier: NCT02532621.
Keywords
Endovascular Procedures; Vascular Grafting; Arteriovenous Anastomosis; Graft Survival; Renal Dialysis; Patency
Introduction
Despite the recent resurgence of arteriovenous grafts (AVGs) as an access option in the end stage renal disease (ESRD) “life-plan [1] , as articulated by the latest Kidney Disease Outcomes Quality Initiative (KDOQI) Guidelines for Vascular Access, traditional sutured AVG patency outcomes continue to be plagued by venous anastomotic stenosis, leading to thrombosis and eventual AVG abandonment [2] . Venous stenosis in AVGs most frequently arises from progressive neointimal hyperplasia caused by surgical trauma, inflammation, and flow turbulence [3] . The EndoForce (formerly InterGraft) Venous Anastomotic Connector offers an alternative, less-traumatic endovascular venous anastomotic technique for AVG creation. It was recently studied in a multicenter, single-arm pivotal trial, evaluating the safety and effectiveness of AVG implantation using the connector throughout a 6-month follow-up period [4] . A total of 158 participants were enrolled from 10 study sites, among which 144 participants were evaluable at six months. The study is registered on clinicaltrials.gov with identifier NCT02532621. By Kaplan Meier survival analysis, cumulative patency was 92.08% with a lower 95% confidence bound of 86.98%. This compares favorably with the AVG outcomes described in a recent meta-analysis, which reported a 6-month primary patency rate of 56% and a 6-month cumulative patency of 80% [5] . The Enforce connector's endovascular design may reduce surgical trauma at the venous anastomosis, mitigate intimal hyperplasia, and promote laminar flow compared to traditional sutured techniques, potentially accounting for these favorable patency outcomes.
The authors concluded that the results demonstrate that the Enforce endovascular connector can be used for successful creation of a graft for hemodialysis, with acceptable cumulative patency and safety profile at six months. This pivotal trial also provided sufficient safety and effectiveness evidence for premarket approval from the United States Food and Drug Administration (FDA) in 2025.
By study design, participant follow-up was limited to six months; therefore, the total number of interventions required maintaining AVG patency beyond six months was not reported. The purpose of this extended follow-up study is to evaluate the number of interventions performed to achieve cumulative patency for an additional six months beyond the most recent intervention reported in the Enforce pivotal trial.
Methods
Study Design and Outcomes
This was retrospective study of the original data set of the EndoForce pivotal trial, with an extended follow-up of participants from the pivotal trial who had at least one graft intervention. No new procedures were required. In 2023, participating sites collected data via medical record review and/or interviews. Outcomes evaluated included post-intervention graft patency information and the number and details of any new graft interventions during an additional six months of follow-up, starting from the date of the last reported graft intervention in the pivotal trial.
Participants for Extended Follow-up
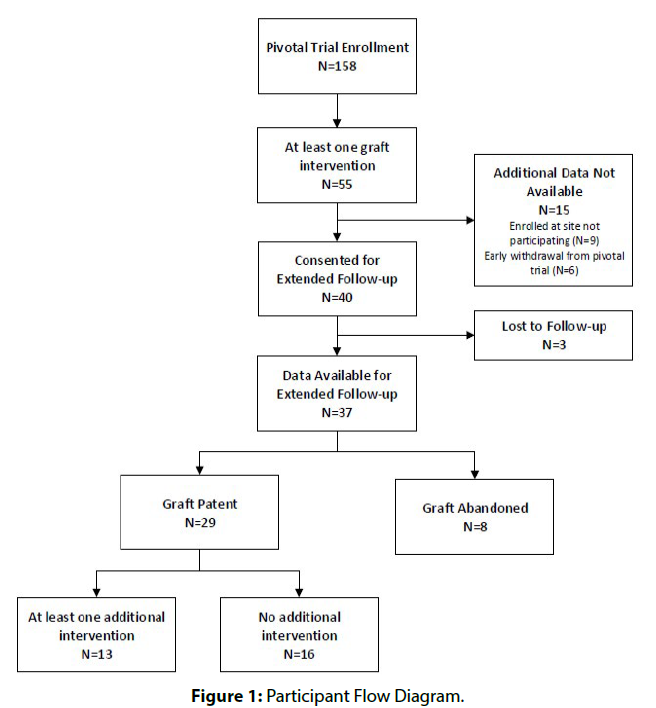
Of the 158 pivotal trial participants, 55 had at least one graft intervention, six were excluded (withdrawn or graft abandonment), and nine were from non-participating sites. Of the 40 eligible participants, three were lost to follow-up, leaving 37 for final analysis [Figure 1] . Institutional review board (IRB) informed consent requirements were waived for deceased participants; otherwise, written informed consent was obtained.
