Research Article - Journal of Interventional Nephrology (2026) Volume 9, Issue 1
Endovascular AVG venous anastomosis improves cumulative patency compared to traditional sutured anastomosis: a meta-analysis
Brad C. Astor1, KMLST Moorthi2, Lisa Grant3* and Terry Litchfield4
1Departments of Medicine and Population Health Sciences, University of Wisconsin School of Medicine and Public Health
2Northwestern University Feinberg School of Medicine
3LB Grant Clinical Research Consulting
- *Corresponding Author:
- Lisa Grant
2425 France Ave S., St. Louis Park, MN 55416
Tel: 612-308-0958
E-mail: lgrant@phraxis.com
Received: 02-Feb-2026, Manuscript No: oain-26-185112, Editor Assigned: 04- Feb-2026, pre QC No: oain-26-185112 (PQ), Reviewed: 16-Feb-2026, QC No oain-26-185112, Revised: 20-Feb-2026, Manuscript No: oain-26-185112 (R), Published: 27-Feb-2026, DOI: 10.47532/oain.2026.9(1).388
Abstract
Objective: Arteriovenous graft (AVG) patency outcomes continue to be plagued by venous anastomotic stenosis, which can lead to eventual thrombosis and AVG abandonment. Venous stenosis occurs most commonly at the graft-vein and juxta-anastomotic vein segments. A recent publication of a novel endovascular AVG anastomosis (EndoForce) showed excellent outcomes with a cumulative patency of 92% for AVGs. The purpose of this investigation was to compare the outcomes of the EndoForce endovascular venous anastomotic connector to traditional sutured anastomosis AVGs via meta-analysis.
Methods: A meta-analysis was conducted to estimate the 6-month patency of AVGs implanted using traditional surgical techniques. We compared those estimates to the results achieved with the EndoForce venous anastomotic device.
Results: A total of 31 studies, with 45 distinct arms, provided data on 6-month cumulative patency. The random-effects pooled estimate of 6-month cumulative patency was 83.23% (95% confidence interval [CI]: 80.18, 86.28). A total of 32 studies, including 46 arms, provided data on primary patency. The pooled estimate for 6-month primary patency was 60.35% (95% CI: 56.21, 64.49). The novel device achieved better 6-month cumulative patency (92.08%; 95% CI: 86.98-97.18) than the pooled estimate (p<0.001) and similar primary patency (60.21% [95% CI: 50.84, 69.59]; p=0.75).
Conclusion: This study provides direct evidence that the EndoForce venous anastomotic device improves 6-month cumulative patency compared to traditional sutured AVG venous anastomosis.
Keywords
Endovascular procedures; Vascular grafting; Arteriovenous anastomosis; Graft survival; Renal dialysis; Patency
Introduction
Arteriovenous grafts (AVGs) are an essential vascular access option for end stage kidney disease (ESKD) patients requiring hemodialysis. The 2019 Kidney Disease Outcomes Quality Initiative (KDOQI) guidelines recommend that the choice of AV-access (arteriovenous fistula (AVF) or AVG) be based on the clinician’s best clinical judgment that considers the vessel characteristics, patient comorbidities, health circumstances, and patient preference [1,2].
The 2019 guidelines further state that there is inadequate evidence for KDOQI to make a recommendation on choice of AVF versus AVG for incident vascular access creation. Despite these guidelines, AVGs continue to be used sparingly in the ESKD “life-plan” ; in 2024 only 3.9% of incident and 16.4% of prevalent patients were using an AVG 9 [3]. This relative underutilization of AVGs is due in part to the fact that traditional sutured AVG patency outcomes continue to be plagued by venous anastomotic stenosis, which can lead to eventual thrombosis and AVG abandonment. Venous stenosis occurs most commonly at the graft-vein and juxta-anastomotic vein segments [4].
A recent publication of a novel endovascular AVG anastomosis ((EndoForce; Phraxis, Inc., Minneapolis, MN) showed excellent outcomes with a cumulative patency of 92% for AVGs compared to a historical control of 75% for traditional sutured venous anastomosis AVGs [5]. The aforementioned pivotal clinical trial was limited by the absence of a direct comparison group within the study. The purpose of this investigation was to compare the outcomes of the EndoForce endovascular venous anastomotic connector to traditional sutured anastomosis AVGs via meta-analysis of more recent studies.
