Mini Review - Interventional Cardiology (2025) Volume 17, Issue 4
Clinical significance of left atrial appendage edema and dense spontaneous echo contrast after pulsed field ablation with planned concomitant device closure
- Corresponding Author:
- Muhammad Munawar
Binawaluya Cardiac Centre, Jakarta, Indonesia
E-mail: muna286@gmail.com
Received date: 10-Nov-2025, Manuscript No. FMIC-25-173500; Editor assigned: 12-Nov-2025, PreQC No. FMIC-25-173500 (PQ); Reviewed date: 26-Nov-2025, QC No. FMIC-25-173500; Revised date: 03-Dec-2025, Manuscript No. FMIC-25-173500 (R); Published date: 10-Dec-2025, DOI: 10.37532/1755-5310.2025.17(4).1030
Abstract
Pulsed Field Ablation (PFA) is an emerging non-thermal ablation modality that achieves selective myocardial injury while preserving adjacent non-cardiac structures. Its rapid energy delivery and favourable safety profile make it a promising candidate for integration with Left Atrial Appendage Closure (LAAC) in a single procedural session. This combined approach offers a comprehensive strategy addressing both rhythm control and stroke prevention in atrial fibrillation management. However, Transient Left Atrial Appendage (LAA) edema and Spontaneous Echo Contrast (SEC) may occur after PFA, leading to temporary alterations in LAA geometry, flow velocity, and imaging interpretation. Consequently, the feasibility and safety of concomitant pulmonary vein isolation and LAAC using PFA remain under active investigation. Although these tissue changes are typically self-limiting, they underscore the importance of meticulous procedural anticoagulation and careful imaging evaluation. Further studies are warranted to clarify their temporal evolution, clinical relevance, and implications for optimizing combined PFA-LAAC strategies.
Keywords
Pulsed field ablation; Left atrial appendage edema; Dense spontaneous echo contrast
Introduction
Pulsed Field Ablation (PFA) has emerged as an innovative energy source in cardiac electrophysiology and represents a major advancement in atrial arrhythmia management [1]. Unlike conventional thermal ablation techniques, PFA employs non-thermal irreversible electroporation to achieve selective myocardial ablation while preserving adjacent non-cardiac structures. This unique mechanism enables faster lesion creation, shorter procedure times, and a lower risk of collateral injury [2, 3]. Several clinical studies have demonstrated that PFA can effectively isolate pulmonary veins with high procedural success and a reduced recurrence rate of Atrial Fibrillation (AF) [4]. Despite these promising outcomes, current evidence regarding the spectrum and incidence of PFA-related complications remains limited, given the technology’s recent introduction into clinical practice.
The shorter procedural time and superior efficacy of PFA compared to conventional ablation offer potential advantage for performing PFA and Left Atrial Appendage Closure (LAAC) simultaneously. The dual simultaneous approach may reduce arrhythmia burden and stroke risk while minimizing procedural complications associated with separate procedures [5, 6]. However, the feasibility of this combined approach remains under investigation due to the potential impact on the Left Atrial Appendage (LAA). LAA edema and dense Spontaneous Echo Contrast (SEC) as reported from recent case raises concerns regarding device under-sizing, peridevice leak, device-related embolization and even thromboembolic complications [7]. These observations underscore the importance of careful evaluation when integrating PFA with LAAC in atrial fibrillation management.
Literature Review
Concomitant Pulsed Field Ablation and LAA Closure
The combination of PFA for Pulmonary Vein Isolation (PVI) and LAA closure represents an emerging strategy for comprehensive management of AF. Given the substantial risk of thromboembolic recurrence and the limited evidence supporting stroke prevention after ablation, current clinical guidelines recommend maintaining Oral Anticoagulation (OAC) indefinitely in AF patients who remain at elevated stroke risk [8]. This recommendation applies irrespective of post-ablation rhythm status and is guided by an individualized evaluation of thromboembolic versus bleeding risk. Consequently, catheter ablation alone cannot be considered a definitive stroke-preventive strategy, prompting consideration of whether combining the procedure with LAAC could offer additional protection.
By targeting both rhythm control and stroke prevention in a single session, this approach reduces procedural burden, anesthesia exposure, and cumulative risk. This approach also shortens hospitalization duration, minimizes imaging-related risks, and allows for a single vascular access and transseptal puncture, thereby potentially reducing procedural complications. The non-thermal nature of PFA, which selectively ablates atrial myocardium while sparing surrounding structures, makes it particularly suitable for integration with LAA device implantation, as it minimizes tissue charring and pericardial inflammation that could complicate device deployment [9]. However, combining these two interventions introduces distinct procedural and imaging challenges. Intra-procedural Transesophageal Echocardiography (TEE) thus plays a pivotal role in guiding device sizing and ensuring complete occlusion [10].