Data Collection and Monitoring
Standardized forms were completed and verified by site investigators. Independent monitors reviewed source data and confirmed database accuracy. Data were recorded by study coordinators on standardized data collection forms provided by the Sponsor. All completed data collection forms were reviewed and approved by the site principal investigator. Data were entered into a database by independent data entry personnel.
Data Monitoring and Quality Assurance
Monitoring was performed for source data verification of information recorded on the data collection forms, including review of operative notes for graft intervention procedures and review of available death records, where applicable. A monitor also verified that data entered in the database matched the information recorded on data collection forms and that no data entry errors occurred.
Statistical Analysis
Descriptive statistics including mean and standard deviation were used to summarize continuous variables. Binary outcomes were represented as counts and proportions. Kaplan-Meier survival analysis was performed to estimate the probability of an additional intervention over time. Analyses were performed by an independent statistician using R version (4.3.2). (R Foundation for Statistical Computing, Vienna, Austria).
Results
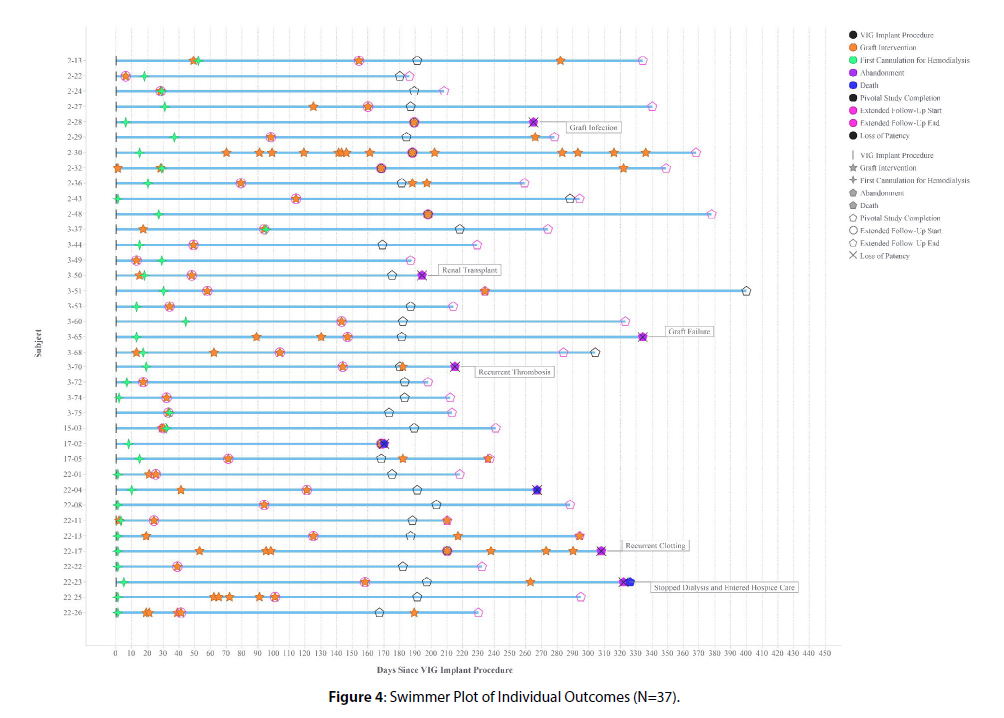
Baseline characteristics are reported in [Table 1] , along with the baseline characteristics of the AVG Intervention Cohort, the No AVG Intervention Cohort, and the Pivotal Trial Cohort. Of 37 participants, five experienced graft abandonment and three died during follow-up. Graft patency and intervention data were collected up to the date of these terminal events. Graft patency and intervention information are summarized in [Table 2] . Among all 37 participants with extended follow-up data, continued access at the end of the extended follow-up period was reported in 29 participants. Thirteen underwent 22 additional interventions, yielding an intervention rate of 1.32 per patient-year, similar to the 1.22 per patient-year rate reported in the pivotal trial. Of these, eight (36%) targeted the endovascular venous anastomotic connector itself. All interventions restored flow and were completed without complications, consistent with technical success. There were no reports of migration, kink, compression, or other damage/defect to the implanted connector. Specific outcomes for each participant are presented in [Figure 4] .