Methods
Study design
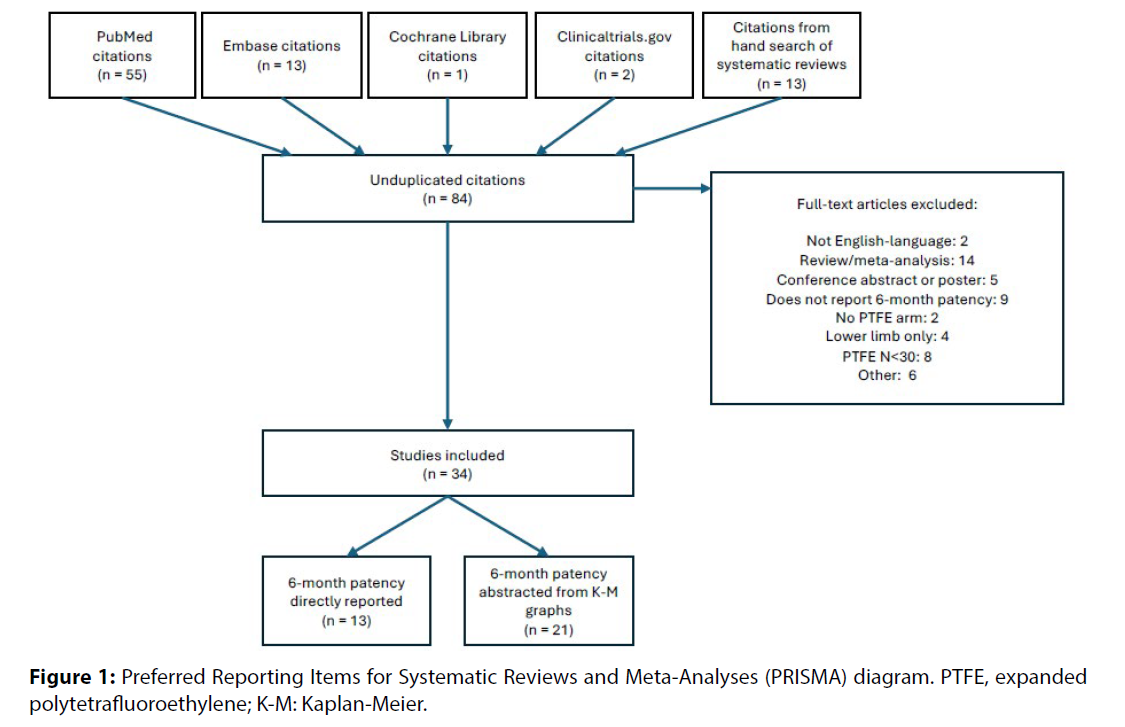
This meta-analysis was conducted in accordance with Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidance [6]. Search terms, inclusion and exclusion criteria, and analytic methods were specified in advance.
The study population consisted of patients who were at least 18 years old who were preparing for or currently receiving chronic hemodialysis treatment using an expanded polytetrafluoroethylene (ePTFE) graft in the upper extremities. Inclusion criteria for studies to be included were: at least one study arm included use of a new ePTFE graft; and the study was published in English. Exclusion criteria were: study arms had fewer than 30 ePTFE grafts; study included only lower-extremity or chest grafts; and study did not report patency measures at 6 months and did not provide Kaplan-Meier graphs from which to abstract these data.
Literature search
Literature searches were conducted in October 2025 in the PubMed, Embase, Cochrane Library, and Clinicaltrials.gov databases. The PRISMA diagram is shown in Figure 1. Additional studies were identified by a hand search of references in selected papers.
Studies were screened for eligibility by two reviewers. Extracted data included study design, graft characteristics (device type/brand and location), patient characteristics (demographics and comorbidities), and 6-month patency outcomes.
End points and definitions
We used the definitions of patency as published by the Journal of Vascular Surgery [7]. Primary patency, or primary unassisted patency, was defined as “the interval from graft placement to the first intervention to relieve stenosis, with or without occlusion.” Secondary patency, or cumulative patency, was defined as the “interval from graft placement to graft abandonment.”
Statistical analyses
We used a random effects model to conduct the meta-analyses in R 4.5.0 [8] using the metafor [9] and forestplot (v3.1.7) [10] packages. Primary outcomes were primary and cumulative patency at 6 months. Heterogeneity between studies was assessed using the I2 statistic.