Discussion
From a clinical standpoint, the concomitant PFA and LAA closure approach underscores the need for a tailored anticoagulation strategy and structured post-procedural imaging follow-up. While early studies suggest that both procedures can be performed safely in a single session, transient LAA dysfunction or SEC may necessitate temporary adjustment of antithrombotic therapy [11]. As this combined strategy becomes more widespread, recognizing and managing electroporation-related tissue responses will be critical to optimizing outcomes and achieving durable rhythm control and stroke prevention.
Left atrial appendage edema following PFA
The LAA ridge, located between the left superior pulmonary vein and the LAA, is a critical anatomic region frequently targeted during pulmonary vein isolation. After PFA, this region often exhibits transient wall thickening and tissue enhancement consistent with interstitial edema [12, 13]. The underlying mechanism differs from that of thermal ablation as electroporation temporarily increases cell membrane permeability in cardiomyocytes and endothelial cells, leading to ionic shifts, osmotic water influx, and localized capillary leakage without inducing coagulative necrosis. Consequently, tissue swelling is observed as T2-hyperintensity on Cardiac Magnetic Resonance (CMR) or as ridge thickening on Transesophageal Echocardiography (TEE) or Intracardiac Echocardiography (ICE). Notably, T2-weighted CMR studies have shown that the extent of edema following PFA is approximately 20% smaller compared with thermal ablation. Because the PFA effect is highly tissue-selective, this edema represents a reversible physiological response rather than permanent injury.
The development of edema after PFA follows a predictable course. Wall thickening and hyperintensity typically become evident within four hours compared to one hour after ablation, and gradually resolve over six to twelve weeks [14, 15]. Imaging studies consistently demonstrate that these changes are temporary and non-fibrotic, contrasting with the outcomes seen in Radiofrequency (RF) or cryothermal ablation. In patients undergoing combined ablation and LAAO, Peri-Device Leak (PDL) occurred in 38.6% of those treated with RF ablation and 34.4% with cryoablation at three-month follow-up [16]. Importantly, no consensus has been reached regarding which ablation modality RF, cryothermal, or the newer PFA is most suitable when performing concomitant Pulmonary Vein Isolation (PVI) and LAA closure.
From a procedural standpoint, the presence of ridge edema can be interpreted as a transient marker of lesion formation following PFA. However, it is important to note that, to date, no studies have demonstrated a direct correlation between the degree of diffuse edema after PFA and ablation adequacy. Evidence supporting such relationships has only been described in the context of thermal ablation modalities, where lesion size and surrounding edema correlate with energy delivery and transmurality [17]. The delivery of pulsed field applications within the Left Superior Pulmonary Vein (LSPV) and in close proximity to the LAA ridge, together with the close anatomic relationship between these structures, may have promoted more pronounced edema in the ridge and LSPV segments directly subjected to ablation.
Functionally, LAA and ridge edema can temporarily reduce appendage compliance, contractility, and flow velocity, manifesting as sluggish flow or spontaneous echo contrast on TEE. Edema at the LAA ridge also carries implications when PFA is performed alongside device-based closure. Swelling can modify ostial geometry, alter device compression, and mimic Peri-Device Leaks (PDL) during immediate post-procedural imaging. Recognition of this reversible phenomenon is crucial to prevent unnecessary device repositioning or oversizing. Additionally, acute inflammation and tissue thickening may theoretically influence early endothelialization over the occluder surface, emphasizing the need for structured follow-up imaging and continued anticoagulation [18]. Overall, LAA edema following PFA represents a benign, self-resolving tissue reaction that reflects the distinctive, non-thermal properties of electroporation and highlights the importance of nuanced interpretation in post-ablation imaging.
Dense spontaneous echo contrast following PFA
Spontaneous Echo Contrast (SEC), often visualized as smoke-like swirling echoes within the cardiac chamber on echocardiography, is a well-recognized marker of low blood-flow velocity and blood stasis. Several pathophysiological mechanisms may contribute to this finding. The most plausible explanation is transient left atrial and LAA stunning after ablation, leading to reduced contractility and diminished LAA emptying velocity [19, 20]. In addition, suboptimal anticoagulation, resulting in an Activated Clotting Time (ACT) below the target range during the procedure, can promote the development of SEC, either intraprocedurally or shortly after PFA. A transient reduction in the ACT, particularly before full heparinization or during the transseptal puncture phase, may promote blood stasis and microthrombus formation within the left atrium and appendage. This highlights the importance of achieving adequate anticoagulation prior to left atrial access and maintaining continuous or minimally interrupted oral anticoagulant therapy in patients on chronic treatment [21, 22]. Current practice recommends initiating unfractionated heparin before or immediately at the time of transseptal puncture to maintain an ACT above 300–350 seconds throughout the ablation procedure, thereby minimizing the risk of dense SEC and thromboembolic complications [23]. Moreover, increasing the intensity of anticoagulation has been shown to reduce the risk of thrombus formation during atrial fibrillation ablation, particularly in patients presenting with pre-existing SEC [24].