| Characteristic | Extended Follow-up Cohort (N=37) Mean ± SD or N (%) | AVG Intervention Cohort (N=55) Mean ± SD or N (%) | No AVG Intervention Cohort (N= 103) Mean ± SD or N (%) | Pivotal Study Cohort (N=158) Mean ± SD or N (%) |
|---|---|---|---|---|
| Age, years | 61 ± 16 | 61 ± 15 | 63 ± 15 | 63 ± 15 |
| Gender: Male Female | 13 (35) 24 (65) | 21 (38) 34 (62) | 40 (39) 63 (61) | 61 (39) 97 (61) |
| Body Mass Index | 30.1 ± 9.3 | 30.2 ± 10.1 | 29.9 ± 7.7 | 30.0 ± 8.6 |
| Ethnicity: Hispanic or Latino Not Hispanic or Latino Unknown/not reported | 1 (3) 35 (95) 1 (3) | 2 (4) 52 (95) 1 (2) | 1 (1) 102 (99) 0 (0) | 3 (2) 154 (97) 1 (1) |
| Race: Asian Black or African American White | 1 (3) 27 (73) 9 (24) | 1 (2) 38 (69) 16 (29) | 0 (0) 85 (83) 18 (17) | 1 (1) 123 (78) 34 (22) |
| Diabetes mellitus | 21 (57) | 32 (58) | 66 (64) | 98 (62) |
| Obesity | 12 (32) | 16 (29) | 35 (34) | 51 (32) |
| Hypertension | 20 (54) | 28 (51) | 61 (59) | 89 (56) |
| Cardiovascular disease | 17 (46) | 24 (44) | 54 (52) | 78 (49) |
| Previous permanent vascular accesses for hemodialysis, any location: 0 1 ≥ 2 | 14 (38) 19 (51) 3 (8) | 18 (33) 27 (49) 10 (18) | 46 (45) 39 (38) 18 (17) | 64 (41) 66 (42) 28 (18) |
| Previous permanent vascular access for hemodialysis in target limb: 0 ≥ 1 | 21 (57) 16 (43) | 27 (49) 29 (53) | 46 (45) 57 (55) | 90 (57) 68 (43) |
| Current vascular access using catheter | 34 (92) | 48 (87) | 86 (83) | 134 (85) |
| Duration of hemodialysis: < 6 months ≥ 6 months and <12 months ≥ 12 months NA (not currently receiving hemodialysis) | 20 (54) 3 (8) 12 (32) 2 (5) | 27 (49) 4 (7) 21 (38) 3 (5) | 42 (41) 11 (11) 38 (37) 12 (12) | 69 (44) 15 (9) 59 (37) 15 (9) |
Table 1: Demographic and Baseline Characteristics for Extended Follow-up, AVG Intervention, No AVG Intervention, and the Pivotal Study Cohorts
| Parameter | Value (Number of participants, unless otherwise indicated) |
|---|---|
| Continued access at end of extended follow-up period | 29 |
| Terminal events during extended follow-up period Death AVG abandoned |
8 5 3 |
| Additional AVG interventions to maintain patency, during extended follow-up period 1 intervention 2 interventions ≥ 3 interventions |
13 8 3 2 |
| Total # of AVG interventions during extended follow-up (among all 13 participants with additional interventions) |