For studies in which the 6-month patency outcomes were reported in tables or text, the reported numbers were used in the analysis. DigitizeIt software was used to abstract the 6-month patency measurements from Kaplan-Meier survival curves from those studies which did not specifically report 6-month patency outcomes but included survival curves.
Results
Studies included
A total of 34 studies were included in the analyses (Table 1). A total of 32 studies, including 46 arms with results listed separately, provided data on primary patency. A total of 31 studies, with 45 distinct arms, provided data on cumulative patency.
| Study | Country | Years | N | Primary Patency* | Cumulative Patency* |
|---|---|---|---|---|---|
| Arhuidese (2017) [19] | US | 2011-2014 | 68 | R | R |
| Baba (2022) – PROPATEN [20] | US | 2015-2019 | 36 | R | R |
| Baba (2022) – ACUSEAL [20] | US | 2015-2019 | 24 | R | R |
| Benedetto (2019) [21] | Italy | 2013-2018 | 145 | R | R |
| Benedetto (2019) [21] | Italy | 2013-2018 | 218 | R | R |
| Chang (2021) [22] | Singapore | 2017-2018 | 48 | R | R |
| Desai (2019) [23] | US | 2014-2018 | 266 | R | |
| Honma & Eguchi (2022) [24] | Japan | 2015-2017 | 113 | R | |
| Kim (2023) <65 [25] | Korea | 2008-2019 | 8,335 | R | R |
| Kim (2023) ≥65 [25] | Korea | 2008-2019 | 13,522 | R | R |
| Kong (2025) [26] | US | 2015-2022 | 40 | R | R |
| Lu (2024) [27] | US | 2010-2022 | 157 | R | R |
| Luo (2025) - period I [28] | China | 2014-2022 | 110 | R | R |
| Luo (2025) - period II [28] | China | 2014-2023 | 379 | R | R |
| Luo (2025) - period III [28] | China | 2014-2024 | 491 | R | R |
| Nissen (2020) - HB-PTFE [29] | US | 2012-2018 | 50 | R | R |
| Nissen (2020) - S-PTFE [29] | US | 2012-2018 | 47 | R | R |
| Tawfik (2022) – standard [30] | Egypt | 2017-2020 | 236 | R | R |
| Tawfik (2022) – early [30] | Egypt | 2017-2020 | 241 | R | R |
| Zhao (2024) – GPVG [31] | China | 2016-2019 | 105 | R | R |
| Zhao (2024) – GAVG [31] | China | 2016-2019 | 109 | R | R |
| Zhao (2024) – BVVG [31] | China | 2016-2019 | 68 | R | R |
| Arhuidese (2019) – combined [32] | US | 2007-2011 | 15,552 | A | A |
| Arhuidese (2018) [33] | US | 2007-2011 | 49,423 | A | A |
| Aziz (2023) [34] | US | 2013-2016 | 48 | A | A |
| Benedetto (2017) – GHVG [35] | Italy | 2010-2013 | 32 | A | A |
| Benedetto (2017) – standard [36] | Italy | 2010-2013 | 43 | A | A |
| Choi (2020) [36] | Korea | 2009-2017 | 416 | A | A |
| Davies (2016) - HB-PTFE [37] | US | 2004-2024 | 234 | A | A |
| Davies (2016) - S-PTFE [37] | US | 2004-2024 | 248 | A | A |
| Drouven (2019) [38] | Netherlands | 2006-2017 | 75 | A | A |
| Fumagalli (2019) [39] | Italy | 2010-2015 | 105 | A | A |
| Habibollahi (2019) [40] | US | 2013-2016 | 46 | A | A |
| Kakisis (2017) [41] | Greece | 2007-2015 | 61 | A | A |
| Khanfar (2023) [42] | Palestine | 2017-2021 | 72 | A | A |
| Kim (2022) [43] | Korea | 2015-2019 | 53 | A | A |
| Lin (2021) [44] | Taiwan | 2008-2012 | 6493 | A | A |
| Marcus (2019) [45] | US | 2010-2017 | 128 | A | A |
| Miao (2022) [46] | China | 2013-2017 | 179 | A | A |
| Mo (2023) [47] | Korea | 2008-2019 | 21,857 | A | A |
| Oh (2024) – elbow [48] | Korea | 2018-2022 | 36 | A | |
| Shemesh (2015) – HB [49] | Israel | 2007-2011 | 80 | A | A |
| Shemesh (2015) – standard [49] | Israel | 2007-2011 | 80 | A | A |
| Sutaria (2020) – elective [50] | UK | 2012-2017 | 66 | A | A |
| Sutaria (2020) – emergency [50] | UK | 2012-2018 | 75 | A | A |
| Zea (2016) – HB [51] | US | 2013-2014 | 32 | A | A |
| Zea (2016) – standard [51] | US | 2013-2014 | 38 | A | A |
Table 1: References included in the meta-analysis.