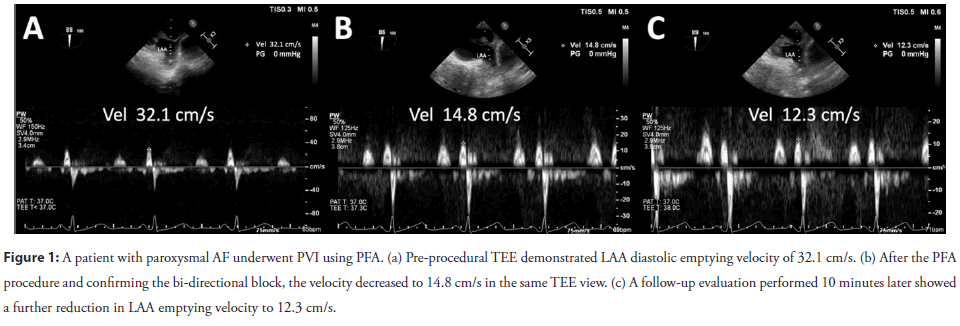
From a hemodynamic perspective, the normal peak LAA emptying velocity typically exceeds 40 cm/s, whereas SEC tends to develop when the velocity falls below approximately 20 cm/s [25, 26]. In the reported case, LAA emptying velocity was observed to decline significantly from 43 cm/s to 23.1 cm/s. Another reported case demonstrated a similar phenomenon, revealing a significant decrease in LAA emptying velocity (Figure 1).
Figure 1: A patient with paroxysmal AF underwent PVI using PFA. (a) Pre-procedural TEE demonstrated LAA diastolic emptying velocity of 32.1 cm/s. (b) After the PFA procedure and confirming the bi-directional block, the velocity decreased to 14.8 cm/s in the same TEE view. (c) A follow-up evaluation performed 10 minutes later showed a further reduction in LAA emptying velocity to 12.3 cm/s.
Clinically, the detection of dense SEC after PFA carries important implications for anticoagulation and procedural interpretation. The associated blood stasis may transiently create a pro-thrombotic milieu, particularly in patients with advanced age, persistent atrial fibrillation, reduced left-ventricular ejection fraction, or enlarged atria. Consequently, therapeutic anticoagulation should be maintained without early interruption until repeat imaging confirms the resolution of SEC and normalization of flow dynamics. If dense SEC is observed immediately post-PFA, deferring device implantation for six to twelve weeks until the edema and SEC resolve may improve safety and reduce thromboembolic risk.
Conclusion
PFA represents a major advancement in atrial fibrillation therapy. When combined with LAAC, it provides an integrated single-session strategy for rhythm control and stroke prevention. However, transient LAA edema and dense SEC may alter appendage geometry, reduce flow velocity, and complicate device implantation or post-procedural imaging interpretation. These reversible findings should be recognized as benign tissue responses rather than complications. Moreover, maintaining adequate anticoagulation is essential to minimize thromboembolic risk. A clear understanding of these transient changes, supported by ongoing research into their temporal evolution, will help refine procedural timing and optimize both safety and efficacy in combined PFA-LAAC interventions—not only to achieve acute procedural success, but also to prevent long-term complications and suboptimal device closure.
References
- Julian Chun KR, MiklavÄiÄ D, Vlachos K, et al. State-of-the-art pulsed field ablation for cardiac arrhythmias: Ongoing evolution and future perspective. Europace. 26(6):euae134 (2024).
- Jiang S, Qian F, Ji S, et al. Pulsed field ablation for atrial fibrillation: Mechanisms, advantages, and limitations. Rev Cardiovasc Med. 25(4):138 (2024).
- Ezzeddine FM, Asirvatham SJ, Nguyen DT, et al. Pulsed field ablation: A comprehensive update. J Clin Med. 13(17):1519 (2024).
- de Campos MCAV, Moraes VRY, Daher RF, et al. Pulsed-field ablation versus thermal ablation for atrial fibrillation: A meta-analysis. Heart Rhythm O2. 5(6):385-395 (2024).
- Mills MT, Calvert P, Velavan P, et al. Concurrent percutaneous left atrial appendage occlusion and catheter ablation for atrial fibrillation: State-of-the-art review. Trends Cardiovasc Med. 34(7):423-433 (2024).