22 |
| Reason(s) for AVG intervention: Thrombosis Poor flow Outflow stenosis Cannulation difficulty Arm swelling Hand/arm pain |
16 1 1 3 2 1 |
| Stenosis identified Stenosis location(s): AVG Edge stent Central vein Outflow vein Inflow artery EndoForce |
22 9 1 6 8 2 8 |
| Intervention success | 22 |
| Major procedural complications during AVG interventions | 0 |
| Evidence of EndoForce migration, kinking, compression, fracture or other damage noted during extended follow-up | 0 |
| Total AVG interventions, pivotal trial plus extended follow-up 1 intervention 2 interventions ≥ 3 interventions |
15 10 12 |
Table 2: Summary of AVG Interventions and Outcomes in Extended Follow-up (N=37)
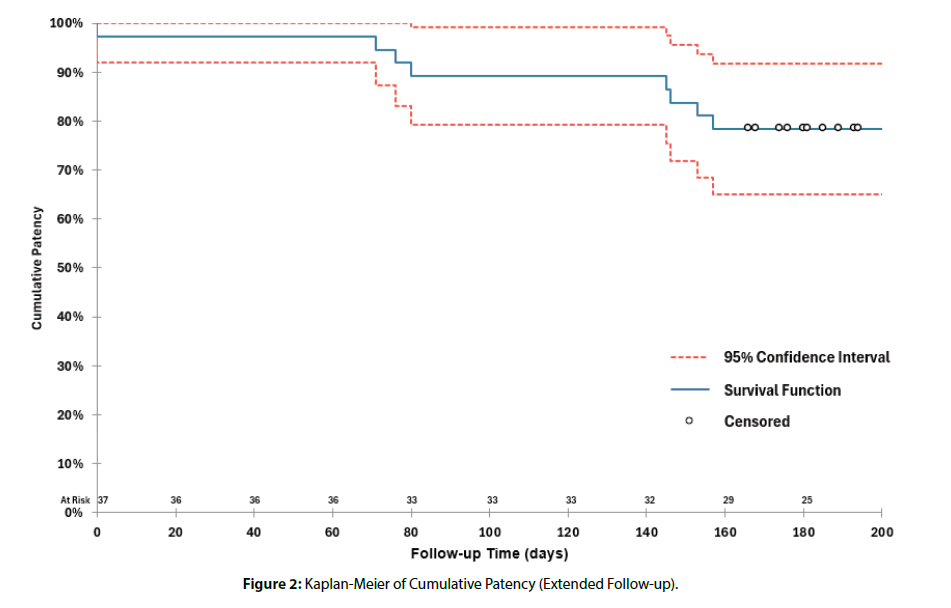
Kaplan-Meier analysis of cumulative patency in the extended follow-up period was performed, with time to loss of patency measured from the date of the last reported graft intervention in the pivotal trial. As shown in [Figure 2] , the estimated cumulative patency at 180 days into the extended follow-up period was 78.4%.
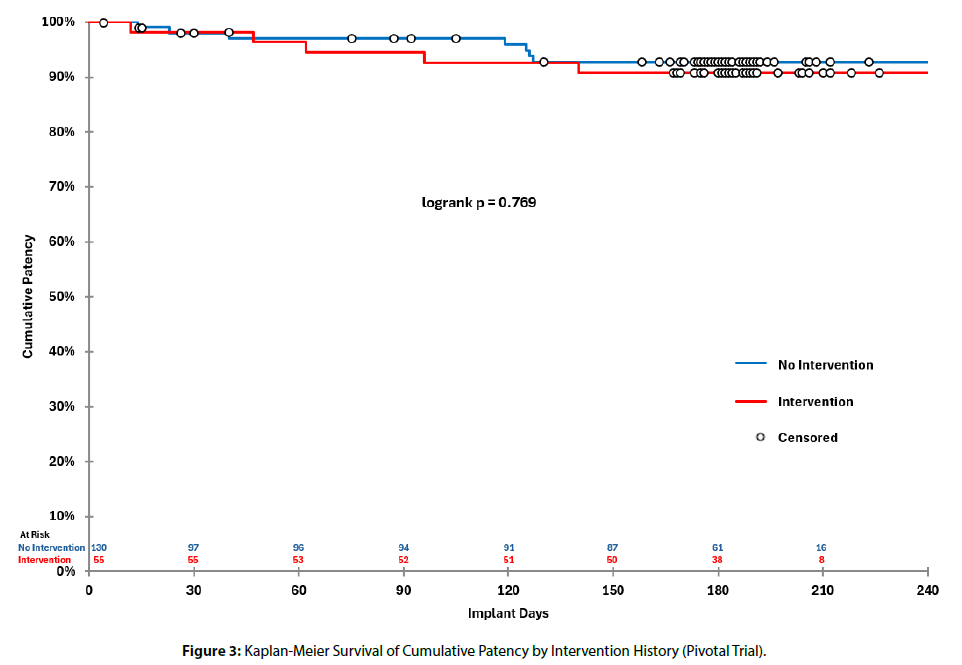
Kaplan-Meier analysis was also performed to compare cumulative patency outcomes in participants with and without graft interventions during the pivotal trial [Figure 3] . Time to loss of patency was measured from the date of implant for those without intervention, and from the date of the first intervention for those with one or more interventions during the pivotal trial. The cumulative patency at six months was nearly identical (91.79% vs. 91.80%; logrank p = 0.769).
Figure 3: Kaplan-Meier Survival of Cumulative Patency by Intervention History (Pivotal Trial).
Discussion
The Fistula First Breakthrough Initiative (FFBI), launched in 2003, shaped vascular access practices in the United States by promoting arteriovenous fistulae (AVFs) as the preferred access for hemodialysis [6] . This initiative successfully increased prevalent use of AVF from around 30% in 2003 to above 60% in 2015, surpassing the original KDOQI guideline of 40% AVF use in prevalent hemodialysis patients and 50% in incident hemodialysis patients [7] . In the ensuing years the demographics of the ESRD population have shifted dramatically, with patients aged 65 years and older comprising more than half of incident hemodialysis patients [8] .