Patency result
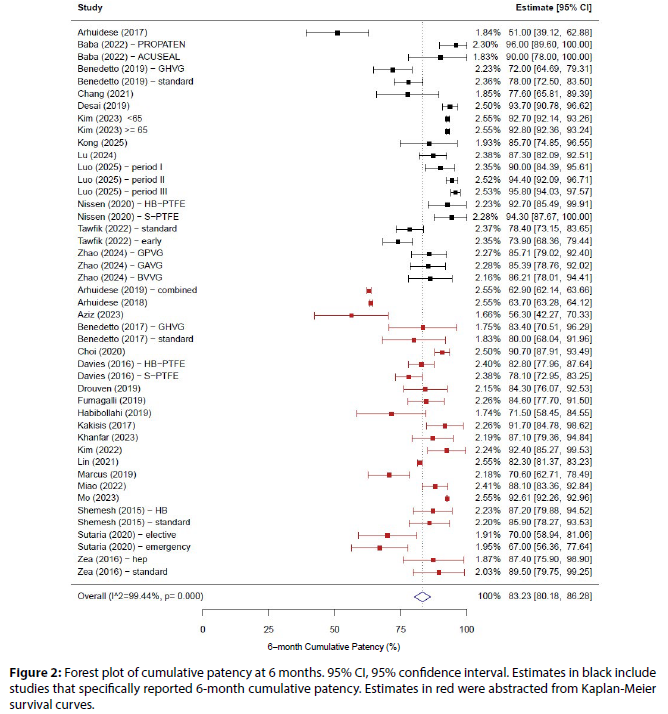
The random-effects pooled estimate of 6-month cumulative patency was 83.23% (95% confidence interval [CI]: 80.18, 86.28), with wide variability (I2=99.36). (Figure 2) The estimated cumulative patency limited to studies which specifically reported values (12 studies, 21 arms) was 86.00% (95% CI: 81.78, 90.22).
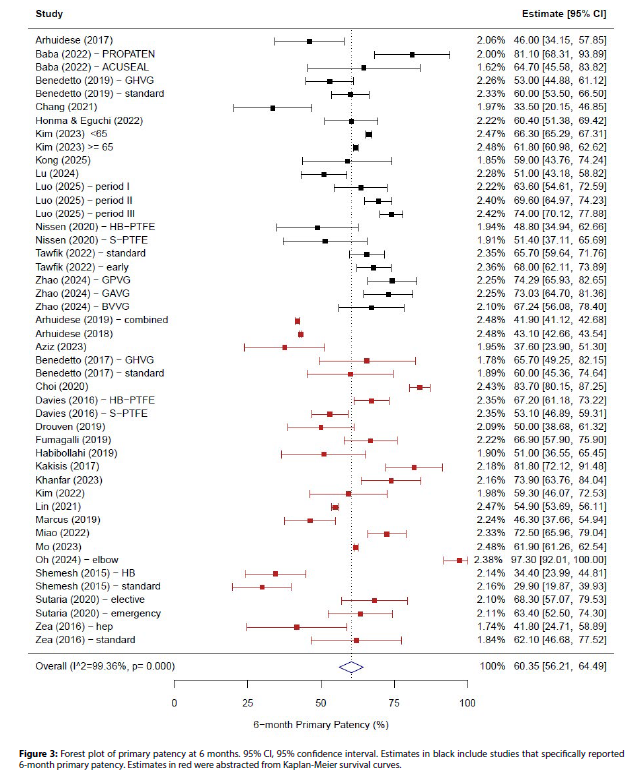
The pooled estimate for 6-month primary patency from the meta-analysis was 60.35% (95% CI: 56.21, 64.49), again with substantial variability (I2=99.44). The estimate was similar when including only those studies (12 studies, 21 arms) which specifically reported 6-month values (62.23% [95% CI: 57.80, 66.66]).