- He B, Jiang LS, Hao ZY, et al. Combination of ablation and left atrial appendage closure as “One-stop” procedure in the treatment of atrial fibrillation: Current status and future perspective. Pacing Clin Electrophysiol. 44(7):1259-1266 (2021).
- Lim J, Hartono B, Gunawan EJ, et al. Left atrial appendage edema and spontaneous echo contrast following pulsed field ablation for pulmonary vein isolation: A case report. Indian Pacing Electrophysiol J. (2025).
- Van Gelder IC, Kotecha D, Rienstra M, et al. 2024 ESC Guidelines for the management of atrial fibrillation developed in collaboration with the European Association for Cardio-Thoracic Surgery (EACTS). Eur Heart J. 45(36):3314-414 (2024).
- Maurhofer J, Kueffer T, Madaffari A, et al. Pulsed-field vs. cryoballoon vs. radiofrequency ablation: A propensity score matched comparison of one-year outcomes after pulmonary vein isolation in patients with paroxysmal atrial fibrillation. J Interv Card Electrophysiol. 67(2):389-397 (2024).
- Garg J, Kabra R, Gopinathannair R, et al. State of the art in left atrial appendage occlusion. JACC Clin Electrophysiol. 11(3):602-641 (2025).
- Beney J, Galea R, Siontis G, et al. Feasibility study on atrial fibrillation ablation with pulsed field ablation and concomitant occlusion of the left atrial appendage. Europace. 26(7):euae176 (2024).
- Nakatani Y, Sridi-Cheniti S, Cheniti G, et al. Pulsed field ablation prevents chronic atrial fibrotic changes and restrictive mechanics after catheter ablation for atrial fibrillation. Europace. 23(11):1767-1776 (2021).
- Saliba W, Delurgio D, Mikaelian B, et al. Combined PVI and LAAC is feasible using RFA or PFA in a canine model despite acute tissue edema in PFA versus RFA. (2024).
- Szegedi N, Gardziejczyk P, Salló Z, et al. Impact of pulsed-field ablation on the left atrial appendage diameter-insights from intra-procedural echocardiography. J Cardiovasc Electrophysiol. (2025).
- Tam MTK, Kojodjojo P, Lam YY, et al. Combined pulsed field ablation and left atrial appendage occlusion: A multicenter comparative study. Heart Rhythm. 22(10):2579-2584 (2025).
- Jiang XH, Tan YJ, Wang RZ, et al. Comparison of prognosis and analysis related risk factors among three different left atrial appendage occlusion procedures in patients with atrial fibrillation. Front Cardiovasc Med. 12:1-11 (2025).
- Hopman LHGA, van Pouderoijen N, Mulder MJ, et al. Atrial ablation lesion evaluation by cardiac magnetic resonance: Review of imaging strategies and histological correlations. JACC Clin Electrophysiol. 9(12):2665-2679 (2023).
- Chen JJ, Chiu FC, Chang SN, et al. Pattern of endothelialization in left atrial appendage occluder by optic coherence tomography: A Pilot Study. J Am Heart Assoc 13(1):e030080 (2024).
- Takami M, Suzuki M, Sugi K, et al. Time course of resolution of left atrial appendage stunning after catheter ablation of chronic atrial flutter. JACC. 41(12):2207-2211 (2003).
- Verhaeghe L, L’Hoyes W, Vijgen J, et al. Left atrial function after atrial fibrillation ablation with pulsed field vs radiofrequency energy: A comparative study. Heart Rhythm. (2025).
- Russo G, Taramasso M, Maisano F, et al. Transseptal puncture: Procedural guidance, challenging situations and management of complications. EuroIntervention. 17(9):720-727 (2021).
- Joglar JA, Chung MK, Armbruster AL, et al. 2023 ACC/AHA/ACCP/HRS guideline for the diagnosis and management of atrial fibrillation: A report of the American College of Cardiology/American Heart Association joint committe on clinical practice guidelines. Circulation. 149:e1-e156 (2024).
- Ma C, Xiao X, Chen Q, et al. Intraprocedural activated clotting time and heparin dosage in pulsed field ablation of paroxysmal atrial fibrillation. Front Cardiovasc Med. (2025).
- Ren JF, Marchlinski FE, Callans DJ, et al. Increased intensity of anticoagulation may reduce risk of thrombus during atrial fibrillation ablation procedures in patients with spontaneous echo contrast. J Cardiovasc Electrophysiol. 16(5):474-477 (2005).
- Bansal M, Kasliwal RR. Echocardiography for left atrial appendage structure and function. Indian Heart J. 64(5):465-475 (2012).
- Ito T, Suwa M. Assessment of left atrial appendage function by echocardiography. Heart Fail Rev. 28:1177-1187 (2023).