Despite the theoretical advantages of AVFs, substantial clinical evidence has emerged documenting significant limitations in their universal application. About 5-20% of fistulae fail because of early thrombosis, and 20-50% fail to mature, requiring assisted maturation by interventional procedures [9] . These challenges are pronounced in elderly patients, who present with diminished vein quality and elasticity, higher prevalence of atherosclerotic disease and increased risk of infection [10], In elderly populations AVGs may offer several distinct advantages [11] . In patients older than 80 years, AVG placement results in higher early utilization rates (38.7% vs. 15.4% within the first six months) and reduced rate of non-use (41.8% never used vs. 63.5% of AVFs never used for hemodialysis). AVGs also demonstrated faster time to successful cannulation (median 75 days vs. 114 days for AVFs) and substantially lower maturation failure rates, in elderly cohorts. In addition to the above, elderly patients also have a higher comorbidity burden and experience a mortality rate of 30-50% within the first year of dialysis initiation. The combination of extended maturation time and high failure rate of AVFs, results in prolonged catheter dependence without the intended benefits of an AVF.
Female gender is another import demographic factor affecting fistula maturation. Even when routine preoperative vascular mapping is utilized to ensure adequate vessel size [12] , and despite a higher frequency of attempted salvage procedures [13] , women have a lower AVF maturation rate.
These complexities were taken into account in the 2019 KDOQI clinical practice guideline for vascular access, which adopted an individualized, patient-centered framework transitioning from the rigid “Fistula First-Catheter Last” mandate to the “Patient First: ESKD Life-Plan”. The emphasis here is on achieving the “right access, in the right patient, at the right time, for the right reasons” [1] . Thus, AVGs are not merely a second option for patient with inadequate vasculature, but rather represent an appropriate and often a preferable primary access choice in elderly individuals with multiple comorbidities and in patients with limited life expectancy, especially if they are already on dialysis via a dialysis catheter.