Comparison to novel device
The novel device achieved better 6-month cumulative patency (92.08%; 95% CI: 86.98-97.18) than the pooled estimate (p<0.001). The novel device achieved a 6-month primary patency of 60.21% (95% CI: 50.84, 69.59). This estimate did not differ significantly (p=0.75) from that achieved with the pooled estimate (60.35% [95% CI: 56.21-64.49]). Results were similar when limited to those with 6-month results specifically reported.
Discussion
Despite recent guidelines that were intended to increase the use of AVGs, AVGs continue to be underutilized as a form of permanent access in both the incident and prevalent populations. The United States Renal Data System (USRDS) reports show a concerning pattern of AVF, central venous catheter (CVC), and AVG use post COVID: In 2018, AVF 64.4%, AVG 17.0%, and CVC 18.6% were used as the primary access in prevalent ESKD hemodialysis patients. In 2024, AVF use had decreased to 57.4%, while AVG decreased to 16.4%, and CVC increased to 26.2% [11]. This represents a 23.7% relative increase in the use of CVCs in the prevalent US hemodialysis population. This is a concerning trend, as a recent meta-analysis showed that CVCs were associated with a 248% increase in all-cause mortality relative to AVFs (OR = 2.48, 95% CI: 1.37–4.48, p < 0.0001) [12].
Traditional AVG 6-month cumulative patency was shown to be approximately 80% in a recent meta-analysis [13]. The pivotal study results of the EndoForce device compare favorably to this estimate, with the EndoForce conferring a 6-month primary patency of 60% and a 6-month cumulative patency of 92%. The cumulative patency of the EndoForce compared to data from the meta-analysis represents a reduction of AVG abandonment at 6 months from 20% to 8%, or a relative 60% reduction in AVG abandonment. However, these results were not compared directly in previous publications. The meta-analysis reported herein compares the 6-month cumulative patency of AVGs implanted using the EndoForce venous anastomotic device with cumulative patency of AVG implanted using the traditional surgical technique directly. We found that the novel EndoForce device achieved better 6-month cumulative patency (92.08% [95% CI: 86.98-97.18] than the random-effects pooled estimate of cumulative 6-month patency of standard AVGs 83.23% (95% confidence interval [CI]: 80.18-86.28) (p=0.002).
This difference could potentially be due to the less invasive, endovascular nature of the procedure that is used to place the EndoForce device as compared to traditional sutured AVG creation. Since AVG abandonment leads to prolonged catheter use and the attendant morbidity and mortality, our study results may represent a possible improvement of an outcomes variable that is relevant to ESKD patients and their families [14]. In addition, the relative improvement in outcomes seen in this study may be due to the hemodynamic benefits of an end-to-end vs. the end-to-side anastomosis. Sheer stress due to unfavorable hemodynamics is a known factor in the development of AVG venous anastomotic stenosis [15, 16]. Further, the anastomotic angle is known to have a direct impact on the flow dynamics, demonstrating a turbulence zone [17].
A previous device, the Gore Hybrid Vascular Graft (GHVG) can also be noted to have an absence of a sewn venous anastomosis, which is replaced by a 5- or 10-cm nitinol covered PTFE stent-graft that is pre-attached to an AVG [18]. It is important to note that the EndoForce study device is not an off-the-shelf, repurposed stent. Rather, it is a patented implant specifically designed for coaxial, endovascular anastomosis of a vein to an AVG. The EndoForce has three elements that distinguish it from other nitinol/ePTFE devices, such as stentgrafts: (1) Barbs on the end secure the endovascular anastomosis to the vein; (2) A flexible nitinol segment provides a wide range of angles from the venotomy site that the AVG can engage the EndoForce; and (3) A rigid segment of the EndoForce designed to provide maximum radial force to maximize a friction fit between the AVG and EndoForce overlap. The self-expanding EndoForce is placed within a peripheral vein using a unique endovascular, transcatheter, over-the-wire technique. Unlike the GHVG, the EndoForce is not pre-attached to a specific AVG. Instead, it is designed for use with any commercially available 6 mm inner diameter ePTFE graft for hemodialysis. Nevertheless, it is important to note that the functional cumulative patency rate of the GHVG did not differ substantially from a standard sutured anastomosis at 6 months (approximately 80% patency).
Conclusion
This study provides direct evidence that the EndoForce venous anastomotic device improves 6-month cumulative patency compared to a traditional sutured AVG venous anastomosis.
References
- Vascular Access 2006 Work Group. Clinical practice guidelines for vascular access. Am J Kidney Dis 48, S176âS247.
[Indexed at], [Google Scholar], [Crossref]
- Lok CE, Huber TS, Lee T, et al. KDOQI Clinical practice guideline for vascular access 2019 update. Am J Kidney Dis 75, S1âS164 (2020).
[Indexed at], [Google Scholar], [Crossref]
- United States Renal Data System. 2025 USRDS Annual Data Report: Epidemiology of kidney disease in the United States. National Institutes of Health, National Institute of Diabetes and Digestive and Kidney Diseases, Bethesda, MD.
[Indexed at], [Google Scholar], [Crossref]
- Yevzlin AS, Chan MR, Becker YT, et al. âVenopathyâ at work: recasting neointimal hyperplasia in a new light. Transl Res 156: 216â225 (2010).
[Indexed at], [Google Scholar], [Crossref]
- Burgess JS, Beaver JD, London M, et al. Prospective multicenter study of a novel endovascular venous anastomotic procedure and device for implantation of an arteriovenous graft for hemodialysis. J Vasc Access 25, 1244-1251(2023).
[Indexed at], [Google Scholar], [Crossref]
- Moher D, Liberati A, Tetzlaff J, et al. PRISMA Group: Preferred reporting items for systematic reviews and meta-analyses: The PRISMA statement. J Clin Epidemiol 62, 1006â1012 (2009).
[Indexed at], [Google Scholar], [Crossref]
- Sidawy AN, Gray R, Besarab A, et al. Recommended standards for reports dealing with arteriovenous hemodialysis accesses. J Vasc Surg 35, 603â610 (2002).
[Indexed at], [Google Scholar], [Crossref]
- R Core Team. R: A Language and Environment for Statistical Computing. R Foundation for Statistical Computing, Vienna, Austria (2025).
[Indexed at], [Google Scholar], [Crossref]
- Viechtbauer W. Conducting meta-analyses in R with the metafor package. J Stat Softw. 36, 1â48 (2010).
[Indexed at], [Google Scholar], [Crossref]
- Gordon M, Lumley T. forestplot: Advanced Forest Plot Using 'grid' Graphics_ .package.forestplot. R package version 3.1.7 (2025).
- United States Renal Data System. 2025 USRDS Annual Data Report: Epidemiology of kidney disease in the United States. National Institutes of Health, National Institute of Diabetes and Digestive and Kidney Diseases, Bethesda, MD.
- Miao P, Tan Z, Yao C, et al. Mortality outcomes associated with vascular access types in hemodialysis for ESRD: a systematic review and meta-analysis. BMC Nephrol 27, 67 (2026).
[Indexed at], [Google Scholar], [Crossref]
- Halbert RJ, Nicholson G, Nordyke RJ, et al. Patency of ePTFE arteriovenous graft placements in hemodialysis patients- systematic literature review and meta-analysis. Kidney 360. 1, 1437â1446 (2020).
[Indexed at], [Google Scholar], [Crossref]
- Gee PO. Kidney health initiative roadmap for kidney replacement therapy: A patient's perspective. Clin J Am Soc Nephrol 15, 585â586 (2020).
[Indexed at], [Google Scholar], [Crossref]
- Fillinger MF, Reinitz ER, Schwartz RA, et al. Beneficial effects of banding on venous intimal-medial hyperplasia in arteriovenous loop grafts. Am J Surg 158, 87â94 (1989).
[Indexed at], [Google Scholar], [Crossref]
- Sivanesan S, How TV, Black RA, et al. Flow patterns in the radiocephalic arteriovenous fistula: an in vitro study. J Biomech 32, 915â925 (1999).
[Indexed at], [Google Scholar], [Crossref]
- Staalsen NH, Ulrich M, Winther J, et al. The anastomosis angle does change the flow fields at vascular end-to-side anastomoses in vivo. J Vasc Surg 21, 460â471 (1995).
[Indexed at], [Google Scholar], [Crossref]
- Benedetto F, Spinelli D, Pipitò N, et al. Initial clinical experience with a polytetrafluoroethylene vascular dialysis graft reinforced with nitinol at the venous end. J Vasc Surg 65, 142â150 (2017).
[Indexed at], [Google Scholar], [Crossref]
- Arhuidese I, Reifsnyder T, Islam T, et al. Bovine carotid artery biologic graft outperforms expanded polytetrafluoroethylene for hemodialysis access. J Vasc Surg 65, 775â782 (2017).