While an attractive option in a select group of patients, stenosis at the graft-vein anastomotic site is a major problem which leads to occlusion and thrombosis. This stenosis almost always arises from progressive neointimal hyperplasia. Surgical trauma, bio incompatibility and mechanical stresses all play a role in the intimal hyperplasia [14] . The current clinical treatment for AVG stenosis is mainly percutaneous transluminal angioplasty (PTA) [15] , although PTA improved the blood flow, the vascular wall injury produced by the balloon promotes hyperplasia and further stenosis. The successful prevention of stenosis is therefore essential for the longevity of AV synthetic grafts.
This extended follow-up study in 37 participants showed an estimated intervention rate of 1.32 per patient-year, which was similar to the rate observed in the pivotal trial (1.22 interventions per patient-year) builds on initial data showing favorable outcomes with the EndoForce connector. These intervention rates compare favorably with the 1.58 interventions per patient-year in a large study reported [16] . Further, a recent large retrospective United States Renal Data System study showed in the AVG group, 5,928 of 10,271 patients (57.7%) required intervention, with a therapeutic intervention rate of 1.87 per patient-year [17] .
Of note, all AVG interventions reported during extended follow-up were successful and no major procedural adverse events occurred. There were no reports of migration, kink, compression, or other damage/defect to the implanted EndoForce connector. In 24 participants (65%), there no additional new interventions were required in the extended follow-up period.
This study has several important limitations. First, the original Pivotal study was a single arm, non-randomized trial. Secondly, the extended follow up cohort was selected and analyzed post hoc, rather than identified as a variable of interest at the initiation of the Pivotal trial. Further, the comparisons made in the Pivotal trial as well as those described in this discussion are not direct comparisons, but rather historical outcomes of Febarate studies.
While this cohort had prior interventions and might represent a higher-risk group, their baseline characteristics aligned with the larger pivotal cohort, minimizing selection bias. Patency at an additional 6 months beyond the most recent intervention (78.4%) exceeds that typically reported for sutured AVGs (70%). The device's reduced intervention burden may positively impact health-related quality of life (HRQoL), which should be explored in future studies using validated tools like the Hemodialysis Access-Related Quality of Life (HARQ) instrument.
Conclusion
The results of the extended follow-up study suggest that, in addition to the patency rate at six months described in the EndoForce pivotal trial, an endovascular venous anastomosis may confer a benefit to the ESRD patient by reducing the number of interventions that need to be performed to achieve patency. Frequent interventions represent a source of quality-of-life reduction in the ESRD population [18] . Frequent interventions may lead ESRD patients to experience increased pain, anxiety, and depression due to the recurring nature of these problems and the constant worry about potential complications [19-21] . Although the reduction in interventions per year reported in this study (1.32 per patient-year) was not directly compared to the interventions per year reported in the literature (1.58-1.87 per patient-year), a potential improvement in quality of life measures should be further investigated. Since both the EndoForce pivotal trial as well as this extended follow up study does not directly compare outcomes to sutured AVGs, a meta-analytic approach to compare endovascular anastomosis of an AVG with a traditional sutured anastomosis may be warranted.
Acknowledgments
We thank the EndoForce Extended Follow-up Study investigators for providing the patient data: Jason Beaver (Flowers Hospital/Department of Surgery, Dothan, AL), Jason Burgess, MD (Surgical Specialists of Charlotte, Charlotte, NC), Loay Kabbani, MD (Henry Ford Health, Detroit, MI), Mark London, MD (The Regional Medical Center/Dialysis Access Institute, Orangeburg, SC); Cindy Setum, PhD (Minneapolis, MN) for study management, and Richard Holcomb, PhD (Minneapolis, MN) for performing the statistical analyses.
Declaration of Conflicting Interest
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: A. Kramer has received honoraria from DaVita, Inc.; Medtronic and/or Mozarc Medical; Merit Medical Systems; Sonavex, Inc.; VenoStent, Inc.; VentureMed Group. K.M.L.S.T. Moorthi has no industry relationships to disclose. L. Grant has served as a consultant to Phraxis, Inc. T. Litchfield has served as a consultant to Mozarc Medical; Sonavex, Inc.; VenoStent, Inc.; and VentureMed Group.
Funding Statement
Funding to support this study was provided by Phraxis, Inc.