[Indexed at], [Google Scholar], [Crossref]
- Baba T, Ohki T, Maeda K, et al. Evaluation of heparin-bonded ePTFE grafts for forearm loop vascular access: Comparison between Gore PROPATEN vascular graft and ACUSEAL vascular graft. J Vasc Access 23, 430â435 (2022).
[Indexed at], [Google Scholar], [Crossref]
- Benedetto F, Spinelli D, Pipitò N, et al. Hybrid arteriovenous graft for hemodialysis vascular access in a multicenter registry. J Vasc Surg 70, 1904â1912.e2 (2019).
[Indexed at], [Google Scholar], [Crossref]
- Chang J, Yap HY, Chan SL, et al. Single Asian Center Experience Using the FlixeneTM Early Cannulation Graft for Hemodialysis Access Creation. Ann Vasc Surg 73, 171â177 (2021).
[Indexed at], [Google Scholar], [Crossref]
- Desai SS. Two-Year Outcomes of Early Cannulation Arteriovenous Grafts for End-Stage Renal Disease. Ann Vasc Surg 59, 158â166 (2019).
[Indexed at], [Google Scholar], [Crossref]
- Honma K and Eguchi D. Short- to midterm results of early cannulation arteriovenous grafts (Gore ACUSEAL) for hemodialysis: Experience with the ACUSEAL in a Japanese cohort. J Vasc Access 23, 75â80 (2022).
[Indexed at], [Google Scholar], [Crossref]
- Kim H, Park HS, Ban TH, et al. Evaluation of outcomes with permanent vascular access in an elderly Korean population based on the National Health Insurance Service database. Hemodial Int 27, 249â258 (2023).
[Indexed at], [Google Scholar], [Crossref]
- Kong D, Lim F, Jabbour G, et al. Comparative study of modified HeRO grafts using bovine carotid artery as conduits versus polytetrafluoreythylene. J Vasc Access Jun18:11297298251343332 (2025).
[Indexed at], [Google Scholar], [Crossref]
- Lu E, Baril DT, Arbabi CN, et al. Outcomes of prosthetic upper arm arteriovenous graft vs brachiobasilic fistula for hemodialysis access. J Vasc Surg 80, 1083â1089.e2 (2024).
[Indexed at], [Google Scholar], [Crossref]
- Luo Y, Bian X, Gu C, et al. Impact of evolving strategies for arteriovenous graft creation and management on patency outcomes. Ren Fail 47, 2549776 (2025).
[Indexed at], [Google Scholar], [Crossref]
- Nissen AP, Sandhu HK, Perlick AP, et al. Heparin-bonded versus standard polytetrafluoroethylene arteriovenous grafts: A Bayesian perspective on a randomized controlled trial for comparative effectiveness. Surgery 168, 1066â1074 (2020).
[Indexed at], [Google Scholar], [Crossref]
- Tawfik AM, Zidan MH, Salem A, et al. A randomized controlled study of early versus standard cannulation of arteriovenous grafts in hemodialysis patients. J Vasc Surg 75, 1047â1053 (2022).
[Indexed at], [Google Scholar], [Crossref]
- Zhao T, Wang W, Lui KHW, et al. Retrospective evaluation of three types of expanded polytetrafluoroethylene grafts for upper limb vascular access. Ren Fail 46, 2371056 (2024).
[Indexed at], [Google Scholar], [Crossref]
- Arhuidese IJ, Cooper MA, Rizwan M, et al. Vascular access for hemodialysis in the elderly. J Vasc Surg 69 517â525.e1 (2019).
[Indexed at], [Google Scholar], [Crossref]
- Arhuidese IJ, Orandi BJ, Nejim B, et al. Utilization, patency, and complications associated with vascular access for hemodialysis in the United States. J Vasc Surg. 68, 1166â1174 (2018).
[Indexed at], [Google Scholar], [Crossref]
- Aziz M, Albert P, Connelly ZM, et al. A Retrospective Review of Bovine Artery Graft Patency: A Single-Site Study. Ann Vasc Surg 94, 272â279 (2023).
[Indexed at], [Google Scholar], [Crossref]
- Benedetto F, Spinelli D, Pipitò N, et al. Initial clinical experience with a polytetrafluoroethylene vascular dialysis graft reinforced with nitinol at the venous end. J Vasc Surg 65, 142â150 (2017).
[Indexed at], [Google Scholar], [Crossref]
- Choi J, Ban TH, Choi BS. et al. Comparison of vascular access patency and patient survival between native arteriovenous fistula and synthetic arteriovenous graft according to age group. Hemodial Int 24, 309â316 (2020).
[Indexed at], [Google Scholar], [Crossref]
- Davies MG, Anaya-Ayala JE, El-Sayed HF. Equivalent outcomes with standard and heparin-bonded expanded polytetrafluoroethylene grafts used as conduits for hemodialysis access. J Vasc Surg 64 715â718 (2016).
[Indexed at], [Google Scholar], [Crossref]
- Drouven JW, de Bruin C, van Roon AM, et al. Outcomes of basilic vein transposition versus polytetrafluoroethylene forearm loop graft as tertiary vascular access. J Vasc Surg 69, 1180â1186 (2019).
[Indexed at], [Google Scholar], [Crossref]
- Fumagalli G, Trovato F, Migliori M, et al. The forearm arteriovenous graft between the brachial artery and the brachial vein as a reliable dialysis vascular access for patients with inadequate superficial veins. J Vasc Surg 70, 199â207.e4 (2019).
[Indexed at], [Google Scholar], [Crossref]
- Habibollahi P, Mantell MP, Rosenberry T, et al. Outcomes of a Polytetrafluoroethylene Hybrid Vascular Graft with Preloaded Nitinol Stent at the Venous Outflow for Dialysis Vascular Access. Ann Vasc Surg 55, 210â215 (2019).
[Indexed at], [Google Scholar], [Crossref]
- Kakisis JD, Antonopoulos C, Mantas G, et al. Safety and efficacy of polyurethane vascular grafts for early hemodialysis access. J Vasc Surg 66, 1792â1797 (2017).
[Google Scholar], [Crossref]
- Khanfar O, Aydi R, Saada S, et al. Mid-term cumulative patency of fistula and PTFE grafts among hemodialysis patients: A retrospective, single-center study from Palestine. J Vasc Access 24, 559â567 (2023).
[Indexed at], [Google Scholar], [Crossref]
- Kim H, Ahn S, Kim M, et al. Comparison between autogenous brachial-brachial upper-arm elevated direct arteriovenous fistulas and prosthetic brachial-antecubital indirect forearm arteriovenous grafts. J Vasc Access 23, 936â945(2022).
[Indexed at], [Google Scholar], [Crossref]
- Lin SJ, Chang SC, Tung CW, et al. Timing of the first cannulation and survival of arteriovenous grafts in hemodialysis patients: a multicenter retrospective cohort study. Ren Fail 43, 1416â1424 (2021).
[Indexed at], [Google Scholar], [Crossref]
- Marcus P, Echeverria A, Cheung M, et al. Early Cannulation of Bovine Carotid Artery Graft Reduces Tunneled Dialysis Catheter-Related Complications: A Comparison of Bovine Carotid Artery Graft Versus Expanded Polytetrafluoroethylene Grafts in Hemodialysis Access. Vasc Endovasc Surg 53, 104â111 (2019).
[Indexed at], [Google Scholar], [Crossref]
- Miao P, Tan ZL, Tian R, et al. Long-term patency and comparisons of venous outflow in hemodialysis forearm arteriovenous grafts. Vascular 30, 238â245 (2022).
[Indexed at], [Google Scholar], [Crossref]
- Mo H, Kwon S, Kim D, et al. Patency of arteriovenous fistulas and grafts for dialysis access: An analysis using the Korean National Health Insurance Service database from 2008 to 2019. J Vasc Access 25, 1544â1552 (2024).
[Indexed at], [Google Scholar], [Crossref]
- Gifford SM, Shepherd MF, Wagner-Drouet EM. Early results of the Gore Acuseal graft for vascular access. Ann Vasc Surg 21, 451â456 (2007).
[Indexed at], [Google Scholar], [Crossref]
- Schild AF, Schuman ES, Noicely K, et al. Early cannulation graft (Flixeneâ¢) for arteriovenous access. J Vasc Surg 48, 1127â1131 (2008).
[Indexed at], [Google Scholar], [Crossref]
- Tordoir JHM, Mickley V. Guidelines for the prevention and management of vascular access complications. Eur J Vasc Endovasc Surg 26, 563â578 (2003).
[Indexed at], [Google Scholar], [Crossref]
- Glickman MH. HeRO Graft: The past, present and future. J Vasc Access 17, S37âS40 (2016).
[Indexed at], [Google Scholar], [Crossref]