References
- Lok CE, Huber TS, Lee T et al. National Kidney Foundation. KDOQI Clinical practice guideline for vascular access 2019 update. Am J Kidney Dis. 75, S1–S16 (2020).
- Yevzlin AS, Chan MR, Becker YT et al. “Venopathy” at work: recasting neointimal hyperplasia in a new light. Transl Res. 156, 216–225 (2010).
- Lee T, Roy-Chaudhury P. Advances and new frontiers in the pathophysiology of venous neointimal hyperplasia and dialysis access stenosis. Adv Chronic Kidney Dis. 16, 329–338 (2009).
- Burgess JS, Beaver JD, London M et al. InterGraft Study Investigators. Prospective multicenter study of a novel endovascular venous anastomotic procedure and device for implantation of an arteriovenous graft for hemodialysis. J Vasc Access. 25, 1244–1251 (2024).
- Halbert RJ, Nicholson G, Nordyke RJ et al. Patency of ePTFE arteriovenous graft placements in hemodialysis patients: systematic literature review and meta-analysis. Kidney360. 1, 1437–1446 (2020).
- Lynch JR, Wasse H, Armistead NC et al. Achieving the goal of the Fistula First breakthrough initiative for prevalent maintenance hemodialysis patients. Am J Kidney Dis. 57, 78–89 (2011).
- Vascular Access 2006 Work Group. Clinical practice guidelines for vascular access. Am J Kidney Dis. 48, S176–S273 (2006).
- S. Renal Data System. USRDS 2014 Annual Data Report: Atlas of End-Stage Renal Disease in the United States. National Institutes of Health, National Institute of Diabetes and Digestive and Kidney Diseases, Bethesda, MD (2014).
- Sabiu G, Gallieni M. Pathophysiology of arteriovenous fistula maturation and nonmaturation. Clin J Am Soc Nephrol. 18, 8–10 (2023).
- Allon M, Robbin ML. Increasing arteriovenous fistulas in hemodialysis patients: problems and solutions. Kidney Int. 62, 1109–1124 (2002).
- Richardson AI, Leake A, Schmieder GC et al. Should fistulas really be first in the elderly patient? J Vasc Access. 10, 199–202 (2009).
- Peterson WJ, Barker J, Allon M. Disparities in fistula maturation persist despite preoperative vascular mapping. Clin J Am Soc Nephrol. 3, 437–441 (2008).
- Miller CD, Robbin ML, Allon M. Gender differences in outcomes of arteriovenous fistulas in hemodialysis patients. Kidney Int. 63, 346–352 (2003).
- Li L, Terry CM, Shiu YT et al. Neointimal hyperplasia associated with synthetic hemodialysis grafts. Kidney Int. 74, 1247–1261 (2008).
- Beathard GA. Percutaneous transvenous angioplasty in the treatment of vascular access stenosis. Kidney Int. 42, 1390–1397 (1992).
- Harms J, Rangarajan S, Young C et al. Outcomes of arteriovenous fistulas and grafts with or without intervention prior to successful use. J Vasc Surg. 64, 155–162 (2016).
- Woodside K, Repeck K, Mukhopadhyay P et al. Arteriovenous vascular access–related procedural burden among incident hemodialysis patients in the United States. Am J Kidney Dis. 78, 369–379 (2021).
- Nordyke RJ, Nicholson G, Gage SM et al. Vascular access-specific health-related quality of life impacts among hemodialysis patients: qualitative development of the hemodialysis access-related quality of life (HARQ) instrument. BMC Nephrol. 21, 16 (2020).
- Sonawane P, Maheshwari R, Singh A et al. Impact of vascular access type on health-related quality of life in patients undergoing hemodialysis: a cross-sectional observational study. Indian J Vasc Endovasc Surg. 7, 63–66 (2020).
- Kim DH, Park JI, Lee JP et al. The effects of vascular access types on the survival and quality of life and depression in the incident hemodialysis patients. Ren Fail. 42, 30–39 (2020).
- Quinn RR, Lamping DL, Lok CE et al. The Vascular Access Questionnaire: assessing patient-reported views of vascular access. J Vasc Access. 9, 122–128 (2008).